Advanced Manufacturing of Dexrazoxane Intermediates via Novel Cyclization Technology
Introduction to Next-Generation Dexrazoxane Manufacturing
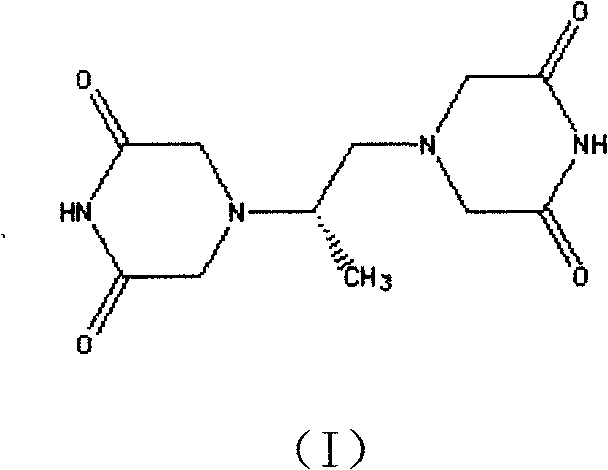
The pharmaceutical landscape for cardioprotective agents continues to evolve, driven by the urgent need for higher purity intermediates that ensure patient safety and regulatory compliance. Patent CN101684100A introduces a transformative preparation method for dexrazoxane, also known as ICRF-187, which serves as a critical chelating agent for preventing anthracycline-induced cardiotoxicity in cancer therapy. This technology represents a significant departure from legacy synthesis routes that have long plagued manufacturers with issues regarding impurity profiles, specifically the formation of persistent pink-colored degradation products. By leveraging a novel condensation pathway starting from chloroacetonitrile, this process achieves a streamlined workflow that directly addresses the pain points of R&D directors seeking robust, scalable chemistry. The method not only simplifies the operational complexity but also ensures the final active pharmaceutical ingredient meets stringent quality specifications without the need for excessive recrystallization cycles.
For global supply chain stakeholders, the implications of this patent are profound, offering a reliable pharmaceutical intermediate supplier pathway that mitigates the risks associated with toxic solvent residues and low yields. The core innovation lies in the generation of N,N-dichloroacetimide as a key reactive intermediate, which subsequently undergoes cyclization with chiral diamines under controlled thermal conditions. This approach effectively bypasses the thermal instability issues inherent in tetraacetic acid precursors used in conventional methods. As we delve deeper into the technical specifics, it becomes clear that this synthesis route is not merely an incremental improvement but a fundamental re-engineering of the manufacturing logic, designed to enhance both economic viability and chemical integrity for high-purity API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
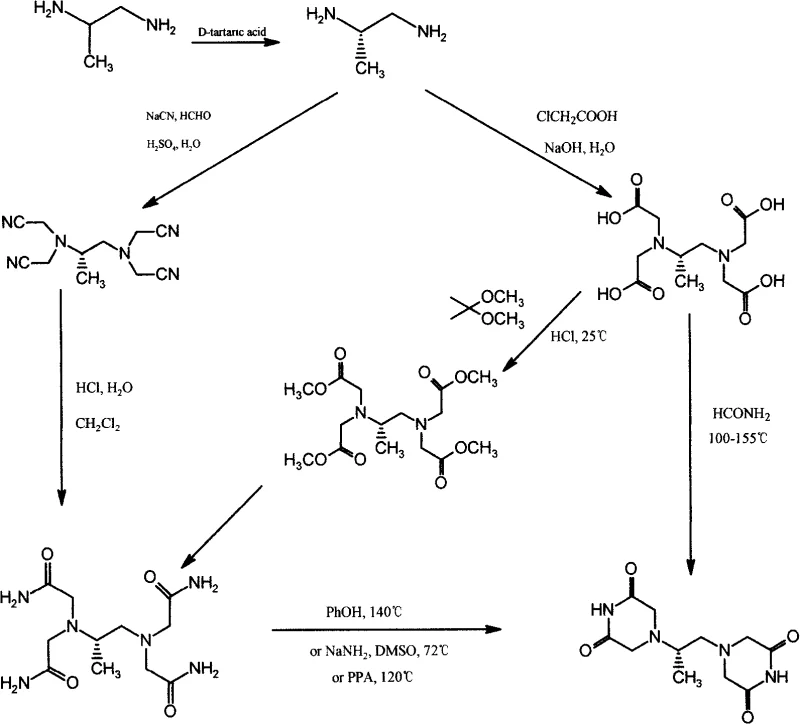
Historically, the synthesis of dexrazoxane has been hindered by convoluted multi-step processes that rely heavily on the resolution of 1,2-propanediamine and subsequent tetraacetylation. Traditional routes often involve reacting the diamine with chloroacetic acid under strongly basic conditions, leading to the formation of 1,2-propanediaminetetraacetic acid intermediates laden with inorganic salts. These salts are notoriously difficult to remove completely without resorting to column chromatography, a technique that is economically unfeasible for multi-ton commercial production. Furthermore, the subsequent cyclization step typically requires harsh conditions, such as heating with formamide or polyphosphoric acid at temperatures exceeding 100°C, which promotes decarboxylation and the generation of open-ring impurities.
