Advanced Biocatalytic Synthesis of Chiral Bis-Aryl Alcohols for Commercial Pharmaceutical Manufacturing
Advanced Biocatalytic Synthesis of Chiral Bis-Aryl Alcohols for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for producing chiral intermediates, particularly for antihistamine drugs like carbinoxamine and bepotastine besilate. Patent CN109837317B introduces a groundbreaking synthetic method for chiral bis-aryl alcohol compounds, specifically focusing on the production of (S)-(4-chlorophenyl)pyridine-2-methanol. This technology leverages engineered mutants of carbonyl reductase SSCR derived from Sporobolomyces Salmonicolor and alcohol dehydrogenase TbSADH from Thermoanaerobacter brockii. By utilizing these highly specific biocatalysts, the process achieves asymmetric reduction of prochiral ketones with unprecedented efficiency. The innovation lies not just in the enzymes themselves, but in the strategic mutation of their catalytic pockets to enhance both substrate conversion and stereocontrol. For procurement and R&D leaders, this represents a shift away from hazardous chemical synthesis towards a greener, high-precision biological manufacturing platform that ensures consistent quality and regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of (S)-(4-chlorophenyl)pyridine-2-methanol has relied heavily on chemical asymmetric reduction methods that pose significant operational and environmental challenges. Conventional routes often involve the use of 2-cyanopyridine as a starting material, requiring a multi-step synthesis involving 4-chlorophenylmagnesium bromide and concentrated sulfuric acid. These processes typically necessitate the use of expensive and toxic transition metal catalysts such as Palladium complexes, for example Pd(Phe3P)4, or Noyori organometallic catalysts based on Ruthenium and Rhodium. Furthermore, the presence of the nitrogen atom in the pyridine ring often mandates complex protection and deprotection strategies to prevent catalyst poisoning or side reactions, which drastically reduces overall atom economy. The requirement for high-pressure hydrogenation (8-10 bar) and harsh acidic conditions increases the capital expenditure for specialized reactor equipment and imposes rigorous safety protocols. Additionally, the removal of trace heavy metal residues from the final product to meet pharmaceutical standards adds substantial cost and complexity to the downstream purification process, creating a bottleneck for scalable manufacturing.
The Novel Approach
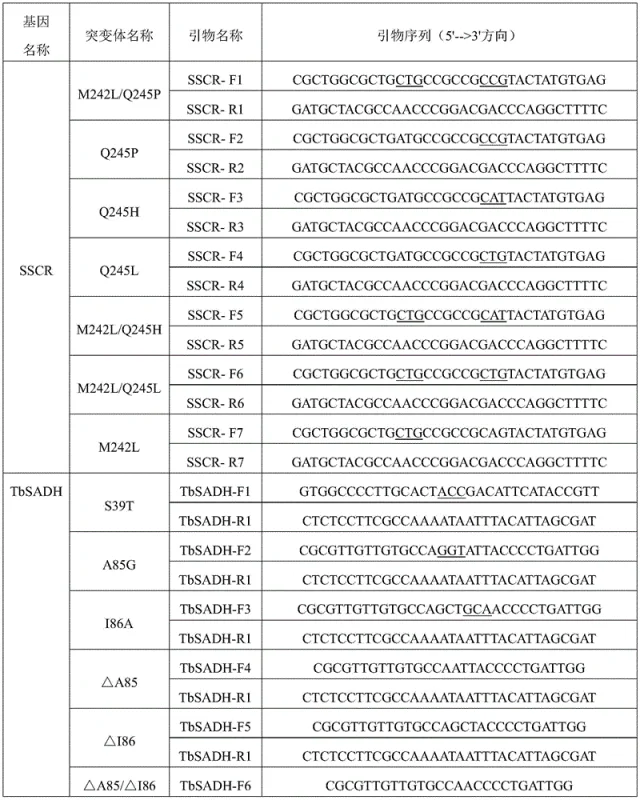
In stark contrast, the novel biocatalytic approach described in the patent utilizes engineered alcohol dehydrogenases to catalyze the direct asymmetric reduction of (4-chlorophenyl)pyridine-2-methanone. This method operates under mild physiological conditions, typically at 30°C and pH 7.0 in a phosphate buffer, eliminating the need for high pressure, extreme temperatures, or corrosive reagents. The core innovation involves specific mutations at key amino acid residues within the enzyme's catalytic center, such as positions 242 and 245 for SSCR or positions 85, 86, and 198 for TbSADH. These modifications optimize the steric environment of the active site, allowing for precise discrimination between enantiomers and resulting in exceptionally high enantiomeric excess (ee) values, often exceeding 99%. Moreover, the process employs isopropanol as a hydrogen donor for cofactor regeneration, which simplifies the reaction system by removing the need for auxiliary enzymes like glucose dehydrogenase. This streamlined approach not only enhances the sustainability profile of the manufacturing process but also significantly reduces the complexity of the supply chain by relying on renewable biological catalysts rather than scarce precious metals.
