Industrial Scale-Up of High-Purity L-Carnosine via Novel Phthaloyl-Silyl Protection Strategy
Introduction to Advanced L-Carnosine Manufacturing Technology
The global demand for bioactive peptides and nutraceutical ingredients has surged, driven by the aging population's need for effective anti-aging and antioxidant supplements. L-carnosine, a naturally occurring dipeptide composed of beta-alanine and L-histidine, stands out as a critical molecule in this sector due to its proven ability to scavenge free radicals, inhibit glycation, and support muscle function. However, the commercial viability of L-carnosine has historically been constrained by synthetic challenges, specifically low yields and difficult purification processes that fail to meet stringent pharmaceutical standards. Patent CN101284862B introduces a transformative synthetic methodology that addresses these bottlenecks through a sophisticated dual-protection strategy involving phthaloyl and silyl groups. This technical breakthrough not only achieves a total yield exceeding 90% but also ensures a final product purity of greater than 99.5% via HPLC analysis, setting a new benchmark for industrial production. By optimizing reaction conditions and solvent systems, this route eliminates the formation of stubborn by-products that typically necessitate expensive chromatographic separations. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a reliable supply chain of high-quality L-carnosine that can withstand the rigorous demands of the global nutraceutical and pharmaceutical markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for L-carnosine have long suffered from inherent inefficiencies that compromise both economic feasibility and product quality. Historically, methods involving the direct acylation of L-histidine with beta-alanine derivatives under free base conditions have resulted in significant formation of by-products, drastically reducing the isolated yield to below 80%. Alternative approaches utilizing esterification of L-histidine followed by acylation and subsequent hydrolysis introduce multiple processing steps, each carrying the risk of racemization and structural degradation of the sensitive imidazole ring. Furthermore, conventional deprotection strategies often rely on strong acidic or basic conditions that can lead to the formation of difficult-to-remove impurities, requiring extensive and costly purification protocols. The reliance on alcohol-based solvents for deprotection in older methods frequently results in the formation of azeotropic mixtures that are energetically expensive to separate and recover, thereby inflating the overall cost of goods sold. These technical deficiencies render many legacy processes unsuitable for modern Good Manufacturing Practice (GMP) environments where consistency and purity are paramount.
The Novel Approach
The methodology disclosed in patent CN101284862B represents a paradigm shift by employing a strategic combination of phthaloyl protection for beta-alanine and silyl protection for L-histidine. This approach begins with the reaction of beta-alanine and phthalic anhydride in a non-polar solvent, catalyzed by an organic amine, to form phthaloyl-beta-alanine with exceptional efficiency. The subsequent conversion to the acid chloride is meticulously controlled to prevent decomposition, utilizing chlorinating agents like triphosgene which offer safer handling profiles compared to traditional reagents. Crucially, the protection of L-histidine via trimethylsilylation creates a highly reactive yet stable intermediate that facilitates smooth condensation without the side reactions typical of unprotected amino acids. The deprotection sequence is elegantly designed to use water for removing the silyl group, avoiding the generation of hazardous organic waste streams associated with alcoholysis. This streamlined workflow reduces the number of unit operations, minimizes solvent consumption, and significantly enhances the overall throughput, making it an ideal candidate for cost reduction in peptide manufacturing.
Mechanistic Insights into Phthaloyl-Silyl Dual Protection Strategy
The core innovation of this synthesis lies in the precise orchestration of protecting group chemistry to maximize regioselectivity and minimize epimerization. The initial step involves the nucleophilic attack of the beta-alanine amino group on phthalic anhydride, forming a stable imide ring that effectively masks the amine functionality while activating the carboxylic acid for subsequent transformation. This phthaloyl group is robust enough to withstand the harsh conditions of acid chloride formation but can be cleanly removed later via hydrazinolysis, a reaction that proceeds rapidly under mild conditions to release the free amine without affecting the peptide bond. The choice of non-polar solvents such as toluene or ethylene dichloride is critical here, as they facilitate the removal of water generated during imide formation via azeotropic distillation, driving the equilibrium towards the desired product. This mechanistic control ensures that the intermediate phthaloyl-beta-alanine is obtained with high purity, serving as a solid foundation for the downstream coupling reaction.