The resulting crude product from these legacy methods frequently exhibits a undesirable pink coloration, indicative of complex degradation byproducts that are resistant to standard purification techniques. Patents such as EP330381 and WO93/08172 describe refining processes using toxic solvents like dioxane, which introduce severe regulatory hurdles regarding residual solvent limits (often exceeding 0.1% versus the pharmacopoeia limit of 0.038%). The reliance on these hazardous reagents not only complicates waste management but also poses significant occupational health risks. Consequently, the overall yield suffers, and the cost of goods sold remains prohibitively high due to the extensive downstream processing required to achieve acceptable purity levels.
The Novel Approach
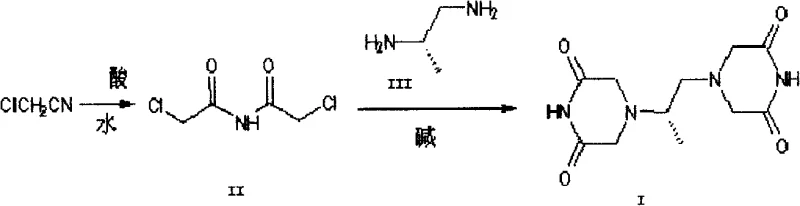
In stark contrast, the methodology disclosed in CN101684100A offers a radical simplification by utilizing chloroacetonitrile as the starting material. This route proceeds through the formation of N,N-dichloroacetimide (II) via an acid-catalyzed decomposition reaction at controlled temperatures between 100°C and 150°C. This intermediate is then reacted directly with D-(+)-1,2-propanediamine (III) in an alcoholic solvent system, facilitating a clean condensation reaction that constructs the piperazine-dione rings in a single pot. The elimination of the tetraacetic acid intermediate removes the risk of thermal decarboxylation entirely, thereby preserving the structural integrity of the molecule throughout the synthesis.
The operational simplicity of this new approach is evident in the workup procedure, which relies on straightforward crystallization and filtration rather than complex extractions or chromatographic separations. By conducting the reaction in C1-3 alkyl alcohols such as ethanol or methanol, the process utilizes green, industrially friendly solvents that are easy to recover and recycle. The product precipitates slowly from the reaction mixture as a white crystalline solid, inherently excluding the colored impurities that characterize older batches. This direct cyclization strategy not only shortens the production timeline significantly but also drastically reduces the consumption of raw materials and energy, positioning it as a superior choice for cost reduction in API manufacturing.
Mechanistic Insights into Acid-Catalyzed Cyclization
The chemical elegance of this process lies in the reactivity of the N,N-dichloroacetimide intermediate, which acts as a potent electrophile capable of undergoing double nucleophilic attack by the amine groups of the chiral diamine. Under alkaline conditions provided by bases like sodium ethoxide, the amine nitrogen attacks the imide carbon, initiating ring closure. The reaction temperature is carefully maintained between 30°C and 80°C, a range that is critical for balancing reaction kinetics with product stability. Unlike the high-temperature regimes of the past that fostered degradation, this moderate thermal window ensures that the cyclization proceeds to completion without triggering the breakdown of the newly formed heterocyclic rings. The slow precipitation of crystals during the reaction further drives the equilibrium towards the product, effectively sequestering the dexrazoxane from the solution before side reactions can occur.
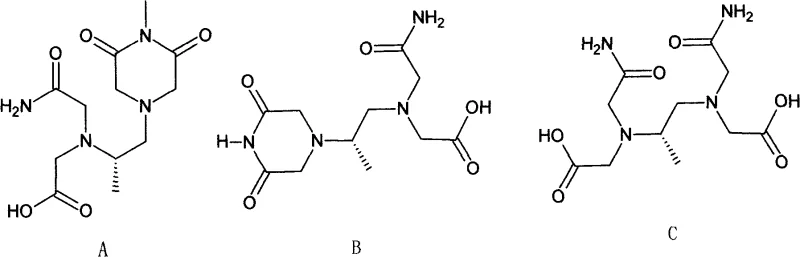
From an impurity control perspective, this mechanism effectively suppresses the formation of the degradation products A, B, and C, which are commonly observed in thermally stressed batches. The avoidance of strong acids like polyphosphoric acid in the final step prevents the hydrolysis of the amide bonds, a common failure mode in previous syntheses. Additionally, the use of alcoholic solvents facilitates the solubility of reactants while ensuring the product remains insoluble enough to crystallize out, acting as a self-purifying mechanism. This results in a final product with an HPLC purity consistently above 99.6%, demonstrating the robustness of the chemical design in maintaining high quality standards without the need for aggressive post-synthesis treatments.
