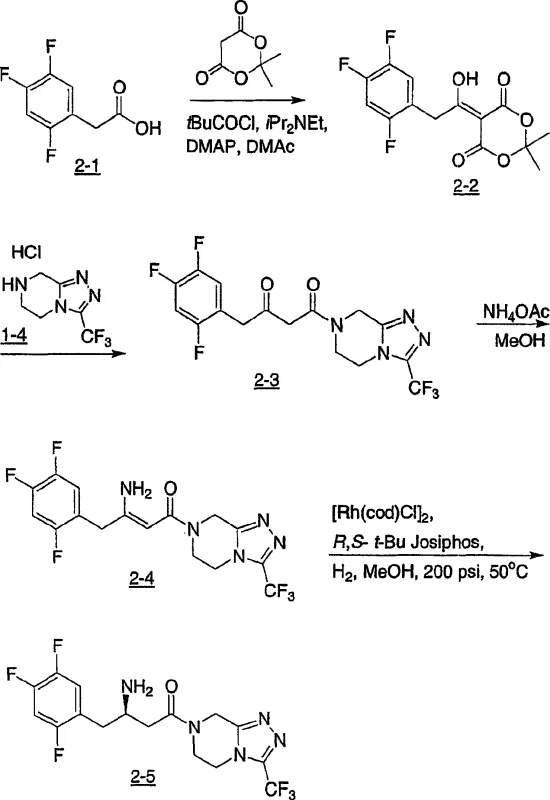
Advanced Asymmetric Hydrogenation for High-Purity Chiral Beta-Amino Acid Intermediates
Advanced Asymmetric Hydrogenation for High-Purity Chiral Beta-Amino Acid Intermediates
The pharmaceutical industry continuously seeks more efficient pathways to construct chiral building blocks, particularly for the synthesis of biologically active molecules like peptidomimetics and enzyme inhibitors. Patent CN1761642A introduces a groundbreaking methodology for the efficient preparation of enantiomerically enriched β-amino acid derivatives. This technology leverages transition metal-catalyzed asymmetric hydrogenation of prochiral β-aminoacrylic acid derivatives in the presence of chiral ferrocenyldiphosphine ligands. Unlike traditional approaches that necessitate cumbersome protection and deprotection sequences, this novel process allows for the direct hydrogenation of substrates containing unprotected primary amino groups. For R&D directors and procurement specialists, this represents a pivotal shift towards greener, more cost-effective manufacturing of high-value pharmaceutical intermediates, specifically those utilized in the production of Type 2 diabetes treatments such as DPP-IV inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric synthesis of β-amino acids has been plagued by inefficiencies inherent to functional group management. Prior art, including methodologies described by Hayashi, Zhu, and Lubell, predominantly relies on the use of N-acetylated or otherwise protected β-aminoacrylic acid derivatives as substrates for hydrogenation. The requirement for amine protection introduces at least two additional chemical steps into the synthetic route: the initial protection of the amine prior to hydrogenation and the subsequent deprotection to reveal the free amine functionality. These extra steps not only increase the consumption of reagents and solvents but also generate significant amounts of chemical waste, thereby negatively impacting the overall atom economy and environmental footprint of the process. Furthermore, the synthesis of these protected reactants can often be chemically challenging, leading to variable yields and complicating the purification of intermediates, which ultimately drives up the cost of goods for the final active pharmaceutical ingredient.
The Novel Approach
The methodology disclosed in CN1761642A circumvents these historical bottlenecks by enabling the enantioselective hydrogenation of β-aminoacrylic acid derivatives where the amino group remains completely unprotected. This streamlined approach significantly reduces the step count and simplifies the overall process flow. As illustrated in the reaction schemes below, the process converts prochiral enamines directly into chiral β-amino acids with high stereoselectivity. The ability to tolerate free amine groups during the catalytic cycle is a major technical achievement, eliminating the need for acylation and hydrolysis steps. This results in a drastic reduction in processing time and raw material costs, making it an highly attractive option for the commercial scale-up of complex pharmaceutical intermediates. The robustness of this method is demonstrated in the synthesis of specific DPP-IV inhibitor precursors, where high yields and excellent optical purity are achieved in a single catalytic step.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the precise coordination chemistry between the transition metal precursor and the chiral ligand. The process utilizes a catalytic system formed from a rhodium precursor, such as [Rh(cod)Cl]2, complexed with a chiral ferrocenyldiphosphine ligand, specifically variants of the Josiphos family. The unique electronic and steric properties of ligands like (R,S)-tert-butyl Josiphos create a highly defined chiral environment around the rhodium center. This chiral pocket effectively differentiates between the enantiotopic faces of the prochiral carbon-carbon double bond in the enamine substrate. During the catalytic cycle, the substrate coordinates to the metal center, and the delivery of molecular hydrogen occurs with strict stereochemical control, dictated by the bulky tert-butyl groups and the planar chirality of the ferrocene backbone. This ensures that the hydrogen atoms are added to the specific face required to generate the desired (R)- or (S)-configuration at the newly formed stereogenic center.