Parallel to this, the activation of L-histidine requires a protecting group that can shield both the alpha-amino group and the nucleophilic nitrogen of the imidazole ring without introducing excessive steric hindrance. The reaction of L-histidine with hexamethyldisilazane generates a trimethylsilyl derivative that fulfills these criteria perfectly. 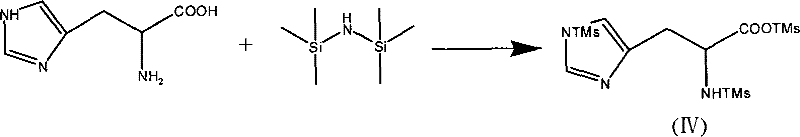 As illustrated in the reaction scheme, the silyl groups attach to the heteroatoms, rendering the histidine residue soluble in organic media and highly reactive towards electrophiles. When this silylated histidine encounters the phthaloyl-beta-alanyl chloride, the condensation proceeds efficiently to form the protected dipeptide. The subsequent hydrolysis step utilizes water to cleave the silicon-oxygen and silicon-nitrogen bonds, a process that is chemically orthogonal to the phthaloyl group, allowing for sequential deprotection. Finally, the treatment with hydrazine hydrate selectively cleaves the phthalimide ring, liberating the final L-carnosine molecule. This mechanistic pathway avoids the use of heavy metal catalysts or toxic reagents, aligning with green chemistry principles.
As illustrated in the reaction scheme, the silyl groups attach to the heteroatoms, rendering the histidine residue soluble in organic media and highly reactive towards electrophiles. When this silylated histidine encounters the phthaloyl-beta-alanyl chloride, the condensation proceeds efficiently to form the protected dipeptide. The subsequent hydrolysis step utilizes water to cleave the silicon-oxygen and silicon-nitrogen bonds, a process that is chemically orthogonal to the phthaloyl group, allowing for sequential deprotection. Finally, the treatment with hydrazine hydrate selectively cleaves the phthalimide ring, liberating the final L-carnosine molecule. This mechanistic pathway avoids the use of heavy metal catalysts or toxic reagents, aligning with green chemistry principles.
The formation of the acid chloride intermediate is another critical juncture where process control dictates success. 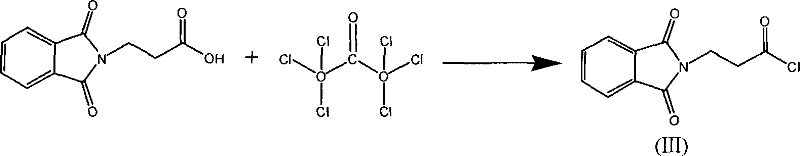 The conversion of phthaloyl-beta-alanine to its corresponding acid chloride using triphosgene, as shown above, must be managed carefully to prevent the formation of symmetric anhydrides or N-acyl urea by-products. The patent specifies maintaining temperatures around 70°C and utilizing solvents like ethylene dichloride to ensure complete dissolution and reaction homogeneity. The evolution of hydrogen chloride gas during this step is managed through absorption systems, which not only ensures safety but also allows for the potential recovery of hydrochloric acid as a valuable by-product. This level of mechanistic detail underscores the robustness of the process, providing R&D teams with a clear roadmap for troubleshooting and optimization during technology transfer.
The conversion of phthaloyl-beta-alanine to its corresponding acid chloride using triphosgene, as shown above, must be managed carefully to prevent the formation of symmetric anhydrides or N-acyl urea by-products. The patent specifies maintaining temperatures around 70°C and utilizing solvents like ethylene dichloride to ensure complete dissolution and reaction homogeneity. The evolution of hydrogen chloride gas during this step is managed through absorption systems, which not only ensures safety but also allows for the potential recovery of hydrochloric acid as a valuable by-product. This level of mechanistic detail underscores the robustness of the process, providing R&D teams with a clear roadmap for troubleshooting and optimization during technology transfer.
How to Synthesize L-Carnosine Efficiently
Implementing this synthesis route requires strict adherence to the stoichiometric ratios and temperature profiles outlined in the patent embodiments to achieve the reported high yields. The process begins with the preparation of phthaloyl-beta-alanine, followed by its activation to the acid chloride, and concurrently, the silylation of L-histidine. These two streams converge in a condensation reaction performed at low temperatures to minimize thermal degradation. The subsequent workup involves a series of aqueous extractions and pH adjustments to isolate the neutralized intermediate before the final hydrazinolysis step. Detailed standard operating procedures regarding solvent recovery, crystallization temperatures, and filtration rates are critical for maintaining batch-to-batch consistency. For a comprehensive breakdown of the specific molar equivalents, reaction times, and purification parameters required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Dissolve beta-alanine in a non-polar solvent like toluene and react with phthalic anhydride under organic amine catalysis to form phthaloyl-beta-alanine.
