Advanced One-Pot Synthesis of Mercapto-Substituted Nitrogen Heterocycles for Commercial Scale Production
Advanced One-Pot Synthesis of Mercapto-Substituted Nitrogen Heterocycles for Commercial Scale Production
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes that balance high efficiency with stringent safety and environmental standards. Patent CN108017594B introduces a transformative preparation method for mercapto-substituted nitrogen heterocyclic compounds, addressing critical pain points associated with traditional synthesis protocols. This technology leverages a novel one-pot cyclization strategy where substituted aniline compounds react directly with disulfide compounds in an organic solvent medium. Unlike conventional methods that rely on hazardous carbon disulfide, this approach utilizes stable disulfide reagents to construct the heterocyclic core under moderate thermal conditions ranging from 60°C to 150°C. The significance of this innovation lies in its ability to streamline the manufacturing process while simultaneously mitigating the severe toxicity risks inherent to sulfur-based chemistry. By eliminating the need for transition metal catalysts and toxic gaseous reagents, this method offers a compelling value proposition for manufacturers aiming to optimize their supply chain resilience and reduce regulatory compliance burdens.
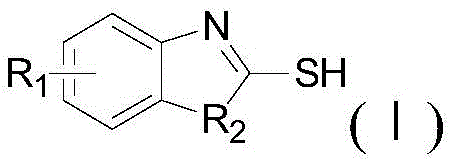
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of vital intermediates such as 2-mercaptobenzothiazole and its analogues has been heavily dependent on the use of carbon disulfide (CS2) as the primary sulfur source. This legacy approach presents formidable challenges for modern chemical manufacturing, primarily due to the extreme volatility and neurotoxicity of carbon disulfide, which necessitates elaborate containment systems and rigorous worker safety protocols. Furthermore, reactions involving CS2 often require harsh conditions and multiple processing steps, leading to lower overall atom economy and increased generation of hazardous waste streams. The reliance on heavy metal catalysts in some variations of these traditional routes further complicates the downstream purification process, as residual metals must be reduced to parts-per-million levels to meet pharmaceutical specifications. These factors collectively contribute to elevated production costs, extended lead times, and a larger environmental footprint, making the conventional CS2-based route increasingly untenable for sustainable large-scale production.
The Novel Approach
In stark contrast to the hazardous legacy methods, the disclosed invention employs a direct condensation reaction between substituted anilines and disulfide compounds, effectively bypassing the need for gaseous sulfur reagents. As illustrated by the reactant structures below, the process utilizes readily available precursors that are significantly safer to handle and store. 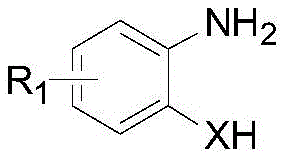
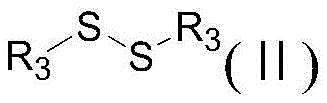 This innovative pathway operates through a thermal cyclization mechanism that proceeds efficiently in common organic solvents such as ethanol, toluene, or dimethylformamide. The reaction conditions are remarkably mild, typically requiring temperatures between 60°C and 150°C for durations of 1 to 15 hours, which reduces energy consumption compared to high-temperature pyrolysis methods. The workup procedure is equally streamlined, involving simple dilution with ethanol and water followed by acidification to precipitate the product, thereby avoiding complex extraction or chromatography steps. This simplicity not only accelerates the production cycle but also enhances the overall yield and purity profile of the final heterocyclic compound.
This innovative pathway operates through a thermal cyclization mechanism that proceeds efficiently in common organic solvents such as ethanol, toluene, or dimethylformamide. The reaction conditions are remarkably mild, typically requiring temperatures between 60°C and 150°C for durations of 1 to 15 hours, which reduces energy consumption compared to high-temperature pyrolysis methods. The workup procedure is equally streamlined, involving simple dilution with ethanol and water followed by acidification to precipitate the product, thereby avoiding complex extraction or chromatography steps. This simplicity not only accelerates the production cycle but also enhances the overall yield and purity profile of the final heterocyclic compound.
