Advanced Organocatalytic Process for Enantiomerically Enriched Beta-Amino Acid Derivatives Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways for the production of high-value chiral intermediates, particularly enantiomerically enriched beta-amino acid derivatives which serve as critical building blocks for antibiotics, alkaloids, and modified peptides. Patent CN101248037A introduces a groundbreaking organocatalytic kinetic resolution method that addresses the long-standing challenges associated with the stereoselective synthesis of these compounds. This technology leverages a novel approach involving 4,5-dihydrooxazin-6-one intermediates and chiral thiourea catalysts to achieve high enantiomeric excess without the need for stoichiometric chiral auxiliaries. By shifting from traditional resolution techniques to this catalytic system, manufacturers can significantly enhance the ecological and economic profile of their production lines. The patent details a robust process that allows for the preparation of both N-acylated beta-amino acid esters and the corresponding free acids through simple hydrolysis, offering remarkable flexibility for downstream processing. This innovation represents a substantial leap forward in the field of asymmetric synthesis, providing a reliable foundation for the commercial manufacturing of complex pharmaceutical intermediates with superior purity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of optically active beta-amino acids has been fraught with significant technical and economic hurdles that limit scalability and increase production costs. Traditional methods often rely on classical racemic resolution through diastereomeric salt formation or diastereoselective synthesis using stoichiometric amounts of chiral reagents such as optically active lithium phenylacetamide. These conventional routes are inherently inefficient because they require equimolar quantities of expensive chiral sources that cannot be recycled, leading to substantial material waste and elevated raw material costs. Furthermore, many of these processes necessitate the use of hazardous and expensive auxiliaries like n-butyllithium for deprotonation, which poses serious industrial safety risks and requires specialized handling protocols. A critical drawback of these legacy methods is the requirement for cryogenic reaction conditions, often around -70°C, to maintain adequate stereoselectivity, which imposes heavy energy demands and necessitates specialized reactor materials capable of withstanding extreme low temperatures. Additionally, biocatalytic alternatives, while effective for aromatic substrates, often fail to accommodate aliphatic or sterically demanding substrates due to enzyme specificity limitations, restricting the scope of applicable chemical structures.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method disclosed in patent CN101248037A utilizes a catalytic kinetic resolution strategy that operates under mild and industrially friendly conditions. This novel approach initiates with racemic 4,5-dihydrooxazin-6-ones, which are reacted with a nucleophile in the presence of a catalytic amount of an enantiomerically enriched thiourea derivative. The process eliminates the need for stoichiometric chiral reagents and cryogenic cooling, instead proceeding optimally at temperatures between 15°C and 40°C, which drastically reduces energy consumption and equipment complexity. The use of modular organocatalysts allows for fine-tuning of the stereochemical outcome, enabling the successful resolution of both aromatic and aliphatic substrates, including sterically hindered examples like beta-neopentylglycine that were previously inaccessible via enzymatic routes. This methodology not only simplifies the reaction setup by using common aprotic solvents like toluene or dichloromethane but also ensures that both enantiomers of the beta-amino acid can be recovered from a single reaction batch, effectively doubling the theoretical yield compared to traditional resolution methods. 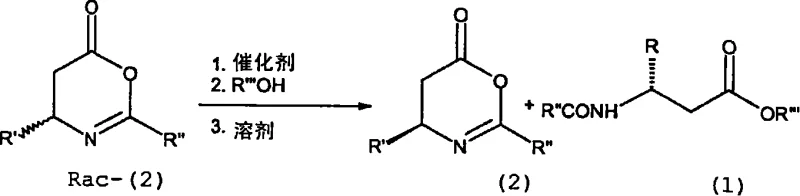
