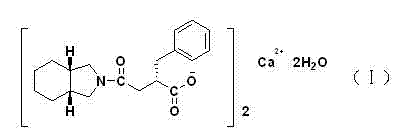
Advanced Manufacturing of High Purity Mitiglinide Calcium for Global Diabetes Care
Advanced Manufacturing of High Purity Mitiglinide Calcium for Global Diabetes Care
The global demand for effective Type 2 diabetes treatments continues to surge, placing immense pressure on the supply chains of key active pharmaceutical ingredients (APIs) like Mitiglinide Calcium (S 21403). As a rapid-acting insulin secretagogue, the clinical efficacy of this drug is directly correlated with its chemical purity and the absence of specific isomeric impurities. A pivotal advancement in this domain is detailed in patent CN103709092A, which outlines a robust, high-purity preparation method that addresses longstanding challenges in regioselectivity and intermediate stability. Unlike traditional routes that struggle with liquid intermediates and expensive catalysts, this innovative process introduces a strategic esterification step that transforms a difficult-to-handle oil into a crystalline solid, thereby revolutionizing the purification landscape for this critical antidiabetic agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Mitiglinide Calcium has been plagued by significant technical and economic bottlenecks that hinder scalable production. Prior art, such as the route described in US6133454A, relies heavily on asymmetric catalysis using rhodium complexes. These transition metal catalysts are not only prohibitively expensive but also present severe challenges in terms of preparation and, crucially, removal from the final product to meet stringent heavy metal specifications. Furthermore, alternative methods reported in patents like CN101492411A utilize hazardous reagents such as thionyl chloride and vinyl chloroformate, which introduce substantial environmental liabilities and safety risks in a manufacturing setting. Perhaps most critically, the key intermediate, (2S)-2-benzyl-3-(cis-hexahydroisoindole-2-carbonyl)propionic acid, exists as a liquid under normal conditions. This physical state makes it exceptionally difficult to purify via crystallization, leading to the carryover of degradation products from the unstable cis-hexahydroisoindoline starting material, ultimately compromising the purity profile of the final API.
The Novel Approach
The methodology disclosed in the referenced patent offers a paradigm shift by decoupling the synthesis from expensive metal catalysis and solving the liquid intermediate problem through a clever "protect-and-purify" strategy. Instead of attempting to purify the oily free acid directly, the process converts the intermediate into its corresponding benzyl ester. This esterification step is the linchpin of the innovation, as it transforms the physical properties of the molecule, allowing it to be isolated as a high-purity crystalline solid. By leveraging standard recrystallization techniques on this solid ester, manufacturers can effectively strip away isomeric impurities and degradation byproducts that would otherwise persist. Following this purification, the benzyl group is cleanly removed under mild acidic conditions to regenerate the pure acid, which is then converted to the calcium salt. This route not only simplifies the operational workflow but also utilizes cost-effective reagents like N,N'-carbonyldiimidazole (CDI) and benzyl chloride, significantly lowering the barrier to entry for high-quality production.
Mechanistic Insights into Esterification-Mediated Purification
The core mechanistic advantage of this synthesis lies in the manipulation of physicochemical properties to drive purification efficiency. The reaction begins with the activation of S-benzyl succinic acid using CDI in an aprotic solvent such as dichloromethane at controlled low temperatures (-15°C to 15°C). This activation generates a reactive acyl imidazole species which subsequently couples with cis-hexahydroisoindoline hydrochloride. The use of the hydrochloride salt of the amine is a critical detail; it mitigates the instability associated with the free amine, preventing the formation of degradation impurities at the source. The resulting amide linkage forms the backbone of the Mitiglinide structure, but the carboxylic acid tail remains a liability for purification until the esterification step occurs.
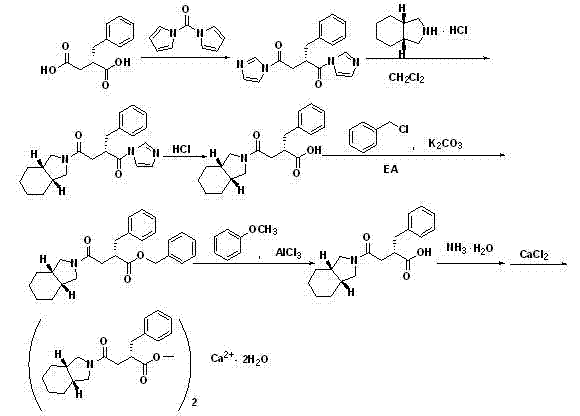
Following the coupling, the process diverges from conventional wisdom by immediately subjecting the crude acid to esterification with benzyl chloride in the presence of potassium carbonate and potassium iodide. This transformation is thermodynamically favorable and results in the formation of the benzyl ester derivative. Mechanistically, this bulky ester group disrupts the molecular symmetry and intermolecular forces in a way that favors lattice formation, allowing the compound to crystallize out of solution upon cooling and the addition of non-solvents like n-hexane. This crystallization acts as a powerful filter, excluding structurally similar impurities that cannot fit into the crystal lattice. Subsequent deprotection using aluminum chloride and sodium phenoxide cleaves the benzyl ester efficiently without racemization, yielding the high-purity free acid required for the final salt formation with calcium chloride. This sequence ensures that the final product meets the rigorous purity standards demanded by regulatory bodies for oral antidiabetic medications.