Mechanistic Insights into Disulfide-Mediated Cyclization
The core of this technological advancement lies in the unique reactivity of the disulfide bond when subjected to nucleophilic attack by the ortho-substituted aniline derivative. Under thermal activation in a polar or semi-polar solvent, the disulfide linkage undergoes cleavage, generating reactive sulfur species that facilitate the formation of the C-S or C-N bonds necessary for ring closure. For instance, when synthesizing 2-mercaptobenzothiazole, the thiol group of 2-aminothiophenol interacts with the tetramethylthiuram disulfide to drive the cyclization forward without external catalytic promotion. 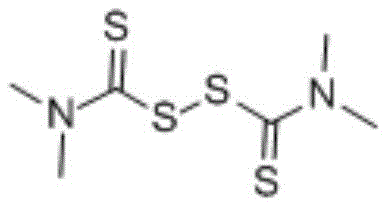 This catalyst-free mechanism is particularly advantageous for pharmaceutical applications, as it inherently prevents the introduction of transition metal impurities that are difficult to remove. The reaction kinetics are favorable, allowing for high conversion rates within a relatively short timeframe, which is critical for maintaining high throughput in a commercial setting. Additionally, the versatility of the disulfide reagent allows for the synthesis of various analogues, including benzoxazoles and benzimidazoles, by simply varying the starting aniline substrate, as demonstrated in the specific reaction schemes for 2-mercaptobenzoxazole and 2-mercaptobenzimidazole shown below.
This catalyst-free mechanism is particularly advantageous for pharmaceutical applications, as it inherently prevents the introduction of transition metal impurities that are difficult to remove. The reaction kinetics are favorable, allowing for high conversion rates within a relatively short timeframe, which is critical for maintaining high throughput in a commercial setting. Additionally, the versatility of the disulfide reagent allows for the synthesis of various analogues, including benzoxazoles and benzimidazoles, by simply varying the starting aniline substrate, as demonstrated in the specific reaction schemes for 2-mercaptobenzoxazole and 2-mercaptobenzimidazole shown below. 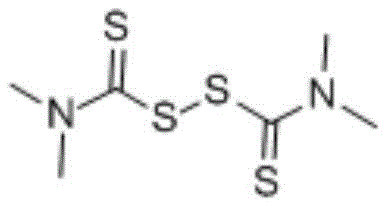
 The structural integrity of the final product is maintained through precise control of the acidification step, ensuring that the mercapto tautomer is stabilized effectively.
The structural integrity of the final product is maintained through precise control of the acidification step, ensuring that the mercapto tautomer is stabilized effectively.
Impurity control is another critical aspect where this mechanism excels, primarily due to the clean nature of the byproduct profile. Since the reaction does not involve oxidative coupling agents or heavy metal catalysts, the resulting crude product contains fewer side products related to metal coordination or over-oxidation. The primary byproducts are generally organic amines or sulfides that can be easily separated during the aqueous workup or recrystallization stages. This high level of chemical selectivity ensures that the final active pharmaceutical ingredient (API) intermediate meets stringent purity specifications, often exceeding 96% purity directly after filtration and drying. For R&D teams focused on impurity profiling, this route offers a significant advantage by simplifying the identification and quantification of potential genotoxic impurities, thereby accelerating the regulatory filing process for new drug candidates.
How to Synthesize Mercapto-Substituted Nitrogen Heterocycles Efficiently
The operational simplicity of this synthesis route makes it highly attractive for process chemists looking to transfer technology from the laboratory to pilot plant scales. The general procedure involves charging a reactor with the substituted aniline and the disulfide compound in a selected organic solvent, followed by heating the mixture to the specified temperature range. This straightforward protocol minimizes the requirement for specialized equipment, allowing existing multipurpose reactors to be utilized effectively for the production of these valuable heterocyclic intermediates.