- Convert phthaloyl-beta-alanine to phthaloyl-beta-alanyl chloride using a chlorinating agent such as triphosgene or thionyl chloride.
- Protect L-histidine using hexamethyldisilazane to form L-histidine trimethylsilane, then condense with the acid chloride followed by hydrazinolysis to yield L-carnosine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for procurement managers and supply chain leaders seeking to optimize their sourcing strategies for peptide intermediates. The elimination of complex esterification and hydrolysis steps significantly reduces the number of processing stages, which directly translates to lower labor costs and reduced equipment occupancy time. By utilizing common, recoverable solvents like toluene and ethylene dichloride, the process minimizes raw material expenditure and simplifies waste management protocols. The high crude yield reported in the patent means that less starting material is required to produce a given amount of finished product, effectively lowering the cost of goods sold without compromising on quality. Furthermore, the ability to use water for key deprotection steps reduces the dependency on expensive anhydrous alcohols and mitigates the environmental risks associated with volatile organic compound emissions.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis eliminates the need for expensive chromatographic purification columns that are often required in lower-yielding routes. By achieving high purity through simple recrystallization and aqueous workups, manufacturers can avoid the capital expenditure associated with preparative HPLC systems and the ongoing costs of stationary phase replacement. The use of triphosgene as a chlorinating agent, while requiring careful handling, offers a solid-state alternative to gaseous phosgene, reducing infrastructure costs related to gas containment and scrubbing systems. Additionally, the recovery of solvents via distillation allows for a closed-loop system that drastically cuts down on fresh solvent purchases, contributing to substantial long-term operational savings.
- Enhanced Supply Chain Reliability: The raw materials for this process, including beta-alanine, phthalic anhydride, and L-histidine, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing without significant yield loss, ensures consistent production output even in large-scale reactors. This reliability is crucial for maintaining continuous supply to downstream customers in the nutraceutical and pharmaceutical sectors who demand just-in-time delivery. The simplified workflow also shortens the overall production cycle time, enabling manufacturers to respond more agilely to fluctuations in market demand and secure their position as a reliable L-carnosine supplier.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial tonnage is facilitated by the use of standard unit operations such as reflux, distillation, and filtration, which are well-understood by chemical engineering teams. The avoidance of heavy metal catalysts simplifies the regulatory approval process for the final API or ingredient, as there is no need for extensive testing and validation of residual metal levels. The aqueous waste streams generated are relatively benign compared to those from traditional peptide synthesis, easing the burden on wastewater treatment facilities and ensuring compliance with increasingly stringent environmental regulations. This environmental compatibility enhances the brand value of the end product, appealing to eco-conscious consumers and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this L-carnosine synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of adopting this method for their specific production needs and ensures alignment with quality standards.
Q: What is the primary advantage of using phthaloyl protection for beta-alanine in L-carnosine synthesis?
A: The phthaloyl protection group offers superior stability during the acyl chloride formation and subsequent condensation steps, preventing racemization and side reactions that commonly plague free amino acid coupling, thereby ensuring high optical purity.
Q: How does the silylation of L-histidine improve the overall yield compared to traditional esterification methods?
A: Silylation using hexamethyldisilazane provides a robust yet easily removable protecting group for the imidazole and amino functionalities, eliminating the need for harsh acidic hydrolysis steps required in esterification routes which often degrade the sensitive dipeptide structure.
Q: Can this synthesis method be scaled for industrial production without significant environmental impact?
A: Yes, the process utilizes recoverable non-polar solvents like toluene and ethylene dichloride, and the water-based recrystallization steps minimize organic waste, making it highly suitable for large-scale GMP manufacturing with reduced environmental footprint.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Carnosine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation health products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering L-carnosine that meets stringent purity specifications, utilizing rigorous QC labs to verify every batch against the highest international standards. Our facility is equipped to handle the specific solvent systems and reaction conditions required by this advanced phthaloyl-silyl protection route, guaranteeing a consistent supply of material that supports your R&D and commercialization timelines.
We invite you to collaborate with us to leverage this cutting-edge synthetic technology for your product pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain resilience and drive down your overall production costs.
