Advanced Biocatalytic Synthesis of Sitagliptin Intermediates via Engineered Transaminase Mutants
The pharmaceutical industry continuously seeks robust biocatalytic solutions to enhance the efficiency of Active Pharmaceutical Ingredient (API) synthesis, particularly for blockbuster drugs like Sitagliptin. Patent CN115975969A introduces a groundbreaking advancement in this domain by disclosing a series of engineered transaminase variants specifically designed to catalyze the asymmetric amination of morpholinedione derivatives. This technology addresses the critical bottleneck of low conversion rates often encountered in earlier enzymatic generations, providing a pathway to produce (R)-3-amino-1-morpholine-4-(2,4,5-trifluorophenyl)-1-butanone with exceptional stereoselectivity. By leveraging site-directed mutagenesis at key residue positions 150, 152, and 155, the disclosed enzymes achieve substantial improvements in catalytic turnover and stability, positioning this innovation as a vital asset for any organization aiming to optimize their supply chain for DPP-4 inhibitor manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Sitagliptin intermediates have historically relied on chemical asymmetric synthesis or early-generation biocatalysts that suffered from suboptimal performance metrics. Prior art, such as that referenced in WO2019011236A1, demonstrated the feasibility of using transaminases but highlighted a persistent challenge: the enzyme activity and conversion rates were insufficient for cost-effective industrial application without extensive process optimization. Conventional enzymes often exhibited poor stability under process conditions, necessitating high enzyme loading which drastically inflates production costs. Furthermore, lower conversion rates result in significant amounts of unreacted starting material, complicating downstream purification and increasing solvent consumption. These inefficiencies create a substantial burden on procurement budgets and extend lead times, making the search for a high-performance biocatalyst a strategic priority for pharmaceutical manufacturers seeking to maintain competitive margins in a crowded market.
The Novel Approach
The novel approach detailed in the patent data overcomes these historical limitations through precise protein engineering focused on the active site and substrate binding pocket of the transaminase. By introducing specific amino acid substitutions at residues 150, 152, and 155 relative to the parent sequence SEQ ID NO: 1, the inventors have created mutants that exhibit markedly superior catalytic properties. For instance, specific variants like Enz.27 (S150W-C152T-Q155K) have demonstrated conversion rates reaching approximately 58%, a significant leap from the roughly 33% observed in the baseline Enz.1. This enhancement is not merely incremental; it represents a fundamental shift in process viability, allowing for higher substrate concentrations and reduced reaction times. The ability to achieve such high conversion while maintaining >99% enantiomeric excess ensures that the resulting intermediate meets the rigorous purity standards required for final API formulation, thereby streamlining the entire manufacturing workflow.
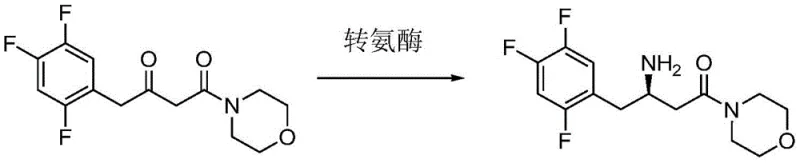
Mechanistic Insights into Site-Directed Mutagenesis of Transaminase
The mechanistic basis for the improved performance lies in the strategic alteration of the enzyme's tertiary structure to better accommodate the bulky 2,4,5-trifluorophenyl moiety of the substrate. The mutations at position 150, such as substituting Serine with Tryptophan (S150W), likely introduce favorable pi-stacking interactions or hydrophobic packing that stabilize the transition state during the amine transfer. Similarly, modifications at position 152, like changing Cysteine to Threonine or Leucine, may optimize the geometry of the binding pocket, reducing steric hindrance and facilitating faster product release. These structural refinements collectively enhance the turnover number (kcat) and potentially improve the Michaelis constant (Km) for the ketone substrate. From a process chemistry perspective, this means the enzyme operates more efficiently at lower loadings, directly translating to reduced raw material costs for the biocatalyst itself. The retention of high stereoselectivity (>99% ee) across all tested mutants indicates that these changes do not compromise the chiral integrity of the active site, a critical factor for regulatory compliance in API synthesis.
Furthermore, the stability of these engineered enzymes under operational conditions is a key differentiator that supports robust manufacturing. The patent data indicates that the mutants retain activity in the presence of organic co-solvents like isopropanol, which is essential for solubilizing the hydrophobic morpholinedione substrate. The use of isopropylamine hydrochloride as the amino donor drives the equilibrium towards product formation, and the enhanced tolerance of the mutants to these reaction components prevents premature deactivation. This resilience allows for longer batch cycles or even potential continuous flow applications, maximizing the utility of each unit of enzyme produced. For R&D teams, understanding these mechanistic advantages provides confidence in scaling the process, as the risk of batch failure due to enzyme instability is significantly mitigated compared to using wild-type or less optimized variants.
