Optimizing Escitalopram Production: A Novel Separation Strategy for High-Purity Intermediates
The pharmaceutical industry continuously seeks robust methodologies for the production of optically active compounds, particularly for blockbuster antidepressants like escitalopram. Patent CN1918112B presents a significant technological advancement in the isolation of intermediates critical for escitalopram synthesis. This invention addresses the persistent challenge of separating enantiomers from racemic mixtures or partially resolved streams with high efficiency. By combining enzymatic resolution with a novel chemical separation technique involving cyclic anhydrides, the process achieves superior optical purity without relying on costly chromatographic columns. For R&D directors and procurement specialists, understanding this pathway is essential for evaluating next-generation supply chains. The core innovation lies in the transformation of the target S-enantiomer into a derivative that possesses distinct physical properties, specifically solubility, compared to its R-counterpart. This allows for a straightforward precipitation and filtration step, drastically simplifying the downstream processing requirements. As a leading entity in fine chemical manufacturing, we recognize the potential of this patented approach to redefine cost structures and purity standards in the antidepressant market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of single-enantiomer pharmaceuticals has been plagued by inefficient resolution techniques that burden both R&D budgets and production timelines. Traditional methods often involve the formation of diastereomeric salts using expensive chiral resolving agents, such as di-p-toluoyl tartaric acid, which require multiple crystallization cycles to achieve acceptable enantiomeric excess. Alternatively, preparative HPLC on chiral stationary phases offers high purity but suffers from severe limitations in throughput and solvent consumption, making it economically unviable for large-scale commercial manufacturing. Furthermore, earlier synthetic routes described in prior art, such as US Patent 4,943,590, often necessitate complex multi-step sequences to install the chiral center, leading to cumulative yield losses. The reliance on stoichiometric amounts of chiral auxiliaries generates significant chemical waste, complicating environmental compliance and increasing disposal costs. For supply chain managers, these inefficiencies translate into longer lead times and higher vulnerability to raw material price fluctuations. The inability to easily separate the desired S-enantiomer from the R-enantiomer without sophisticated equipment has long been a bottleneck in the generic API sector, driving up the cost of goods sold and limiting market accessibility for essential medications.
The Novel Approach
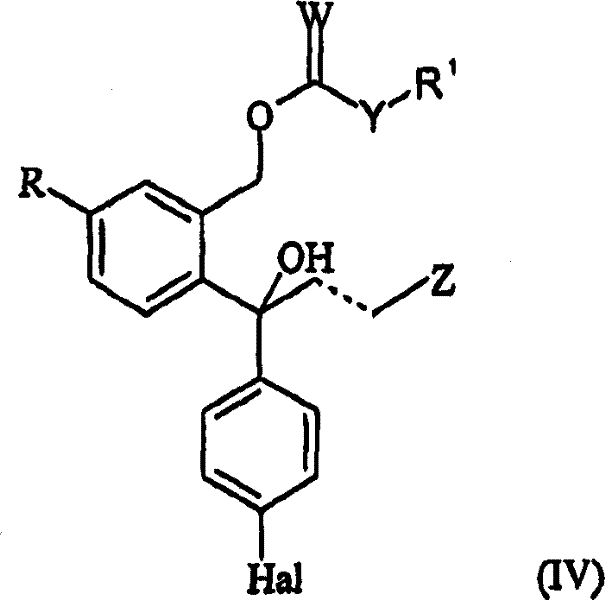
The methodology disclosed in CN1918112B circumvents these traditional bottlenecks by introducing a clever chemical differentiation strategy following an initial enzymatic enrichment. Instead of attempting to separate the diols directly, the process converts the mixture into acylated derivatives using specific cyclic anhydrides. This reaction targets the hydroxyl groups, attaching a moiety that contains a free carboxylic acid group. Crucially, the resulting derivative of the S-enantiomer exhibits significantly lower solubility in the reaction medium compared to the R-enantiomer or the unreacted starting materials. This physical property difference enables the S-enantiomer to precipitate out of the solution spontaneously upon formation. The solid can then be isolated via simple filtration, a unit operation that is infinitely more scalable and cost-effective than chromatography. This approach effectively turns a difficult separation problem into a straightforward crystallization process. By integrating this step after an enzymatic kinetic resolution, the overall optical purity is maximized while minimizing the number of purification stages. For manufacturers, this represents a paradigm shift towards process intensification, where reaction and separation are coupled to enhance efficiency. The result is a streamlined workflow that reduces solvent usage, eliminates the need for exotic chiral columns, and delivers high-purity intermediates ready for the final cyclization step.