Mechanistic Insights into Enzyme-Catalyzed Asymmetric Reduction
The success of this synthetic route hinges on the precise engineering of the enzyme's active site to accommodate the bulky bis-aryl ketone substrate while enforcing strict stereochemical control. The carbonyl reductase SSCR and alcohol dehydrogenase TbSADH function by facilitating the transfer of a hydride ion from the reduced cofactor (NADPH or NADH) to the carbonyl carbon of the substrate. In the wild-type enzymes, the catalytic pocket may be too restrictive or lack the necessary interactions to stabilize the transition state for this specific substrate, leading to low conversion or poor stereoselectivity. However, through site-directed mutagenesis, specific residues lining the catalytic pocket are altered. For instance, mutating Glutamine to Proline at position 245 (Q245P) in SSCR alters the hydrophobicity and spatial arrangement near the binding site. Similarly, mutations in TbSADH, such as A85G/I86A, reshape the small pocket of the catalytic center to better fit the 4-chlorophenyl group. These structural adjustments lower the activation energy for the desired (S)-enantiomer pathway while sterically hindering the formation of the (R)-enantiomer. The result is a highly efficient catalytic cycle where the enzyme repeatedly turns over the substrate with minimal byproduct formation.
Furthermore, the mechanism includes an integrated cofactor regeneration system that is critical for industrial viability. In typical biocatalytic reductions, the stoichiometric consumption of expensive cofactors like NADP+ would be prohibitive. This patent solves that issue by coupling the reduction reaction with the oxidation of isopropanol to acetone. The enzyme catalyzes the oxidation of isopropanol, regenerating the reduced cofactor (NADPH) needed for the ketone reduction in situ. This creates a closed loop where only a catalytic amount of cofactor is required, drastically reducing raw material costs. The stability of the mutant enzymes under reaction conditions (20-35°C) ensures that they remain active throughout the 24-hour reaction period, maintaining high reaction rates without significant denaturation. This robustness is essential for maintaining consistent product quality in large-scale batches, ensuring that the impurity profile remains tightly controlled and predictable, which is a key concern for regulatory approval of pharmaceutical intermediates.
How to Synthesize (S)-(4-chlorophenyl)pyridine-2-methanol Efficiently
The implementation of this biocatalytic route requires a structured approach to enzyme expression and reaction optimization to ensure maximum yield and purity. The process begins with the transformation of host cells, typically E. coli BL21(DE3), with recombinant plasmids carrying the mutated enzyme genes. Following induction with IPTG, the biomass is harvested and processed into a usable biocatalyst form, such as crude enzyme powder or whole cells. The reaction setup involves dissolving the prochiral ketone substrate in a buffered aqueous system containing the biocatalyst and isopropanol. Careful control of pH and temperature is maintained to preserve enzyme activity. While the general framework is straightforward, specific parameters such as enzyme loading and substrate concentration must be optimized for the specific mutant strain used. For a detailed, step-by-step technical guide on the exact fermentation, lysis, and catalytic conditions validated in the patent examples, please refer to the standardized protocol below.
- Express the engineered alcohol dehydrogenase (SSCR or TbSADH mutant) in E. coli BL21(DE3) host cells and prepare crude enzyme powder or whole cells.
- Prepare the reaction system containing the substrate (4-chlorophenyl)pyridine-2-methanone, the biocatalyst, phosphate buffer (pH 7.0), and isopropanol as a hydrogen donor.
