Revolutionizing Chiral Carbonyl Synthesis: Advanced Asymmetric Hydrogenation for Industrial Scale
Revolutionizing Chiral Carbonyl Synthesis: Advanced Asymmetric Hydrogenation for Industrial Scale
The landscape of fine chemical manufacturing is constantly evolving, driven by the demand for higher purity chiral intermediates and more sustainable production methodologies. Patent CN101675020B introduces a groundbreaking approach to synthesizing optically active carbonyl compounds, specifically addressing the longstanding challenges associated with catalyst stability and process control in asymmetric hydrogenation. This technology leverages the unique properties of transition metal catalysts soluble in the reaction mixture, which possess at least one carbon monoxide ligand. By meticulously controlling the carbon monoxide concentration during both the catalyst pretreatment and the actual hydrogenation phases, this method achieves superior enantioselectivity and operational longevity. For industry leaders seeking a reliable fragrance intermediate supplier or partners for complex pharmaceutical synthesis, this patented process represents a significant leap forward in process chemistry, enabling the efficient production of high-value aldehydes and ketones such as citronellal.

The limitations of conventional methods for producing optically active aldehydes and ketones have long hindered the economic feasibility of large-scale operations. Traditional homogeneous catalytic systems, often relying on rhodium and chiral phosphine complexes, suffer from insufficient stability of the catalyst complexes or the catalytically active metals. In many prior art methods, such as those described in EP-A 0000315, the catalyst tends to decompose or lose activity rapidly, necessitating frequent replenishment and driving up operational costs. Furthermore, controlling the reaction environment to prevent side reactions while maintaining high enantiomeric excess has proven difficult. The novel approach detailed in this patent overcomes these hurdles by introducing a specific pretreatment protocol where the catalyst precursor is exposed to a gas mixture of carbon monoxide and hydrogen under pressure. This generates a robust catalyst species stabilized by CO ligands. Crucially, the process involves removing excess CO before the main reaction and then reintroducing it at trace levels (100-1200 ppm) during hydrogenation. This delicate balance ensures the catalyst remains active and stable throughout the cycle, solving the degradation issues that plague older technologies.
Mechanistic Insights into CO-Stabilized Asymmetric Hydrogenation
The core of this technological advancement lies in the precise manipulation of the coordination sphere around the transition metal center, typically rhodium or iridium. The mechanism involves the formation of a catalytically active species where the metal is coordinated with chiral diphosphine ligands, such as chiraphos, and critically, at least one carbon monoxide molecule. This CO ligand acts as a stabilizer, preventing the aggregation of metal atoms or the formation of inactive clusters that typically deactivate homogeneous catalysts. The process begins with the pretreatment of the catalyst precursor at pressures ranging from 5 to 100 bar with a synthesis gas mixture. This step ensures the complete formation of the carbonyl-hydride species necessary for the catalytic cycle. Following this, the removal of excess free carbon monoxide is vital; if left unchecked, high concentrations of CO would poison the catalyst by occupying too many coordination sites, thereby inhibiting the binding of the substrate and hydrogen. By stripping the excess CO and then maintaining a low, steady-state concentration of 100-1200 ppm during the reaction, the system achieves an optimal equilibrium where the catalyst is protected from decomposition yet remains accessible for the substrate.
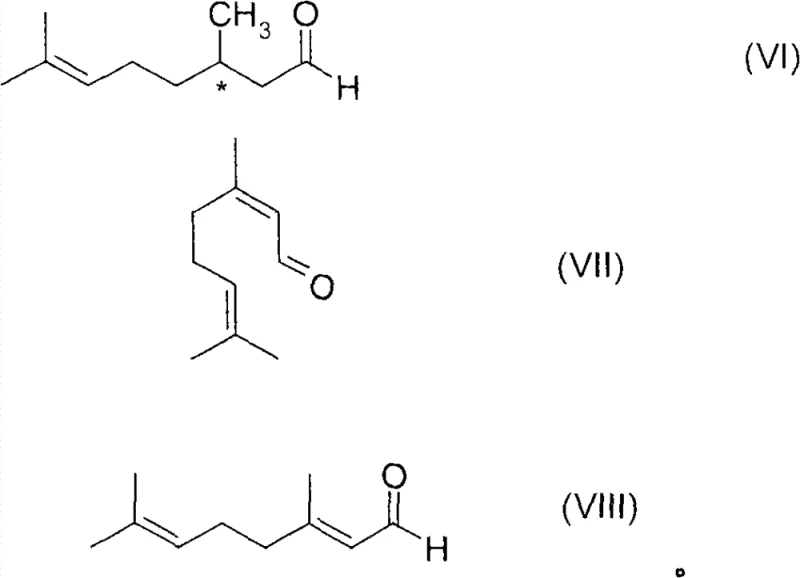
This mechanistic precision allows for exceptional control over the stereochemistry of the product. The chiral environment created by ligands like (R,R)-chiraphos directs the hydrogen addition to the alpha,beta-unsaturated double bond with high fidelity. The patent highlights that this method is particularly effective for substrates like neral and geranial, converting them into optically active citronellal with enantiomeric excess values often exceeding 80% and reaching up to 99% under optimized conditions. The ability to tune the E/Z isomer ratio of the starting material further enhances the optical purity of the final product. This level of control is essential for applications in the flavors and fragrances industry, where the sensory profile is strictly dependent on the specific enantiomer produced. The stabilization provided by the CO ligand also facilitates the potential for catalyst recycling, a feature that is rarely achievable in standard homogeneous hydrogenation processes without significant loss of activity.
