Advanced Gold-Catalyzed Synthesis of Pyranocoumarin Derivatives for Commercial Scale-Up
Advanced Gold-Catalyzed Synthesis of Pyranocoumarin Derivatives for Commercial Scale-Up
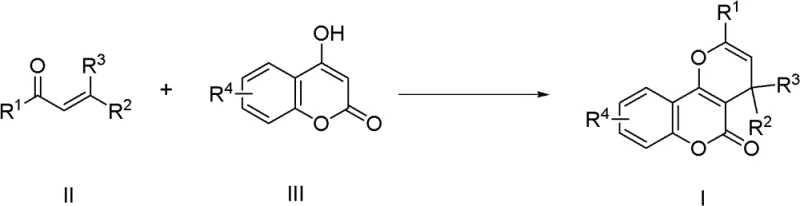
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic methodologies that balance high efficiency with environmental sustainability. Patent CN102336763B introduces a groundbreaking approach to synthesizing pyranocoumarin derivatives, a class of compounds renowned for their potent biological activities including antimalarial, antibacterial, and anti-HIV properties. This technology leverages a sophisticated gold-catalyzed tandem reaction between alpha,beta-unsaturated ketones and 4-hydroxycoumarin compounds, marking a significant departure from conventional acidic conditions. For R&D directors and procurement specialists, this patent represents a pivotal opportunity to access high-purity pharmaceutical intermediates through a streamlined, atom-economical process that minimizes waste and maximizes yield. The versatility of this method allows for the introduction of diverse substituents, enabling the rapid generation of compound libraries for drug discovery programs while maintaining a pathway viable for industrial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functional pyranocoumarins has relied heavily on harsh acidic conditions, such as sulfuric acid or iodine in acetic acid, to facilitate the cyclization of precursors. These traditional routes suffer from significant drawbacks that hinder their applicability in modern green chemistry frameworks. The primary issue lies in the notoriously low reaction yields, which directly impact the cost-efficiency and material throughput required for commercial production. Furthermore, the use of strong mineral acids generates substantial amounts of corrosive waste, posing severe environmental challenges and increasing the operational costs associated with waste treatment and disposal. Additionally, alternative oxidative cross-coupling methods using DDQ intermediates often require excessive catalyst loading and exhibit poor regioselectivity, particularly when dealing with complex 1,3-dipropenylarene substrates. These limitations create a bottleneck for supply chain managers who require consistent, high-volume delivery of key intermediates without the burden of complex purification protocols or regulatory hurdles associated with hazardous reagents.
The Novel Approach
In stark contrast to these legacy methods, the novel synthesis route disclosed in the patent utilizes a transition metal catalyst system, specifically highlighting the efficacy of gold-based complexes like AuCl3 combined with silver salts such as AgOTf. This innovative approach operates under much milder conditions, typically in organic solvents like toluene at temperatures ranging from 80°C to 100°C. The mechanism involves a cascade of carbon-hydrogen bond activation, deprotonation, and intramolecular cyclization, which proceeds with exceptional atom economy. By shifting away from stoichiometric acid promoters to a catalytic cycle, the process drastically reduces the generation of chemical waste. The simplicity of the operation is another key advantage; the reaction mixture can be directly subjected to standard purification techniques like silica gel column chromatography after solvent removal. This streamlined workflow not only enhances the overall yield, with some examples demonstrating yields as high as 98%, but also simplifies the technical requirements for plant operators, thereby reducing the risk of batch-to-batch variability in a manufacturing setting.
Mechanistic Insights into AuCl3-Catalyzed Cyclization
The core of this technological advancement lies in the unique ability of the gold catalyst to activate the unsaturated ketone substrate towards nucleophilic attack by the 4-hydroxycoumarin. The preferred catalytic system, comprising AuCl3 and AgOTf in a 1:3 molar ratio, generates a highly active cationic gold species in situ. This electrophilic metal center coordinates with the alkene moiety of the alpha,beta-unsaturated ketone, increasing its susceptibility to nucleophilic addition. Following the initial Michael-type addition, the intermediate undergoes a series of proton transfers and cyclization steps to form the fused pyran ring system characteristic of the target derivatives. The presence of the silver salt serves to abstract the chloride ligand from the gold precursor, enhancing the Lewis acidity of the metal center and driving the catalytic turnover. This precise control over the reaction pathway ensures high regioselectivity, preventing the formation of unwanted isomers that often plague non-catalytic thermal reactions. Understanding this mechanistic nuance is crucial for process chemists aiming to optimize reaction parameters for specific substrate classes, ensuring that the electronic and steric properties of substituents on the aromatic rings do not impede the catalytic cycle.
Furthermore, the impurity profile of the final product is significantly improved due to the specificity of the gold-catalyzed transformation. In traditional acid-catalyzed processes, side reactions such as polymerization of the unsaturated ketone or degradation of the coumarin scaffold are common, leading to complex mixtures that are difficult to separate. The mild nature of the gold catalysis mitigates these decomposition pathways, preserving the integrity of sensitive functional groups such as halogens, methoxy, or nitro groups present on the aromatic rings. This tolerance for diverse functional groups expands the chemical space accessible to medicinal chemists, allowing for the synthesis of analogs with tailored pharmacokinetic properties. For quality control teams, this translates to a cleaner crude reaction mass, which simplifies the analytical validation process and ensures that the final API intermediate meets stringent purity specifications required by regulatory bodies. The ability to consistently produce high-quality material with minimal byproduct formation is a critical factor in reducing the overall cost of goods and accelerating the timeline from bench-scale discovery to pilot plant production.
