Advanced Iodine-Catalyzed Synthesis of Pyranocoumarin Derivatives for Commercial Pharmaceutical Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex heterocyclic scaffolds, particularly those exhibiting potent biological activities such as anticoagulant and antiviral properties. Patent CN101613355A introduces a groundbreaking synthetic route for pyranocoumarin derivatives, utilizing a dual-catalytic system involving molecular iodine and concentrated sulfuric acid. This innovation addresses critical bottlenecks in traditional synthesis by replacing expensive reagents with readily available commodity chemicals, thereby streamlining the production workflow. The method operates under relatively mild thermal conditions, eliminating the need for rigorous air-free techniques that often complicate large-scale operations. By leveraging the unique electrophilic activation capabilities of iodine followed by acid-mediated cyclization, this process achieves high structural fidelity. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more economically viable and operationally simple manufacturing protocols for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyranocoumarin core has relied on methodologies that impose significant economic and operational burdens on manufacturers. Prior art, such as the methods reported in Tetrahedron (2006) and Tetrahedron Letters (2007), frequently necessitates the use of specialized and costly reagents like isocyanides, diethyl butynedioate, or chiral organocatalysts such as (S)-proline. These reagents not only inflate the raw material costs but also introduce complex purification challenges, often requiring extensive chromatographic separation to remove trace impurities that could compromise the safety profile of the final API. Furthermore, many conventional routes involve multi-step sequences with stringent environmental controls, such as inert atmosphere protection, which increases capital expenditure on equipment and operational overhead. The reliance on precious metal catalysts in some alternative pathways further exacerbates the cost structure and introduces regulatory hurdles regarding heavy metal residues in the final drug substance.
The Novel Approach
In stark contrast, the methodology disclosed in CN101613355A offers a streamlined, one-pot strategy that drastically simplifies the synthetic landscape. By employing 4-hydroxycoumarin and 1,3-diaryl-2-propyn-1-ol as starting materials, the process utilizes molecular iodine as a primary catalyst to initiate the alkylation phase, followed by concentrated sulfuric acid to drive the cyclization. This sequential addition allows for precise control over reaction kinetics without the need for exotic reagents. The reaction proceeds efficiently in common organic solvents like nitromethane or nitroethane, and crucially, it does not require isolation from air, significantly reducing engineering controls. This approach not only lowers the barrier to entry for production but also enhances the overall atom economy of the transformation. The visual representation of this efficient transformation highlights the direct conversion of simple precursors into the complex fused ring system.
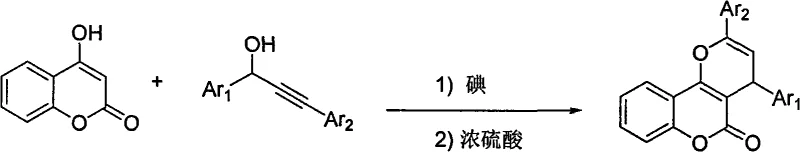
Mechanistic Insights into Iodine and Sulfuric Acid Dual Catalysis
The mechanistic elegance of this synthesis lies in the synergistic action of the two catalysts, which orchestrate distinct phases of the molecular assembly. Initially, molecular iodine acts as a mild Lewis acid, activating the hydroxyl group of the 1,3-diaryl-2-propyn-1-ol to generate a reactive propargylic cation species. This electrophilic intermediate is then attacked by the nucleophilic carbon at the 3-position of the 4-hydroxycoumarin ring, forming a C-C bond in a highly regioselective manner. The use of iodine is particularly advantageous as it avoids the harsh oxidative conditions associated with other halogen sources, thereby preserving the integrity of the sensitive coumarin lactone ring. Following this alkylation step, the introduction of concentrated sulfuric acid shifts the reaction pathway towards intramolecular cyclization. The strong Brønsted acidity protonates the alkyne moiety, facilitating a 6-endo-dig cyclization that closes the pyran ring, followed by dehydration to restore aromaticity or establish the conjugated system.
From an impurity control perspective, this dual-catalyst system offers superior selectivity compared to single-catalyst systems. The moderate temperature range of 50°C to 80°C during the iodine-catalyzed phase prevents thermal degradation of the starting materials, while the subsequent heating to 80°C to 100°C is carefully timed (8 to 12 hours) to ensure complete conversion without promoting polymerization of the alkyne substrate. The quenching step using saturated sodium sulfite solution effectively reduces any residual iodine to water-soluble iodide salts, ensuring that the final organic extract is free from colored iodine impurities that could interfere with downstream crystallization. This meticulous control over reaction parameters results in a crude product profile that is significantly cleaner than those obtained via traditional methods, reducing the load on purification columns and improving overall yield consistency across different batches.
