Optimized Industrial Synthesis of Dexrazoxane: Enhancing Purity and Scalability for Global Pharma Supply Chains
Introduction to Advanced Dexrazoxane Manufacturing Technologies
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic efficiency, particularly for critical cardioprotective agents like dexrazoxane. Patent CN102952088A introduces a transformative preparation method that addresses the longstanding inefficiencies of conventional synthesis routes. Historically, the production of dexrazoxane has been plagued by complex multi-step sequences, excessive generation of inorganic waste, and the necessity for cumbersome purification techniques such as column chromatography. This novel technical disclosure outlines a streamlined approach wherein the key intermediate, (S)-1,2-diaminopropane-tetraacetate, is obtained through a highly efficient single-step alkylation process. By optimizing reaction conditions to avoid extreme temperatures and toxic reagents, this methodology not only shortens the overall production cycle but also significantly enhances the environmental profile of the manufacturing process. For global supply chain stakeholders, this represents a pivotal shift towards more sustainable and cost-effective production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art technologies, such as those described in patent WO9308172A1, typically rely on the reaction of (S)-1,2-propylene diamine with monochloroacetic acid under strongly alkaline conditions using excessive sodium hydroxide. While chemically feasible, this approach results in a crude product heavily contaminated with inorganic salts. The presence of these salts drastically reduces the reaction yield and complicates downstream processing, often necessitating hot column chromatography to achieve acceptable purity levels. Furthermore, alternative routes disclosed in European patent EP 2045235A1 involve the hydrolysis of tetranitriles under strong acidic conditions or the use of highly toxic cyclization agents like phenol and polyphosphoric acid. These harsh conditions pose significant safety risks, increase the complexity of waste disposal, and frequently lead to product decomposition, thereby compromising both the yield and the optical purity of the final dexrazoxane active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology presented in CN102952088A employs a mild and controlled alkylation strategy. The process initiates with the reaction of (S)-1,2-diaminopropane (or its hydrochloride salt) with chloroacetate esters in a basic solvent environment, preferably utilizing potassium carbonate in acetone. This specific combination of base and solvent minimizes side reactions and facilitates the easy removal of inorganic byproducts through simple filtration. Subsequent hydrolysis is conducted under controlled pH conditions (pH 2-5), allowing for the direct precipitation of the tetraacetic acid intermediate without the need for chromatographic separation. The final cyclization step utilizes formamide at moderate temperatures (140-160°C) under reduced pressure, effectively closing the piperazine ring to form dexrazoxane. This route eliminates the need for hazardous reagents and simplifies the isolation of the final product to a straightforward crystallization process, marking a substantial improvement in operational safety and efficiency.
Mechanistic Insights into N-Alkylation and Cyclization
The core of this synthetic innovation lies in the precise control of the N-alkylation mechanism. In the initial step, the nucleophilic nitrogen atoms of the (S)-1,2-diaminopropane attack the electrophilic carbon of the chloroacetate ester. The choice of a weak organic base or mild inorganic base like potassium carbonate is critical; it serves to scavenge the hydrochloric acid generated during the reaction without promoting excessive hydrolysis of the ester groups or racemization of the chiral center. The solvent system, often a mixture of acetone and water or pure acetone, ensures that the starting amine is soluble while the resulting inorganic salt (potassium chloride) precipitates out, driving the equilibrium forward and simplifying the workup. This mechanistic elegance prevents the formation of polymeric byproducts that are common in less controlled alkylation reactions.
Following the formation of the tetraester, the hydrolysis mechanism is equally vital for impurity control. By adjusting the pH of the reaction mixture to the range of 2 to 5 after alkaline hydrolysis, the tetraacetic acid intermediate precipitates selectively. This pH-dependent solubility behavior acts as a built-in purification step, leaving soluble impurities in the mother liquor. The final cyclization involves the nucleophilic attack of the amine nitrogen on the carbonyl carbon of the carboxylic acid group, facilitated by formamide which acts as both a solvent and a dehydrating agent. This intramolecular condensation forms the stable diketopiperazine ring characteristic of dexrazoxane. The rigorous control of temperature and pressure during this stage prevents thermal degradation, ensuring the structural integrity and high optical purity of the final molecule.
