Advanced Asymmetric Hydrogenation Technology for High-Purity Optically Active Carbonyl Compounds
Advanced Asymmetric Hydrogenation Technology for High-Purity Optically Active Carbonyl Compounds
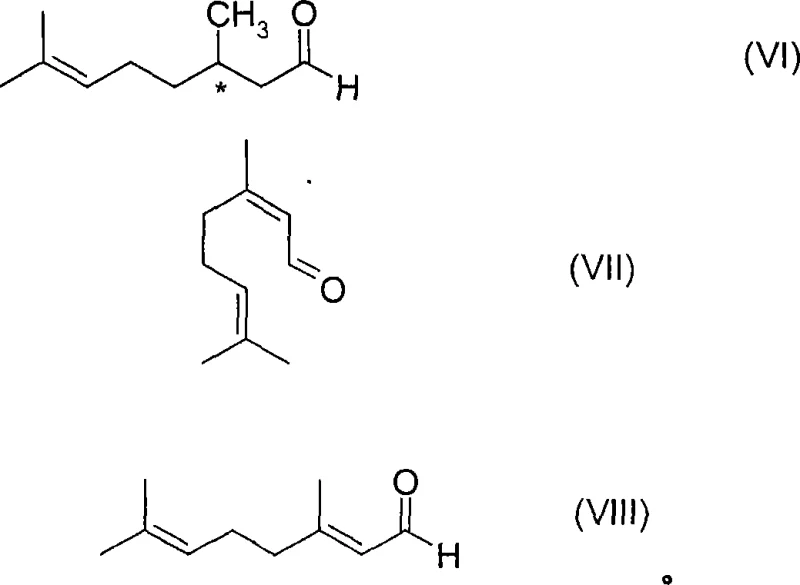
The production of high-value chiral substances remains a cornerstone of modern fine chemical manufacturing, particularly within the flavors, fragrances, and pharmaceutical sectors. Patent CN101039894A introduces a groundbreaking methodology for the preparation of optically active carbonyl compounds through the asymmetric hydrogenation of alpha,beta-unsaturated precursors. This technology specifically addresses the historical instability of homogeneous transition metal catalysts by incorporating carbon monoxide ligands, thereby enabling robust industrial-scale operations. By utilizing soluble optically active transition metal catalysts containing at least one carbon monoxide ligand, this process achieves exceptional enantioselectivity and catalyst longevity. The innovation is particularly transformative for the synthesis of valuable intermediates such as citronellal, a key precursor in the menthol supply chain, offering a reliable pathway for manufacturers seeking to enhance purity profiles while optimizing production efficiency.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the homogeneous catalytic hydrogenation of alpha,beta-unsaturated aldehydes and ketones has been plagued by significant technical hurdles regarding catalyst stability and economic feasibility. Prior art, such as that described in EP-A 0 000 315, utilized rhodium and chiral phosphine complexes which, while effective in laboratory settings, often suffered from rapid deactivation under industrial conditions. The catalytically active metal species or the transition metal complexes formed were frequently insufficiently stable to withstand the rigorous demands of large-scale continuous processing. This instability necessitated high catalyst loading rates, driving up the cost of goods significantly due to the expensive nature of precious metals like rhodium and iridium. Furthermore, the inability to recycle these unstable catalysts resulted in substantial waste generation and complicated downstream purification processes, creating a bottleneck for the cost-effective manufacturing of high-purity fragrance and pharmaceutical intermediates.
The Novel Approach
The novel approach detailed in the patent data fundamentally shifts the paradigm by stabilizing the catalytic species through the strategic integration of carbon monoxide ligands. This method involves either pretreating the catalyst with a gas mixture comprising carbon monoxide and hydrogen or conducting the asymmetric hydrogenation in the presence of additionally fed carbon monoxide. Surprisingly, carbon monoxide, traditionally viewed as a catalyst poison in many hydrogenation contexts, serves here as a crucial stabilizing agent that extends the operational lifetime of the optically active transition metal complex. This stabilization allows for the use of significantly lower catalyst concentrations while maintaining high activity and selectivity. Consequently, the process becomes economically viable for industrial application, enabling the production of optically active aldehydes and ketones with superior enantiomeric excess and facilitating the potential for catalyst reuse in continuous flow systems, which is a critical advancement for supply chain reliability.
Mechanistic Insights into CO-Stabilized Asymmetric Hydrogenation
The core mechanistic advantage of this technology lies in the formation of a robust catalytic cycle involving Group VIII transition metals, preferably Rhodium or Iridium, coordinated with chiral phosphine ligands and carbon monoxide. The transition metal compound, which may be a salt or complex such as Rh(CO)2acac or Rh4(CO)12, reacts with optically active ligands to form the active catalyst species. The presence of the CO ligand modifies the electronic environment of the metal center, preventing aggregation or decomposition pathways that typically deactivate the catalyst during the hydrogenation of the olefinic double bond at the alpha,beta-position. This results in the formation of a new tetrahedral carbon atom at the beta-position with high stereocontrol, yielding non-racemic forms of the target carbonyl compounds. The process accommodates a wide range of substrates, including those with various alkyl, aryl, and heteroaryl substituents, demonstrating remarkable versatility in generating complex chiral architectures required for advanced chemical synthesis.