Mechanistic Insights into Anhydride-Mediated Enantiomer Separation
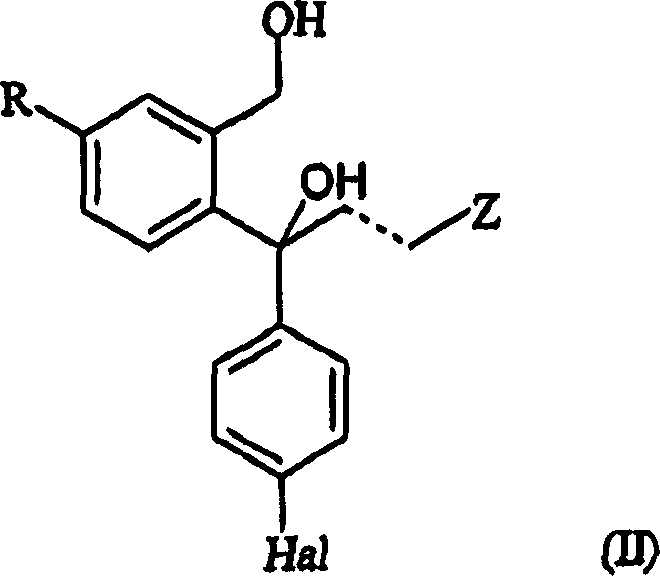
The chemical mechanism underpinning this separation technology relies on the differential reactivity and solubility profiles of the enantiomers when exposed to cyclic anhydrides. The process begins with a mixture containing the S-enantiomer of the diol (Formula II) and its acylated counterpart (Formula IV). When this mixture is treated with a cyclic anhydride, such as succinic anhydride or glutaric anhydride, a ring-opening esterification occurs. The anhydride reacts with the primary hydroxyl group of the diol to form a mono-ester possessing a terminal carboxylic acid functionality. The patent specifies that the S-enantiomer derivative (Formula V) formed in this reaction has a strong tendency to crystallize or precipitate from the organic solvent, such as tetrahydrofuran. This phenomenon is likely due to the specific spatial arrangement of the S-configuration allowing for more efficient packing in the crystal lattice or stronger intermolecular hydrogen bonding via the carboxylic acid dimerization. In contrast, the R-enantiomer derivative remains dissolved in the mother liquor. This selective precipitation acts as a powerful driving force, pulling the equilibrium towards the formation of the S-product and effectively removing it from the reaction environment. The use of anhydrides like succinic anhydride is particularly advantageous because the resulting succinate half-ester is stable yet reactive enough for the subsequent cyclization step. This mechanistic elegance ensures that the separation is not just a physical partitioning but is chemically driven, offering high selectivity and robustness against minor variations in reaction conditions.
Impurity control is another critical aspect where this mechanism excels, particularly regarding the removal of the unwanted R-enantiomer and residual enzymes from the previous biocatalytic step. Since the separation relies on the formation of a solid precipitate, soluble impurities, including the R-enantiomer acylated derivatives and any unreacted racemic material, remain in the filtrate. This provides a high degree of purification in a single operation. Furthermore, the subsequent cyclization step, which involves treating the isolated S-enantiomer ester with a base like sodium hydride, is highly specific. The base induces an intramolecular nucleophilic attack by the phenolic oxygen (after deprotonation or activation) onto the side chain, closing the phthalane ring to form the escitalopram core. Because the starting material for this cyclization is already enriched to high optical purity via the precipitation step, the risk of racemization during the harsh basic conditions is minimized. The patent notes that optical purity can be further enhanced if necessary through recrystallization of the final salt, but the primary separation efficiency renders this often unnecessary. This dual-stage purification—first by selective precipitation of the intermediate, then by crystallization of the final salt—ensures that the final API meets stringent regulatory specifications for chiral purity, typically exceeding 98% ee, which is vital for patient safety and regulatory approval.
How to Synthesize Escitalopram Intermediates Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to maximize yield and optical purity. The process initiates with the preparation of the substrate mixture, often achieved through selective enzymatic acylation of the racemic diol using lipases such as Candida antarctica lipase B. Once the enzymatic step reaches the desired conversion, the reaction mixture is subjected to the chemical separation protocol. The detailed standardized synthesis steps are outlined below to guide process development teams in replicating this efficient route.
- Prepare a mixture of racemic diol and its acylated derivative via selective enzymatic acylation using lipases such as Candida antarctica lipase B.