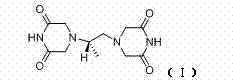
How to Synthesize Dexrazoxane Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and thermal management to maximize yield and purity. The process is divided into three distinct operational phases: the formation of the tetraester intermediate, the hydrolysis to the tetraacetic acid, and the final cyclization to dexrazoxane. Each phase utilizes standard chemical engineering unit operations, making the transition from laboratory scale to industrial production seamless. Operators must ensure precise pH control during the acidification step to optimize the recovery of the intermediate, and maintain strict temperature profiles during the formamide cyclization to prevent charring or decomposition. The detailed standardized synthetic steps, including specific molar ratios and reaction times validated by experimental data, are outlined in the guide below.
- Perform N-alkylation of (S)-1,2-diaminopropane with chloroacetate in a basic solvent environment (e.g., potassium carbonate in acetone) at 30-82°C to form the tetraester intermediate.
- Hydrolyze the tetraester intermediate using an aqueous alkali solution (e.g., NaOH or KOH), followed by acidification to pH 2-5 to precipitate the (S)-1,2-diaminopropane-tetraacetic acid.
- React the purified tetraacetic acid with formamide under reduced pressure at 140-160°C for 3-8 hours to effect cyclization, followed by hot filtration and crystallization to obtain high-purity dexrazoxane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling strategic advantages beyond mere technical superiority. The elimination of column chromatography, a batch process that is notoriously difficult to scale and expensive to operate, translates directly into significant reductions in manufacturing overhead. By replacing complex purification steps with simple filtration and crystallization, the process throughput is dramatically increased, allowing for larger batch sizes and more consistent supply continuity. Furthermore, the avoidance of toxic reagents such as phenol and strong mineral acids reduces the regulatory burden associated with hazardous material handling and waste disposal, leading to substantial cost savings in environmental compliance and worker safety protocols.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis significantly lowers the cost of goods sold (COGS) by minimizing raw material waste and energy consumption. Traditional methods often suffer from low yields due to side reactions and difficult purification, whereas this optimized route achieves high conversion rates with minimal loss of chiral material. The ability to recover and recycle solvents like acetone and methanol further enhances the economic viability of the process. Additionally, the reduction in processing time—shortening the production cycle by eliminating tedious post-treatment steps—allows for better asset utilization and faster turnaround times for custom manufacturing orders.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and commodity-grade starting materials. (S)-1,2-diaminopropane and chloroacetate esters are commercially accessible in bulk quantities, reducing the risk of raw material shortages that can plague specialized synthetic routes. The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing without compromising product quality, ensures consistent batch-to-batch reproducibility. This reliability is crucial for maintaining long-term supply agreements with pharmaceutical clients who require guaranteed availability of critical intermediates for their own drug formulation schedules.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a greener alternative to legacy methods. The reduction in inorganic salt waste and the absence of heavy metal catalysts or toxic organic solvents simplify the wastewater treatment process. This aligns with increasingly stringent global environmental regulations and corporate sustainability goals. The scalability of the process is evidenced by its reliance on standard reactor types and separation equipment, facilitating a smooth scale-up from pilot plant trials to multi-ton commercial production without the need for specialized or exotic infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of dexrazoxane using this advanced methodology. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on purity standards, process safety, and scalability potential. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their broader supply chain strategies.
Q: How does this new synthesis route improve product purity compared to conventional methods?
A: Unlike traditional methods that generate excessive inorganic salts requiring difficult column chromatography purification, this optimized route utilizes controlled hydrolysis and precipitation (pH 2-5) to isolate intermediates. This avoids thermal degradation and salt contamination, consistently achieving purity levels exceeding 99.75%.
Q: What are the primary safety advantages of avoiding strong acid hydrolysis in dexrazoxane production?
A: Conventional routes often employ strong acid hydrolysis of nitriles or toxic cyclization agents like phenol and polyphosphoric acid. The patented method replaces these with milder alkaline hydrolysis and formamide cyclization, significantly reducing operator exposure to hazardous chemicals and simplifying waste treatment protocols.
Q: Is this synthetic pathway suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for industrial scalability. It eliminates complex separation steps like column chromatography and relies on standard unit operations such as reflux, filtration, and crystallization. The use of common solvents like acetone and methanol further enhances its feasibility for multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dexrazoxane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of dexrazoxane meets the highest international standards, including the >99.75% purity benchmarks demonstrated in recent technical advancements.
We invite pharmaceutical partners to collaborate with us to leverage these cost-saving synthetic innovations for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your project benefits from the most efficient and reliable manufacturing solutions available in the market.