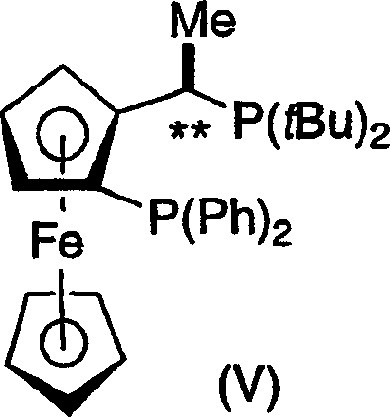
Furthermore, the mechanism inherently supports high levels of impurity control, which is critical for regulatory compliance in API manufacturing. The high enantioselectivity of the catalyst minimizes the formation of the unwanted enantiomer at the source, rather than relying solely on downstream purification. In the specific embodiment described for the synthesis of compound 2-5, the hydrogenation proceeds with an initial optical purity of approximately 94% ee. The process design includes a subsequent crystallization step that leverages the solubility differences between the enantiomers to upgrade the optical purity to greater than 98-99% ee. This combination of highly selective catalysis and effective crystallization ensures that the final product meets the stringent purity specifications required for biological testing and clinical applications, reducing the risk of toxicological issues associated with chiral impurities.
How to Synthesize Chiral Beta-Amino Acid Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a laboratory or pilot plant setting. The process begins with the preparation of the enamine substrate from readily available beta-keto esters or acids using simple ammonia sources like ammonium acetate. Following substrate preparation, the active catalyst is generated in situ or pre-formed by mixing the rhodium dimer with the chiral ligand in a suitable solvent such as methanol. The hydrogenation is then conducted under moderate pressure and temperature conditions, ensuring safety and scalability. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Preparation of the prochiral beta-aminoacrylic derivative (enamine) from the corresponding beta-keto ester or acid using an ammonia source like ammonium acetate.
- Formation of the active catalytic species by mixing a Rhodium precursor such as [Rh(cod)Cl]2 with a chiral ferrocenyldiphosphine ligand like (R,S)-t-Bu Josiphos in methanol.
- Execution of the hydrogenation reaction under controlled pressure (e.g., 200 psi) and temperature (50°C), followed by crystallization to enhance optical purity to >98% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this asymmetric hydrogenation technology offers substantial strategic advantages beyond mere technical elegance. The primary value driver is the significant reduction in manufacturing costs achieved through process intensification. By eliminating the protection and deprotection steps, the process reduces the total volume of solvents and reagents required, lowers energy consumption associated with additional reaction and isolation steps, and minimizes waste disposal costs. This streamlined workflow translates directly into a lower cost of goods sold (COGS), providing a competitive edge in pricing negotiations for long-term supply contracts. Additionally, the reduction in unit operations shortens the overall production cycle time, enhancing the responsiveness of the supply chain to market demands.
- Cost Reduction in Manufacturing: The removal of amine protection steps fundamentally alters the cost structure of producing chiral beta-amino acids. Traditional routes incur costs for protecting group reagents, additional solvents for washing and extraction, and the labor associated with managing extra synthetic stages. By performing the hydrogenation on the unprotected enamine, the process achieves a higher atom economy and reduces the physical footprint required for production. This efficiency gain allows for substantial cost savings without compromising on the quality or purity of the final intermediate, making it an economically superior choice for large-scale API manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the Rhodium-Josiphos catalyst system contributes to greater supply chain stability. The reagents involved, including the rhodium precursor and the commercially available Josiphos ligands, are well-established in the fine chemical industry, ensuring consistent availability and reducing the risk of supply disruptions. Furthermore, the process operates under relatively mild conditions (e.g., 50°C and moderate hydrogen pressure), which reduces the stress on equipment and lowers the likelihood of batch failures due to thermal runaway or pressure excursions. This reliability ensures consistent delivery schedules and helps maintain inventory levels for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns perfectly with modern green chemistry principles. The reduction in chemical waste and solvent usage simplifies effluent treatment and lowers the environmental burden of the manufacturing site. The scalability of the hydrogenation step is well-documented, with the patent citing successful operations at pressures suitable for industrial reactors. This ease of scale-up facilitates the transition from kilogram-scale development to multi-ton commercial production, ensuring that the supply chain can grow seamlessly alongside the clinical and commercial success of the downstream drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and embodiments disclosed in the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these details is crucial for making informed decisions about process adoption and supplier selection.
Q: Why is the elimination of amine protection groups significant in this synthesis?
A: Conventional methods require N-acetylation and subsequent deprotection, adding two synthetic steps and generating additional waste. This patent enables direct hydrogenation of unprotected enamines, drastically improving atom economy and reducing processing time.
Q: What level of enantiomeric excess (ee) can be achieved with this process?
A: The process typically yields products with greater than 90% ee directly from hydrogenation. Through optimized crystallization techniques described in the patent, optical purity can be further upgraded to exceed 98-99% ee, meeting stringent pharmaceutical standards.
Q: Is this method scalable for industrial production of DPP-IV inhibitors?
A: Yes, the patent explicitly mentions applicability on pilot and industrial scales. The use of robust Rhodium-Josiphos catalysts and standard hydrogenation equipment (up to 200 psi) ensures the process is viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Beta-Amino Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient chiral synthesis in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN1761642A can be successfully translated into robust manufacturing processes. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle the specific requirements of transition metal catalysis, including safe hydrogenation protocols and advanced purification techniques to guarantee optimal enantiomeric excess.
We invite you to collaborate with us to leverage this advanced technology for your pipeline projects. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this streamlined route can optimize your budget. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and to discuss how we can support your supply chain with reliable, high-quality chiral building blocks.