How to Synthesize Pyranocoumarin Derivatives Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The protocol begins with the dissolution of stoichiometric amounts of 4-hydroxycoumarin and the propargylic alcohol in a polar aprotic solvent, establishing a homogeneous reaction mixture. The addition of catalytic iodine initiates the first stage of the transformation, which is monitored to ensure the formation of the alkylated intermediate before proceeding. Once the initial coupling is complete, the careful addition of concentrated sulfuric acid triggers the cyclization cascade. The reaction mixture is then subjected to elevated temperatures for an extended period to drive the equilibrium towards the desired tricyclic product. Upon completion, a straightforward aqueous workup involving sodium sulfite and ethyl acetate extraction isolates the crude material, which can be further purified via standard column chromatography to achieve pharmaceutical-grade purity. Detailed standardized operating procedures for this synthesis are provided below.
- Dissolve 4-hydroxycoumarin and 1,3-diaryl-2-propyn-1-ol in nitromethane or nitroethane with a molar ratio of 1: 1.0 to 1.2.
- Add molecular iodine (0.08-0.20 equivalents) and react at 50-80°C for 1 hour to facilitate alkylation.
- Introduce concentrated sulfuric acid and stir at 80-100°C for 8-12 hours to complete cyclization, followed by standard aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iodine-catalyzed methodology presents a compelling value proposition centered on cost efficiency and supply security. The replacement of expensive, specialty reagents with commodity chemicals like molecular iodine and sulfuric acid fundamentally alters the cost structure of the manufacturing process. Since these catalysts are produced on a massive global scale, their pricing is stable and immune to the volatility often seen with niche organocatalysts or precious metals. Furthermore, the elimination of air-sensitive conditions means that existing standard reactor infrastructure can be utilized without the need for costly upgrades to glovebox lines or specialized inert gas manifolds. This compatibility with standard equipment accelerates the timeline for technology transfer from R&D to commercial production, allowing companies to bring products to market faster.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic reduction in raw material expenses. By utilizing non-metallic iodine and sulfuric acid instead of palladium or rhodium-based catalysts, the direct material cost per kilogram of product is significantly lowered. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps required for transition metal catalysis, reduces the consumption of auxiliary processing agents and waste disposal costs. The high atom economy of the one-pot design minimizes solvent usage and energy consumption associated with intermediate isolation, leading to substantial overall operational savings. These factors combine to create a highly competitive cost position for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. 4-Hydroxycoumarin and substituted propargylic alcohols are commercially accessible from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate ambient air and moisture to a reasonable degree, reduces the likelihood of batch failures due to environmental excursions. This reliability ensures consistent output volumes, allowing supply chain planners to forecast production schedules with greater confidence. The stability of the reagents also simplifies logistics and storage requirements, as there is no need for cryogenic shipping or specialized containment for air-sensitive compounds.
- Scalability and Environmental Compliance: Scaling this process from gram to ton quantities is straightforward due to the absence of exothermic hazards typically associated with organometallic reagents. The reaction exotherm is manageable within standard jacketed reactors, and the use of common solvents like nitromethane facilitates established recovery and recycling protocols. From an environmental standpoint, the avoidance of heavy metals aligns with increasingly stringent regulatory standards for pharmaceutical manufacturing, reducing the burden of wastewater treatment and hazardous waste disposal. The process generates minimal toxic byproducts, supporting corporate sustainability goals and simplifying the regulatory filing process for new drug applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and advantageous effects detailed in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provide clarity on catalyst loading, reaction times, and purification strategies to ensure successful adoption.
Q: What are the primary advantages of using molecular iodine over traditional catalysts for pyranocoumarin synthesis?
A: Molecular iodine serves as a cost-effective, non-metallic Lewis acid that activates propargylic alcohols efficiently without the need for expensive transition metals or complex organocatalysts, significantly reducing raw material costs and simplifying metal removal processes.
Q: How does the two-stage temperature protocol improve product purity?
A: The initial lower temperature phase (50-80°C) ensures selective alkylation, while the subsequent higher temperature phase (80-100°C) with sulfuric acid drives the cyclization to completion, minimizing side reactions and polymerization often seen in single-step high-temperature processes.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process utilizes common solvents like nitromethane and standard reagents like concentrated sulfuric acid, avoiding air-sensitive conditions or cryogenic temperatures, which makes it highly amenable to large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyranocoumarin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iodine-catalyzed synthesis described in CN101613355A for the production of high-value pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped to handle the specific thermal and corrosive requirements of sulfuric acid-mediated reactions safely and efficiently. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the absence of iodine residues and ensure the structural integrity of the final pyranocoumarin derivatives.
We invite pharmaceutical partners to leverage our technical expertise to optimize this synthesis for their specific pipeline needs. Our team is prepared to conduct a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this route can improve your margin structure. We encourage you to contact our technical procurement team to request specific COA data from our pilot runs and comprehensive route feasibility assessments. By collaborating with us, you gain access to a secure, scalable, and cost-effective supply chain for complex heterocyclic intermediates, positioning your organization for success in a competitive global market.