How to Synthesize Optically Active Citronellal Efficiently
The synthesis of high-purity optically active citronellal serves as a prime example of the utility of this patented technology. The process is designed to be robust enough for industrial application while maintaining the rigorous stereochemical control required for fine chemicals. The procedure involves dissolving the transition metal compound and the chiral ligand in a suitable solvent or directly in the substrate, followed by the critical pressurization with the CO/H2 mixture. After the activation period, the pressure is carefully released to remove excess CO before the main hydrogenation charge is introduced. This sequence ensures that the catalyst enters the reaction zone in its most active and stable form. For detailed operational parameters regarding temperature, pressure, and specific catalyst loading ratios, please refer to the standardized synthesis protocols derived from the patent data.
- Pretreat the catalyst precursor with a gas mixture containing 20-90% carbon monoxide and 10-80% hydrogen at 5-100 bar pressure to form the active CO-ligand complex.
- Remove excess unbound carbon monoxide from the pretreated catalyst mixture by depressurization or stripping with inert gas before the hydrogenation step.
- Perform asymmetric hydrogenation of the alpha,beta-unsaturated substrate in the presence of hydrogen containing a controlled low concentration of carbon monoxide (100-1200 ppm).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the implementation of this CO-stabilized asymmetric hydrogenation technology offers profound advantages for procurement and supply chain management within the fine chemical sector. The primary benefit stems from the enhanced stability of the catalyst system. In traditional processes, the rapid degradation of expensive noble metal catalysts like rhodium necessitates high consumption rates and complex recovery procedures, which inflate the cost of goods sold. By extending the lifespan of the catalyst through CO stabilization, this method significantly reduces the consumption of precious metals. This reduction translates directly into substantial cost savings in flavor manufacturing, allowing for more competitive pricing structures without compromising on quality. Furthermore, the ability to operate the process continuously, as demonstrated in the patent examples, improves throughput and reduces downtime associated with batch processing and catalyst changeovers.
Supply chain reliability is another critical area where this technology excels. The process is designed to be scalable from laboratory benchtops to multi-ton industrial reactors, ensuring a consistent supply of high-purity intermediates. The robustness of the catalyst against deactivation means that production schedules are less likely to be disrupted by unexpected catalyst failures or the need for frequent recharging. This predictability is invaluable for supply chain heads managing just-in-time inventory for downstream pharmaceutical or fragrance production. Additionally, the process eliminates the need for stoichiometric amounts of additives or complex workup procedures often required to remove degraded catalyst residues. This simplification of the downstream processing not only reduces waste generation but also shortens the overall production cycle time. Consequently, manufacturers can respond more agilely to market demands, reducing lead time for high-purity fragrance intermediates and securing a stronger position in the global supply chain.
Environmental compliance and scalability are inherently improved by this methodology. The use of a homogeneous catalyst that can be stabilized and potentially recycled aligns with green chemistry principles by minimizing heavy metal waste. The process operates under relatively mild temperatures and pressures compared to some alternative high-energy synthetic routes, contributing to a lower carbon footprint. The ability to run the reaction in a continuous mode using gas循环 reactors further enhances energy efficiency and safety profiles. For organizations aiming to scale up complex fragrance intermediates, this technology provides a clear pathway to commercialization that meets stringent regulatory standards for impurity profiles and residual metal content. The combination of high yield, high enantiomeric excess, and operational stability makes this a superior choice for long-term manufacturing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the operational boundaries and potential applications of the method. Understanding these nuances is crucial for R&D teams evaluating the feasibility of adopting this process for their specific product portfolios.
Q: How does the addition of carbon monoxide improve catalyst stability in this process?
A: The process utilizes a transition metal catalyst, typically rhodium or iridium, coordinated with at least one carbon monoxide ligand. This CO ligand stabilizes the catalytically active species, preventing decomposition and allowing for catalyst reuse, which is a common failure point in conventional homogeneous hydrogenation systems.
Q: What is the optimal carbon monoxide concentration during the hydrogenation step?
A: While the catalyst pretreatment uses high concentrations of CO, the actual asymmetric hydrogenation step requires a strictly controlled low concentration of carbon monoxide, specifically between 100 ppm and 1200 ppm in the hydrogen gas stream, to maintain activity without inhibiting the reaction.
Q: Can this method be applied to the synthesis of menthol precursors?
A: Yes, the method is particularly effective for converting neral or geranial into optically active citronellal with high enantiomeric excess. This citronellal serves as a critical intermediate for the subsequent cyclization and hydrogenation steps required to produce high-purity optically active menthol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Carbonyl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN101675020B for the production of high-value chiral intermediates. Our commitment to excellence is reflected in our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that the transition from a patented laboratory method to a robust industrial process requires deep expertise in process engineering and quality control. Our facilities are equipped with state-of-the-art high-pressure hydrogenation reactors and rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of optically active carbonyl compounds meets the exacting standards required by the global pharmaceutical and fragrance industries.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. Whether you require custom synthesis of complex aldehydes or large-scale production of key fragrance ingredients like citronellal, our technical team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and enhance your product quality.