Furthermore, the control of impurity profiles is intrinsically linked to the stability of the catalyst system. In conventional methods, catalyst degradation often leads to the formation of side products and reduced enantiomeric purity over time. By maintaining the catalyst in its active CO-ligated form through continuous or batch-wise addition of carbon monoxide, the reaction maintains consistent selectivity throughout the conversion. The patent specifies that the process can achieve enantiomeric excess values ranging from 80% to over 99%, depending on the purity of the starting E/Z isomers. This high level of stereochemical control minimizes the need for costly downstream resolution steps. Additionally, the ability to tune the ratio of hydrogen to carbon monoxide in the pretreatment phase allows process chemists to optimize the catalyst preformation, ensuring that the active species is generated efficiently before the substrate is introduced, thereby maximizing yield and minimizing the formation of unwanted by-products.
How to Synthesize Optically Active Carbonyl Compounds Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to leverage the full potential of the CO-stabilized catalyst system. The process generally begins with the dissolution of the transition metal precursor and the chiral ligand in an inert solvent or the substrate itself, followed by a critical pretreatment step with synthesis gas. This preformation ensures the generation of the active catalytic species prior to hydrogenation. Detailed operational protocols involve specific pressure ranges, typically between 50 to 100 bar, and temperature controls to prevent isomerization of the double bond. For a comprehensive understanding of the exact stoichiometric ratios, solvent choices, and safety protocols required for implementation, please refer to the standardized synthesis guidelines provided below.
- Prepare the catalyst by reacting a soluble transition metal compound (e.g., Rhodium or Iridium) with a chiral phosphine ligand in an inert solvent.
- Pretreat the catalyst mixture with a gas blend of hydrogen and carbon monoxide at elevated pressure and temperature to form the active CO-ligand species.
- Introduce the alpha,beta-unsaturated carbonyl substrate and perform asymmetric hydrogenation under controlled pressure, maintaining CO presence to ensure catalyst stability and high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this asymmetric hydrogenation technology presents a compelling value proposition centered around cost efficiency and supply continuity. The primary economic driver is the drastic improvement in catalyst turnover numbers (TON), with experimental data indicating TON values exceeding 10,000 in continuous operations. This efficiency translates directly into a significant reduction in the consumption of expensive precious metal catalysts, which is a major cost component in fine chemical manufacturing. By extending the life of the catalyst and enabling its potential reuse or recycling, the overall cost of production for optically active intermediates is substantially lowered, providing a competitive edge in pricing strategies for downstream fragrance and pharmaceutical products.
- Cost Reduction in Manufacturing: The elimination of frequent catalyst replacement and the ability to operate with lower metal loadings directly reduce raw material costs. Since the catalyst remains stable due to CO coordination, the process avoids the economic losses associated with catalyst deactivation and the subsequent need for fresh catalyst charges. This stability also simplifies the purification process, as there is less metal contamination to remove, reducing the burden on downstream processing units and lowering utility costs associated with purification steps like distillation or chromatography.
- Enhanced Supply Chain Reliability: The robustness of the catalyst system ensures consistent production output, minimizing the risk of batch failures that can disrupt supply schedules. The ability to run the process in a continuous mode, as demonstrated in the patent examples, allows for a steady stream of product, which is essential for meeting the demanding delivery timelines of global multinational corporations. Furthermore, the use of readily available starting materials like geranial and neral, combined with a stable catalytic process, mitigates the risk of supply bottlenecks often associated with more fragile or exotic synthetic routes.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale-up, capable of transitioning from laboratory autoclaves to large-scale continuous reactors without loss of efficiency. From an environmental perspective, the high selectivity and yield minimize waste generation, aligning with green chemistry principles. The potential for solvent-free operation, where the substrate acts as the medium, further reduces the environmental footprint by eliminating the need for large volumes of organic solvents, thereby simplifying waste treatment and ensuring compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this process for their supply chains.
Q: How does the addition of carbon monoxide improve catalyst stability in this process?
A: According to patent CN101039894A, carbon monoxide acts as a stabilizing ligand for the transition metal catalyst (such as Rhodium), preventing decomposition and allowing for significantly extended catalyst lifetime and potential reuse in continuous processes.
Q: What represents the primary commercial advantage of this asymmetric hydrogenation method?
A: The primary advantage is the ability to operate on an industrial scale with high turnover numbers (TON) due to enhanced catalyst stability, which drastically reduces the cost per kilogram of high-purity optically active products like citronellal.
Q: Can this method be applied to substrates other than citronellal precursors?
A: Yes, the patent describes a broad scope covering various alpha,beta-unsaturated aldehydes and ketones, including cyclic and acyclic structures, making it versatile for producing diverse pharmaceutical and fragrance intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Citronellal Supplier
The technological advancements described in patent CN101039894A represent a significant leap forward in the synthesis of optically active carbonyl compounds, particularly for the production of high-purity citronellal. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our rigorous QC labs and stringent purity specifications guarantee that every batch meets the exacting standards required by the global flavors and pharmaceutical industries, making us a trusted partner for complex chiral synthesis.
We invite you to engage with our technical procurement team to discuss how this advanced hydrogenation technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits specific to your production needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial viability.