- Incubate the reaction mixture at 30°C for approximately 24 hours to achieve high conversion and enantiomeric excess, followed by extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic technology offers transformative benefits that extend beyond simple yield improvements. The shift from chemical to biological catalysis fundamentally alters the cost structure and risk profile of the manufacturing process. By eliminating the reliance on precious metal catalysts like Ruthenium and Palladium, manufacturers can insulate themselves from the volatile pricing and geopolitical supply risks associated with these commodities. Furthermore, the mild reaction conditions reduce the energy consumption required for heating, cooling, and pressurization, leading to substantial operational expenditure savings. The simplicity of the work-up procedure, which avoids complex protection groups and heavy metal scavenging steps, shortens the production cycle time and increases the throughput of existing manufacturing facilities. This efficiency gain allows for more flexible response to market demand fluctuations without the need for significant capital investment in new infrastructure.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the drastic simplification of the synthesis route. Traditional chemical methods incur high costs due to the purchase of expensive organometallic catalysts and the subsequent need for rigorous purification to remove metal traces to ppm levels. In this biocatalytic process, the catalyst is a renewable protein produced via fermentation, which is inherently cheaper and more sustainable. The use of isopropanol as a cheap hydrogen donor eliminates the need for costly auxiliary enzymes or external cofactor addition. Additionally, the high stereoselectivity means that less material is wasted on the unwanted enantiomer, improving the overall mass balance and reducing the cost of goods sold (COGS). The removal of protection and deprotection steps further cuts down on reagent costs and waste disposal fees, creating a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the robust nature of the biological materials involved. Unlike sensitive organometallic complexes that may require special storage conditions or have limited shelf lives, the engineered enzyme strains can be stored as stable glycerol stocks or lyophilized powders. The raw materials for the reaction, such as the ketone substrate and isopropanol, are commodity chemicals with reliable global availability. This reduces the risk of production stoppages due to raw material shortages. Moreover, the process does not require high-pressure hydrogen gas, removing a significant safety hazard and logistical burden associated with transporting and storing hazardous gases. This makes the technology suitable for deployment in a wider range of manufacturing sites, diversifying the supply base and reducing dependency on single-source specialized facilities.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is generally more straightforward than scaling high-pressure chemical hydrogenations. The reaction operates at atmospheric pressure and moderate temperatures, meaning standard stainless steel reactors can be used without the need for specialized high-pressure vessels. This lowers the barrier to scale-up from pilot to commercial production. From an environmental perspective, the process aligns perfectly with green chemistry principles. It generates less hazardous waste, avoids toxic heavy metals, and uses water as the primary solvent. This simplifies wastewater treatment and reduces the environmental footprint of the facility. Meeting stringent environmental regulations becomes easier and less costly, avoiding potential fines and reputational damage. The high atom economy of the direct reduction also means less raw material is converted into waste, supporting corporate sustainability goals and enhancing the brand value of the final pharmaceutical product.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the patent's capabilities and limitations. These insights are derived directly from the experimental data and technical specifications outlined in the intellectual property documentation. Understanding these nuances is critical for integrating this biocatalytic route into existing development pipelines. The following responses address key concerns regarding performance metrics, operational parameters, and comparative advantages over legacy methods.
Q: What are the primary advantages of this enzymatic method over traditional chemical synthesis?
A: Unlike traditional chemical methods that require heavy metal catalysts like Palladium or Ruthenium and harsh conditions such as high pressure and concentrated sulfuric acid, this biocatalytic approach operates under mild physiological conditions (30°C, pH 7.0). It eliminates the need for complex protection and deprotection steps on the pyridine ring and avoids toxic heavy metal residues, significantly simplifying downstream purification and environmental compliance.
Q: How does the patented method address cofactor regeneration costs?
A: The method utilizes isopropanol as a hydrogen donor for cofactor regeneration, which removes the necessity of adding additional auxiliary enzymes like glucose dehydrogenase (GDH) often required in other biocatalytic systems. This simplification reduces the overall enzyme loading cost and streamlines the reaction components, making the process more economically viable for large-scale manufacturing.
Q: What level of stereoselectivity can be achieved with these enzyme mutants?
A: Through specific site-directed mutagenesis of key amino acid residues in the catalytic pocket, the engineered SSCR and TbSADH mutants achieve exceptional stereoselectivity. Specific mutants have demonstrated enantiomeric excess (ee) values exceeding 99.99% and conversion rates greater than 99%, meeting the stringent purity requirements for active pharmaceutical ingredient (API) intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-(4-chlorophenyl)pyridine-2-methanol Supplier
The technological potential of enzyme-engineered synthesis for chiral bis-aryl alcohols is immense, offering a pathway to high-purity intermediates that define the quality of modern antihistamine medications. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are successfully translated into robust industrial realities. Our commitment to stringent purity specifications and operation of rigorous QC labs guarantees that every batch meets the exacting standards required by global regulatory bodies. We understand that consistency is key in pharmaceutical supply, and our advanced biocatalytic capabilities allow us to deliver products with superior enantiomeric excess and minimal impurity profiles consistently.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how this biocatalytic approach can optimize your supply chain. Let us partner with you to drive innovation and efficiency in the production of high-value pharmaceutical intermediates.