- Mix substituted aniline compound and disulfide compound in an organic solvent such as ethanol or toluene.
- Stir the reaction mixture at temperatures between 60°C and 150°C for a duration of 1 to 15 hours.
- Cool to room temperature, dilute with ethanol and water, acidify to pH 1-6, then filter and dry the solid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this disulfide-based synthesis route offers substantial opportunities for cost optimization and supply chain stabilization. The elimination of carbon disulfide removes a major bottleneck associated with the storage and handling of highly regulated hazardous materials, thereby reducing insurance premiums and facility compliance costs. Furthermore, the absence of expensive transition metal catalysts translates directly into raw material savings, as there is no need to procure costly palladium or copper salts nor invest in dedicated metal scavenging resins for purification. The simplified workup procedure, which relies on basic acidification and filtration rather than complex distillation or chromatography, significantly reduces utility consumption and solvent waste disposal fees. These cumulative efficiencies result in a markedly lower cost of goods sold (COGS), providing a competitive edge in pricing negotiations with downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the drastic simplification of the reaction setup and the removal of high-cost reagents. By utilizing stable disulfide compounds instead of gaseous carbon disulfide, manufacturers avoid the capital expenditure associated with specialized gas handling infrastructure and leak detection systems. Additionally, the catalyst-free nature of the reaction eliminates the financial burden of metal removal technologies, which often account for a significant portion of purification costs in fine chemical synthesis. The high atom economy and direct precipitation of the product further minimize solvent usage and waste treatment expenses, leading to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Supply continuity is significantly improved by relying on commercially abundant and stable raw materials like tetramethylthiuram disulfide and substituted anilines. Unlike carbon disulfide, which is subject to strict transportation regulations and supply volatility due to safety concerns, disulfide reagents are solids or high-boiling liquids that are easy to stockpile and transport globally. This stability ensures that production schedules are not disrupted by raw material shortages or logistical delays, allowing suppliers to maintain consistent delivery timelines for their clients. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further enhancing the reliability of the supply chain.
- Scalability and Environmental Compliance: The environmental profile of this method aligns perfectly with modern green chemistry initiatives, facilitating easier permitting and regulatory approval for capacity expansions. The reduction in toxic emissions and hazardous waste generation simplifies the environmental impact assessment process, enabling faster deployment of new production lines. Moreover, the use of common solvents like ethanol allows for efficient solvent recovery and recycling, contributing to a circular economy model within the manufacturing facility. This commitment to sustainability not only mitigates regulatory risk but also enhances the brand reputation of the supplier among environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new method improve safety compared to traditional carbon disulfide routes?
A: The traditional method utilizes highly toxic and volatile carbon disulfide (CS2), posing severe health risks and environmental hazards. This novel protocol replaces CS2 with stable disulfide compounds like tetramethylthiuram disulfide, drastically reducing operator exposure to hazardous vapors and eliminating the need for complex gas scrubbing systems.
Q: What are the typical purity levels achievable with this catalyst-free process?
A: Experimental data from the patent indicates that the process yields products with purity greater than or equal to 96% without the need for transition metal catalysts. The absence of metal catalysts simplifies the purification workflow, avoiding expensive heavy metal removal steps often required in pharmaceutical grade synthesis.
Q: Is this synthesis route scalable for industrial manufacturing of API intermediates?
A: Yes, the process is designed for industrial applicability using common organic solvents like ethanol and toluene. The simple workup procedure involving acidification and filtration allows for straightforward scale-up from laboratory benchtop to multi-ton production facilities without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mercaptobenzothiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging the innovative disulfide-mediated cyclization technology, we can offer our partners a superior product profile with reduced impurity levels and enhanced batch-to-batch consistency.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific project requirements. Our experts are ready to discuss how this greener, more efficient synthesis route can optimize your supply chain and reduce overall manufacturing costs. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments for your next development program.