How to Synthesize Mitiglinide Calcium Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and optical purity. The process is divided into five distinct operational stages: activation and coupling, esterification and crystallization, deprotection, acid recovery, and final salt formation. Each stage builds upon the purity established in the previous step, ensuring that impurities are progressively eliminated rather than carried through to the final API. The use of common solvents like dichloromethane and ethyl acetate facilitates easy solvent recovery and recycling, further enhancing the green chemistry profile of the method. For a detailed breakdown of the specific molar ratios, temperature profiles, and workup procedures required to replicate this high-purity outcome, please refer to the standardized protocol below.
- Activate S-benzyl succinic acid using N,N'-carbonyldiimidazole (CDI) in an aprotic solvent at low temperature.
- Couple the activated acid with cis-hexahydroisoindoline hydrochloride to form the core amide structure.
- Convert the resulting liquid acid intermediate into a crystalline benzyl ester to facilitate purification via recrystallization.
- Remove the benzyl protecting group using aluminum chloride and sodium phenoxide to regenerate the free acid.
- Neutralize the purified acid with ammonia and react with calcium chloride to precipitate the final Mitiglinide Calcium dihydrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route represents a significant opportunity to optimize cost structures and mitigate supply risks. The elimination of noble metal catalysts removes a major variable cost component and eliminates the need for specialized scavenging resins or complex filtration steps required to meet heavy metal limits. Furthermore, the ability to purify via crystallization rather than column chromatography or difficult distillations of heat-sensitive oils translates directly into lower processing costs and higher throughput capabilities. The reliance on stable, commodity-grade reagents like benzyl chloride and CDI ensures that raw material sourcing is robust and less susceptible to the geopolitical volatility often associated with specialized catalytic ligands.
- Cost Reduction in Manufacturing: The substitution of expensive rhodium catalysts with organic coupling reagents results in a drastic reduction in direct material costs. Additionally, the crystallization-based purification strategy minimizes product loss typically associated with chromatographic separations, leading to improved overall mass balance and yield efficiency. The simplified equipment requirements, avoiding the need for high-pressure hydrogenation or specialized metal-handling reactors, further decrease capital expenditure and maintenance overheads for manufacturing facilities.
- Enhanced Supply Chain Reliability: By utilizing cis-hexahydroisoindoline hydrochloride instead of the free base, the process circumvents the stability issues that often lead to batch failures and supply interruptions. The solid nature of the key intermediate ester allows for easier storage and transportation compared to liquid acids, providing greater flexibility in production scheduling and inventory management. This stability ensures a consistent supply of high-quality intermediates, reducing the risk of production delays caused by raw material degradation.
- Scalability and Environmental Compliance: The avoidance of toxic reagents like thionyl chloride aligns the process with increasingly stringent environmental regulations, reducing the burden on waste treatment systems and lowering disposal costs. The use of standard organic solvents and ambient pressure reactions makes the process highly scalable from pilot plant to commercial tonnage without requiring significant re-engineering. This scalability ensures that suppliers can rapidly ramp up production to meet surging market demand for diabetes medications without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Mitiglinide Calcium using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on why this route is superior for industrial application. Understanding these nuances is essential for technical teams evaluating potential manufacturing partners or optimizing existing production lines for better efficiency and compliance.
Q: Why is the benzyl ester intermediate critical for purity?
A: The free acid intermediate is a liquid that is difficult to purify and prone to retaining isomeric impurities. Converting it to a benzyl ester allows it to crystallize, enabling effective removal of impurities through standard recrystallization techniques before the final deprotection step.
Q: How does this method improve upon previous rhodium-catalyzed routes?
A: Previous methods relied on expensive and difficult-to-prepare asymmetric rhodium catalysts. This novel approach eliminates the need for transition metal catalysts entirely, utilizing standard organic coupling reagents like CDI, which drastically reduces raw material costs and simplifies heavy metal removal processes.
Q: What stability advantages does using cis-hexahydroisoindoline hydrochloride offer?
A: Free cis-hexahydroisoindoline is unstable and difficult to preserve, often degrading into impurities that are chemically similar to the product. Using the hydrochloride salt form enhances stability during storage and reaction, preventing the introduction of degradation byproducts into the final API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mitiglinide Calcium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect even trace levels of isomeric impurities, guaranteeing that every batch of Mitiglinide Calcium meets the highest global pharmacopoeia standards.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring your supply chain is built on a foundation of scientific excellence and commercial reliability.