- React the mixture with a cyclic anhydride (e.g., succinic anhydride) in an inert organic solvent like tetrahydrofuran to form a carboxylic acid derivative.
- Induce precipitation of the S-enantiomer acid derivative, filter the solid, and subsequently cyclize using a base like NaH to form the final escitalopram structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this separation technology offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the purification train. By replacing chromatographic separation with precipitation and filtration, the process eliminates the need for expensive chiral stationary phases and the associated high-volume solvent consumption. This reduction in material intensity directly translates to significant cost savings in raw materials and waste disposal. Moreover, filtration is a continuous or semi-continuous operation that scales linearly with production volume, unlike batch chromatography which faces diminishing returns at larger scales. This scalability ensures a more reliable supply of intermediates, reducing the risk of production bottlenecks that can delay API delivery. The use of common reagents like succinic anhydride further enhances supply chain security, as these chemicals are commodity items with stable pricing and widespread availability, unlike specialized chiral resolving agents which may have limited suppliers. Additionally, the robustness of the precipitation method means that the process is less sensitive to minor fluctuations in feedstock quality, providing greater operational stability. Overall, this technology supports a leaner, more agile manufacturing model that aligns with modern principles of green chemistry and cost-effective production.
- Cost Reduction in Manufacturing: The elimination of preparative HPLC and expensive chiral resolving agents significantly lowers the variable cost per kilogram of the intermediate. The precipitation method utilizes inexpensive cyclic anhydrides and standard solvents, reducing the overall bill of materials. Furthermore, the ability to recover the unwanted R-enantiomer from the mother liquor for potential racemization and recycling adds value by improving the overall atom economy of the process. This comprehensive approach to cost management ensures that the final API can be produced at a competitive price point, enhancing market competitiveness for generic manufacturers.
- Enhanced Supply Chain Reliability: Reliance on commodity chemicals like succinic anhydride and standard filtration equipment mitigates the risk of supply disruptions associated with specialized reagents. The process is robust and tolerant to variations, ensuring consistent output quality even under fluctuating production conditions. This reliability is crucial for maintaining uninterrupted supply to downstream API manufacturers, preventing costly downtime. The simplified workflow also reduces the dependency on highly specialized technical personnel for operation, making it easier to transfer technology between manufacturing sites or contract manufacturing organizations, thereby diversifying the supply base and enhancing resilience.
- Scalability and Environmental Compliance: The transition from chromatography to crystallization/filtration dramatically reduces solvent waste, aligning with increasingly strict environmental regulations. Solvent recovery is more straightforward in a precipitation process, allowing for higher recycling rates and a smaller environmental footprint. The process avoids the use of heavy metal catalysts often found in alternative synthetic routes, simplifying the purification of the final product to meet residual metal specifications. This environmental stewardship not only reduces compliance costs but also enhances the corporate sustainability profile, which is becoming a key factor in supplier selection for major pharmaceutical companies committed to green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this separation technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and licensees.
Q: How does this patent improve upon traditional resolution methods?
A: Traditional methods often rely on expensive chiral acids or complex HPLC separations. This patent introduces a chemical differentiation strategy where the desired S-enantiomer is converted into a carboxylic acid derivative that spontaneously precipitates, allowing for simple filtration rather than chromatography.
Q: What specific reagents are used for the separation step?
A: The process utilizes cyclic anhydrides such as succinic anhydride, glutaric anhydride, or phthalic anhydride. These reagents react selectively with the hydroxyl groups to form esters containing a free carboxylic acid moiety, which alters the solubility profile of the target enantiomer.
Q: Is this process scalable for commercial API production?
A: Yes, the reliance on precipitation and filtration makes this highly scalable. Unlike chromatographic methods which are difficult to scale economically, solid-liquid separation is a standard unit operation in large-scale pharmaceutical manufacturing, ensuring consistent supply and lower operational costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Escitalopram Intermediate Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the one described in CN1918112B are translated into efficient industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral purity analysis via SFC and HPLC. We understand the critical nature of antidepressant intermediates in the global supply chain and are committed to delivering consistent quality. Our team of expert chemists can optimize the enzymatic and chemical steps to maximize yield and minimize cycle time, providing a seamless bridge from laboratory innovation to commercial reality. We invite you to leverage our technical expertise to secure a stable and cost-effective supply of high-quality escitalopram intermediates.
We encourage potential partners to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes. By collaborating with us, you can access specific COA data and route feasibility assessments that demonstrate the viability of this advanced separation method for your portfolio. Let us help you navigate the complexities of chiral synthesis and achieve your commercial goals with confidence and precision.