How to Synthesize (R)-3-amino-1-morpholine-4-(2,4,5-trifluorophenyl)-1-butanone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a pilot or production setting, emphasizing simplicity and reproducibility. The process begins with the fermentation of recombinant E. coli BL21 strains harboring the specific transaminase genes, followed by a straightforward cell disruption and homogenization step to yield a crude enzyme preparation. This eliminates the need for expensive and time-consuming protein purification steps, further driving down the cost of goods. The reaction is conducted in a biphasic or co-solvent system utilizing isopropanol and water, with precise control over pH and temperature to maximize yield. Detailed standardized synthesis steps are provided in the guide below to ensure consistent results.
- Cultivate engineered E. coli BL21 strains containing the specific transaminase gene variants (e.g., Enz.27 with S150W-C152T-Q155K mutations) in LB medium with kanamycin selection until OD600 reaches 0.8.
- Induce protein expression with IPTG at 18°C overnight, harvest cells via centrifugation, and homogenize with phosphate buffer to prepare the crude enzyme solution.
- Conduct the transamination reaction at 45°C using isopropanol as solvent and isopropylamine hydrochloride as the amino donor, achieving high conversion rates and >99% ee value.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this engineered transaminase technology offers compelling economic and operational benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic reduction of raw material waste; by achieving significantly higher conversion rates, the amount of expensive fluorinated starting material lost to unreacted substrate is minimized. This efficiency gain directly lowers the variable cost per kilogram of the intermediate, improving the overall margin profile of the final API. Additionally, the high stereoselectivity eliminates the need for costly chiral resolution steps or recrystallization processes that are often required to meet purity specifications, simplifying the downstream processing train and reducing solvent usage. These factors combine to create a more lean and agile manufacturing process that is less susceptible to fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The implementation of high-efficiency enzyme mutants leads to substantial cost savings by reducing the enzyme loading required per batch and minimizing substrate waste. Since the conversion rates are significantly improved over prior art, less starting material is needed to produce the same amount of product, effectively lowering the material cost basis. Furthermore, the ability to use crude enzyme lysates instead of purified proteins removes a major expense from the biocatalyst production budget, making the overall process economically attractive for large-scale commercialization without compromising on quality or performance metrics.
- Enhanced Supply Chain Reliability: Utilizing a robust biological catalyst expressed in a common host like E. coli ensures a stable and scalable supply of the biocatalyst itself. Unlike rare metal catalysts which are subject to geopolitical supply risks and price volatility, fermented enzymes can be produced reliably in multiple facilities globally. The improved stability of the mutants also means the enzyme can be stored for longer periods or transported with less stringent cold-chain requirements, reducing logistics costs and ensuring continuity of supply even during disruptions. This reliability is crucial for maintaining uninterrupted API production schedules and meeting delivery commitments to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently green, operating at mild temperatures around 45°C and avoiding the use of toxic heavy metals or hazardous reagents typical of chemical asymmetric synthesis. This aligns with increasingly strict environmental regulations and corporate sustainability goals, reducing the burden of waste treatment and disposal. The scalability is proven by the use of standard fermentation and reaction equipment, allowing for seamless transition from laboratory development to multi-ton annual production capacities. This ease of scale-up reduces the time-to-market for generic versions of the drug and ensures that manufacturing capacity can be rapidly expanded to meet surges in demand without significant capital investment in specialized infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this transaminase technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational parameters and benefits for stakeholders evaluating this route for their own manufacturing portfolios. Understanding these details is essential for making informed decisions about technology transfer and process integration.
Q: What specific amino acid mutations improve transaminase activity for Sitagliptin synthesis?
A: The patent identifies critical mutations at positions 150, 152, and 155 relative to SEQ ID NO: 1. Specific combinations such as S150W, C152T, and Q155K (Enz.27) have demonstrated conversion rates exceeding 58%, significantly outperforming the wild-type baseline.
Q: How does this enzymatic route compare to traditional chemical synthesis?
A: Unlike traditional chemical routes that often require harsh conditions and complex chiral resolution steps, this biocatalytic approach utilizes engineered transaminases to achieve >99% enantiomeric excess (ee) directly, simplifying downstream purification and reducing environmental impact.
Q: Is this process scalable for industrial API production?
A: Yes, the method utilizes standard E. coli BL21 fermentation and simple crude enzyme preparation protocols. The high stability and conversion efficiency of the mutants facilitate scale-up from laboratory benchtop to multi-ton commercial manufacturing without significant loss of catalytic performance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this engineered transaminase technology for the efficient production of Sitagliptin intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale innovation to full-scale manufacturing is seamless and compliant. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical enantiomeric excess values required for this chiral intermediate. We are committed to delivering high-quality pharmaceutical intermediates that meet the exacting standards of the global healthcare industry.
We invite you to collaborate with us to leverage this advanced biocatalytic route for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this technology can optimize your COGS. Please contact us to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the production of next-generation diabetes therapeutics through our reliable supply of high-purity Sitagliptin intermediates.