How to Synthesize Dexrazoxane Efficiently
Implementing this synthesis protocol requires precise control over reaction parameters to maximize yield and optical purity. The process begins with the preparation of the key intermediate, N,N-dichloroacetimide, by heating chloroacetonitrile with an acid source such as hydrogen chloride or sulfuric acid. Following hydrolysis, the intermediate is dissolved in an alcohol solvent containing a base, creating the necessary environment for the subsequent condensation. The chiral diamine is then introduced slowly to manage the exotherm and ensure uniform crystal growth. Detailed standardized operating procedures regarding stoichiometry, addition rates, and cooling profiles are essential for reproducibility.
- Prepare N,N-dichloroacetimide (II) by reacting chloroacetonitrile with acid at 100-150°C, followed by hydrolysis.
- Dissolve the intermediate in C1-3 alkyl alcohol with a base such as sodium ethoxide.
- Slowly add D-(+)-1,2-propanediamine (III) at 30-80°C to induce cyclization and crystallization of Dexrazoxane (I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the substantial cost savings achieved through the elimination of expensive and hazardous reagents. By removing the need for toxic solvents like dioxane and avoiding the use of column chromatography for salt removal, the operational expenditure is significantly reduced. The simplified workflow means fewer unit operations, which translates to lower labor costs, reduced equipment downtime, and decreased utility consumption. Furthermore, the use of commodity chemicals like chloroacetonitrile and ethanol ensures a stable and resilient supply chain, minimizing the risk of raw material shortages that can disrupt production schedules.
Enhanced supply chain reliability is another critical benefit, as the process is inherently more scalable and easier to industrialize. The robust nature of the reaction conditions allows for seamless scale-up from pilot plant to commercial manufacturing without the unpredictability often associated with complex resolution steps. The high purity of the crude product reduces the burden on quality control laboratories and shortens the release time for batches, enabling faster time-to-market for finished dosage forms. Additionally, the environmental profile of the process is markedly improved, aligning with modern sustainability goals and reducing the costs associated with hazardous waste disposal and regulatory compliance. This makes the technology not only economically attractive but also environmentally responsible.
- Cost Reduction in Manufacturing: The elimination of column chromatography and toxic solvents drastically lowers processing costs. By utilizing a direct cyclization method, the number of processing steps is minimized, leading to significant reductions in solvent consumption and energy usage. The high yield and purity reduce the need for reprocessing, further optimizing the cost structure.
- Enhanced Supply Chain Reliability: The reliance on readily available bulk chemicals ensures a consistent supply of raw materials. The simplified process flow reduces the potential for bottlenecks and equipment failures, guaranteeing steady production output. This stability allows for better inventory planning and more reliable delivery commitments to downstream partners.
- Scalability and Environmental Compliance: The process is designed for easy scale-up, utilizing standard reactor configurations and safe solvents. The reduction in hazardous waste generation simplifies environmental permitting and lowers disposal costs. This alignment with green chemistry principles enhances the corporate sustainability profile and mitigates regulatory risks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dexrazoxane synthesis technology. These insights are derived directly from the patent data and practical manufacturing considerations, providing clarity for stakeholders evaluating this route for adoption. Understanding these nuances is crucial for making informed decisions about process integration and supply partnerships.
Q: How does the new synthesis route improve product purity compared to traditional methods?
A: The novel route avoids high-temperature decarboxylation and the use of toxic solvents like dioxane, resulting in a white crystalline product with HPLC purity exceeding 99.6%, free from the pink impurities common in older processes.
Q: What are the key raw materials required for this manufacturing process?
A: The process utilizes readily available industrial chemicals including chloroacetonitrile, mineral or organic acids, C1-3 alkyl alcohols (such as ethanol or methanol), and resolved D-(+)-1,2-propanediamine.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method eliminates complex column chromatography and uses simple filtration and crystallization steps, making it highly scalable and easier to industrialize compared to previous multi-step resolution pathways.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dexrazoxane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving oncology therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering dexrazoxane and its salts with stringent purity specifications, leveraging advanced analytical capabilities in our rigorous QC labs to guarantee every batch meets global pharmacopoeia standards. Our expertise in heterocyclic chemistry allows us to optimize this novel route further, ensuring maximum efficiency and consistency for our partners.
We invite you to collaborate with us to unlock the full potential of this advanced manufacturing technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your supply chain optimization goals. Let us be your trusted partner in delivering high-purity pharmaceutical intermediates that drive your success in the global market.