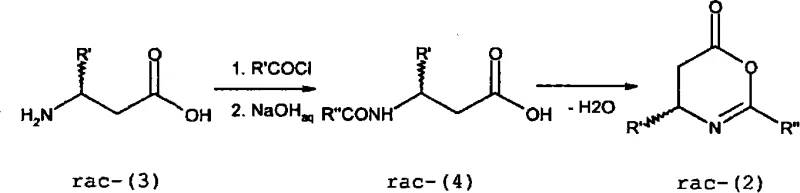
Mechanistic Insights into Organocatalytic Kinetic Resolution
The core of this technological advancement lies in the sophisticated mechanism of the organocatalytic kinetic resolution, which relies on the precise activation of the oxazinone substrate through hydrogen bonding interactions. The chiral thiourea catalyst, typically used in loading ranges from 0.5 mol% to 10 mol%, acts as a bifunctional activator that simultaneously coordinates with the carbonyl group of the oxazinone and the incoming nucleophile, such as allyl alcohol or water. This dual activation lowers the energy barrier for the nucleophilic attack on one specific enantiomer of the racemic substrate, leading to a highly stereoselective ring-opening reaction that yields an enantiomerically enriched beta-amino acid ester. As the reaction progresses, the unreacted oxazinone becomes increasingly enriched in the opposite enantiomer, allowing for the isolation of both chiral forms with high optical purity, often exceeding 98% ee under optimized conditions. The modular nature of the thiourea catalyst structure permits chemical modifications to the aryl or alkyl substituents, enabling process chemists to adapt the catalyst to specific substrate requirements and maximize enantioselectivity for diverse chemical libraries. This level of control over the stereochemical outcome is critical for pharmaceutical applications where even trace impurities can impact drug safety and efficacy.
Impurity control is inherently built into this mechanism due to the high chemoselectivity of the organocatalyst, which minimizes side reactions such as polymerization or non-selective hydrolysis that are common in metal-catalyzed systems. The reaction pathway avoids the use of transition metals, thereby eliminating the risk of heavy metal contamination in the final product, a crucial consideration for regulatory compliance in API manufacturing. The resulting N-acylated beta-amino acid esters can be easily converted to the free acids through standard acidic or basic hydrolysis without racemization, preserving the hard-won enantiomeric purity throughout the downstream processing steps. Furthermore, the compatibility of the catalyst with a wide range of nucleophiles, including amines and thiols, expands the synthetic utility of this method beyond simple ester formation, allowing for the direct synthesis of diverse beta-amino acid derivatives. The robustness of the catalytic cycle ensures consistent performance across multiple batches, providing the reliability needed for continuous manufacturing processes. 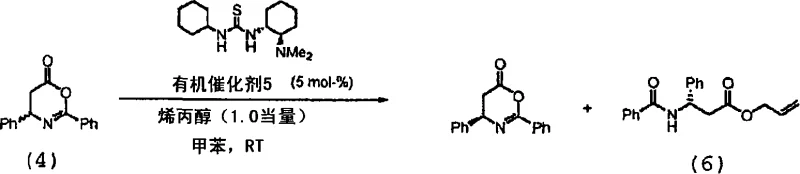
How to Synthesize Beta-Amino Acid Derivatives Efficiently
Implementing this synthesis route in a production environment begins with the preparation of the racemic oxazinone starting material, which is readily accessible from commercially available beta-amino acids through a straightforward Schotten-Baumann acylation followed by cyclization under dehydrating conditions. Once the substrate is prepared, the kinetic resolution is conducted by dissolving the oxazinone in an inert organic solvent such as toluene and adding the chiral thiourea catalyst along with the chosen nucleophile, typically allyl alcohol for ester formation. The reaction mixture is maintained at a mild temperature range of 20°C to 30°C, allowing the catalytic cycle to proceed with high efficiency while minimizing thermal stress on the reagents and equipment. Detailed standardized synthesis steps see the guide below.
- Prepare the racemic 4,5-dihydrooxazin-6-one starting material from beta-amino acids via acylation and cyclization.
- Perform kinetic racemic resolution using a chiral thiourea catalyst and a nucleophile like allyl alcohol.