How to Synthesize Pyranocoumarin Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the preparation of the catalytic system and the control of reaction parameters. The protocol generally involves pre-mixing the gold and silver salts in the chosen solvent, typically toluene, to ensure the active catalyst species is fully generated before the addition of substrates. The molar ratio of the 4-hydroxycoumarin to the alpha,beta-unsaturated ketone is optimized between 1:1.0 and 1:1.2 to drive the reaction to completion while minimizing excess reagent waste. Maintaining the reaction temperature within the 80°C to 100°C window is essential for achieving optimal kinetics without triggering thermal degradation. Detailed standardized operating procedures for this synthesis, including specific workup and purification steps, are outlined in the technical documentation below to ensure reproducibility and safety during scale-up operations.
- Prepare the catalytic system by mixing AuCl3 and AgOTf in toluene solvent under magnetic stirring at room temperature.
- Dissolve the alpha,beta-unsaturated ketone and 4-hydroxycoumarin substrates in toluene and add to the catalyst mixture.
- Heat the reaction mixture to 80-100°C for 6-8 hours, then purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this gold-catalyzed methodology offers profound advantages for procurement managers and supply chain heads focused on cost reduction and reliability. The shift from hazardous acid reagents to a reusable or low-loading metal catalyst system fundamentally alters the cost structure of manufacturing these valuable intermediates. By eliminating the need for expensive and dangerous acid handling infrastructure, facilities can reduce their capital expenditure on corrosion-resistant equipment and waste neutralization systems. Moreover, the high atom economy of the reaction means that a greater proportion of the raw material mass is converted into the desired product, directly lowering the material cost per kilogram of output. This efficiency is compounded by the simplified downstream processing; the ability to purify the product via straightforward silica gel chromatography rather than complex crystallization or distillation sequences reduces both energy consumption and labor hours. These factors collectively contribute to a more resilient supply chain capable of meeting fluctuating market demands without compromising on margin or delivery timelines.
- Cost Reduction in Manufacturing: The implementation of this catalytic route eliminates the reliance on stoichiometric amounts of strong acids, which are not only costly to purchase but also expensive to dispose of in compliance with environmental regulations. The use of a highly active gold catalyst at low loading levels (2.5-5.0 mol%) ensures that the catalyst cost does not offset the savings gained from improved yields. Additionally, the simplified workup procedure reduces the consumption of solvents and stationary phases during purification, further driving down the variable costs associated with production. This economic efficiency makes the process highly competitive for the manufacturing of high-purity pharmaceutical intermediates, allowing suppliers to offer more attractive pricing models to their downstream clients while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically alpha,beta-unsaturated ketones and 4-hydroxycoumarins, are commercially available and can be sourced from multiple established vendors, reducing the risk of supply bottlenecks. The robustness of the reaction conditions, which tolerate a wide range of substituents and moisture levels better than sensitive organometallic alternatives, ensures consistent batch quality even when scaling up to multi-kilogram quantities. This reliability is critical for long-term supply agreements where continuity of supply is paramount. By adopting a process that is less prone to failure due to minor variations in raw material quality or environmental conditions, manufacturers can guarantee shorter lead times and more predictable delivery schedules for their customers, strengthening their position as a reliable partner in the global pharmaceutical supply chain.
- Scalability and Environmental Compliance: As regulatory pressure mounts on the chemical industry to adopt greener practices, this synthesis route positions manufacturers favorably by minimizing the generation of hazardous waste. The use of toluene, a common industrial solvent with well-established recovery and recycling protocols, aligns with current sustainability goals. The high selectivity of the reaction reduces the formation of toxic byproducts, simplifying the effluent treatment process and lowering the environmental footprint of the facility. This compliance advantage is increasingly becoming a deciding factor for multinational corporations when selecting suppliers, as it helps them meet their own corporate social responsibility targets. Furthermore, the scalability of the process from gram to ton scale has been demonstrated through the broad scope of substrates tested, confirming its viability for commercial scale-up of complex pharmaceutical intermediates without the need for specialized high-pressure or cryogenic equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios or for procurement officers assessing the quality standards of potential suppliers.
Q: What are the primary advantages of the gold-catalyzed method over traditional acid catalysis?
A: The gold-catalyzed method described in CN102336763B offers significantly higher reaction yields and superior regioselectivity compared to traditional sulfuric or iodine-catalyzed methods, while avoiding the environmental pollution associated with strong acid waste streams.
Q: Is this synthesis route suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process utilizes readily available raw materials and common organic solvents like toluene, with a simple workup procedure involving silica gel adsorption and distillation, making it highly amenable to commercial scale-up for API intermediate production.
Q: What is the typical purity profile of the resulting pyranocoumarin derivatives?
A: The method demonstrates high atom economy and selectivity, producing white or pale yellow solids with high purity after standard column chromatography purification, suitable for downstream biological testing and drug development applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyranocoumarin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic methodologies like the one described in CN102336763B for the development of next-generation therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity pyranocoumarin derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex gold-catalyzed reactions allows us to offer customized synthesis solutions that optimize both cost and quality for our global partners.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage potential partners to reach out for specific COA data and route feasibility assessments to understand how our manufacturing capabilities can support your supply chain objectives. Let us be your trusted partner in bringing high-value pharmaceutical intermediates to market with speed, precision, and economic efficiency.