- Hydrolyze the resulting enantiomerically enriched esters and oxazinones to obtain the free beta-amino acids.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organocatalytic process translates into tangible strategic advantages that directly impact the bottom line and operational resilience. By replacing stoichiometric chiral reagents with catalytic amounts of organocatalysts, the material cost per kilogram of product is significantly reduced, as the expensive chiral source is no longer consumed in equimolar quantities but rather recycled through the catalytic cycle. The elimination of cryogenic cooling requirements removes the need for specialized low-temperature reactors and reduces energy consumption drastically, leading to lower utility costs and a smaller carbon footprint for the manufacturing facility. Furthermore, the avoidance of hazardous reagents like n-butyllithium simplifies safety protocols and reduces the costs associated with hazardous waste disposal and regulatory compliance. The ability to produce both enantiomers from a single racemic starting material effectively doubles the yield of valuable chiral intermediates from the same amount of raw material, optimizing resource utilization and reducing the overall cost of goods sold. These factors combine to create a supply chain that is not only more cost-effective but also more robust against fluctuations in the availability of specialized reagents.
- Cost Reduction in Manufacturing: The transition from stoichiometric chiral auxiliaries to catalytic organocatalysts removes the burden of purchasing large quantities of expensive chiral pool materials that are traditionally discarded after a single use. This shift fundamentally changes the cost structure of the synthesis, as the catalyst loading is minimal and the potential for catalyst recovery or reuse further enhances economic efficiency. Additionally, the mild reaction conditions eliminate the high energy costs associated with maintaining cryogenic temperatures, resulting in substantial savings on utilities and equipment maintenance. The simplified workup procedures, which avoid complex metal removal steps, further reduce processing time and labor costs, contributing to an overall leaner manufacturing operation.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable organocatalysts rather than sensitive organometallic reagents ensures a more stable and predictable supply of critical processing materials. Since the reaction does not depend on specialized enzymes with narrow substrate scopes, the process is adaptable to a wider range of raw materials, reducing the risk of supply bottlenecks caused by specific precursor shortages. The use of common industrial solvents like toluene and acetonitrile means that solvent procurement is straightforward and not subject to the volatility of specialized chemical markets. This flexibility allows for faster response times to changes in demand and ensures continuous production capability even when specific niche reagents are unavailable.
- Scalability and Environmental Compliance: The absence of heavy metal catalysts simplifies the regulatory approval process for pharmaceutical intermediates, as there is no need for extensive and costly testing to ensure residual metal levels are within strict limits. The process generates less hazardous waste compared to traditional methods that use stoichiometric lithium reagents, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The mild operating conditions and standard pressure requirements make the technology easily scalable from laboratory to commercial production volumes without the need for significant re-engineering of the process. This scalability ensures that supply can be ramped up quickly to meet market demand without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this organocatalytic kinetic resolution technology for beta-amino acid production. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing workflows. The information covers catalyst performance, substrate scope, and downstream processing considerations.
Q: What are the advantages of this organocatalytic method over traditional resolution?
A: This method avoids stoichiometric chiral reagents and expensive auxiliaries like n-butyllithium, operating at mild temperatures between 15°C and 40°C instead of cryogenic conditions.
Q: Can this process be scaled for industrial production of beta-amino acids?
A: Yes, the use of catalytic amounts of modular thiourea catalysts and common organic solvents like toluene makes the process highly suitable for commercial scale-up.
Q: What types of beta-amino acid derivatives can be synthesized using this route?
A: The method is particularly effective for both aromatic and aliphatic substituted beta-amino acids, including sterically demanding substrates like beta-neopentylglycine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Amino Acid Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing innovation, leveraging advanced technologies like the organocatalytic kinetic resolution described in patent CN101248037A to deliver superior pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel chemistry are fully realized in large-scale operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of beta-amino acid derivatives meets the highest international standards for enantiomeric excess and chemical purity. Our commitment to technical excellence allows us to navigate the complexities of chiral synthesis, providing our partners with a reliable source of high-quality intermediates that accelerate their drug development timelines.
We invite global pharmaceutical and chemical companies to collaborate with us to optimize their supply chains and reduce manufacturing costs through the adoption of this efficient synthesis route. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your project's success. Let us partner with you to bring these advanced chemical solutions from the laboratory to the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
