Advanced Ruthenium-Catalyzed Asymmetric Michael Addition for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously demands high-purity chiral intermediates to ensure the safety and efficacy of final drug products. Patent CN1626501A introduces a groundbreaking methodology for the preparation of optically active nitro compounds and cyano compounds, which serve as critical building blocks for numerous therapeutic agents, including GABA-B receptor agonists like Baclofen and endothelin antagonists. This technology leverages a sophisticated asymmetric Michael addition reaction catalyzed by a novel metal complex derived from optically active nitrogenous compounds and Periodic Table Group VIII metals, specifically ruthenium. By shifting away from traditional, harsh synthetic routes, this patent offers a pathway to achieve exceptional enantiomeric excess (up to 98% ee) and high yields under significantly milder conditions. For R&D directors and procurement specialists, understanding this catalytic innovation is key to securing a reliable pharmaceutical intermediate supplier capable of delivering complex chiral molecules with stringent purity specifications.
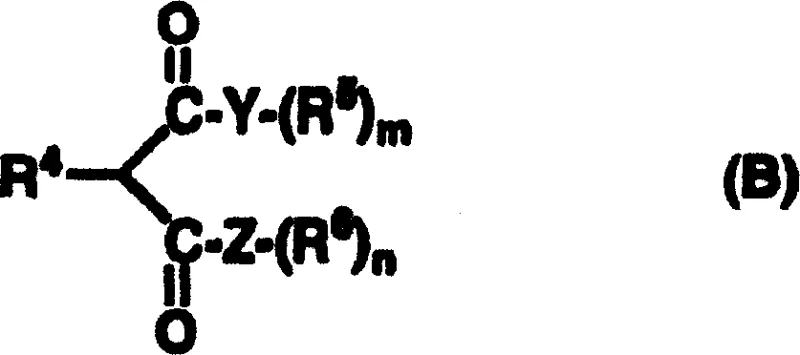
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active nitro compounds has been fraught with significant technical and operational challenges that hinder efficient commercial manufacturing. Conventional methods often rely on asymmetric metal catalysts based on rhodium, copper, or magnesium, each presenting distinct drawbacks. For instance, rhodium-catalyzed processes frequently necessitate the use of alkylboronic acids as nucleophilic reagents, which must be pre-modulated or added in excess, complicating the reaction workflow and increasing raw material costs. Furthermore, copper-based systems typically employ unstable dialkyl zinc reagents that are pyrophoric and hazardous to handle on a large scale, requiring rigorous safety protocols and specialized equipment. Perhaps most critically, many of these legacy processes demand cryogenic reaction temperatures ranging from -30°C to -78°C to achieve acceptable enantioselectivity, leading to substantial energy consumption and limiting the practical throughput of manufacturing facilities. Additionally, magnesium complex methods often require high catalyst loadings (around 5 mol%) alongside promoters like molecular sieves, yet still struggle to provide sufficient deprotonation promotion, resulting in lower overall efficiency.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a highly efficient asymmetric metal complex formed by the interaction of an optically active nitrogenous compound and a ruthenium complex. This system fundamentally alters the reaction landscape by enabling the asymmetric Michael addition to proceed at ambient or near-ambient temperatures, typically between -30°C and 40°C, thereby eliminating the need for energy-intensive cryogenic cooling. The catalyst system exhibits remarkable versatility, effectively accommodating a wide range of electron-withdrawing groups on the Michael acceptor, including both nitro and cyano functionalities, which was previously a point of uncertainty for basic catalysts. By employing specific chiral diamine ligands coordinated to ruthenium, the process achieves high stereoselectivity with significantly reduced catalyst loading compared to older magnesium-based methods. This advancement not only simplifies the operational procedure but also enhances the safety profile by avoiding unstable organometallic reagents, positioning it as a superior choice for cost reduction in API manufacturing.
Mechanistic Insights into Ruthenium-Catalyzed Asymmetric Michael Addition
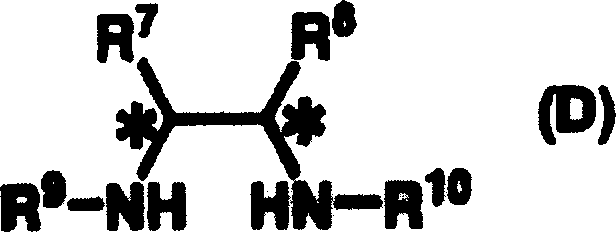
The core of this technological breakthrough lies in the precise design of the chiral catalyst, which creates a highly defined steric environment to control the stereochemical outcome of the Michael addition. The active catalytic species is generated through the coordination of a chiral diamine derivative, represented by General Formula (D), with a ruthenium precursor. This interaction forms either an asymmetric ruthenium amide complex or a ruthenium hydride complex, depending on the presence of hydrogen donors during preparation. The chiral diamine ligand, often featuring bulky aryl sulfonyl groups such as p-toluenesulfonyl or 2,4,6-triisopropylbenzenesulfonyl, imparts significant steric bulk around the metal center. This bulk is crucial for differentiating the enantiotopic faces of the nitro or cyano alkene substrate during the nucleophilic attack by the 1,3-dicarbonyl compound. The ruthenium center acts as a Lewis acid to activate the electrophile while the basic nitrogen of the ligand assists in the deprotonation of the nucleophile, facilitating a concerted and highly selective bond formation.
Impurity control in this process is inherently managed by the high specificity of the catalyst-substrate interaction. Unlike non-selective base-catalyzed reactions that can lead to polymerization of nitro alkenes or racemic byproduct formation, the chiral ruthenium complex directs the reaction exclusively towards the desired enantiomer. The mechanism minimizes side reactions such as anionic polymerization, which is a common issue with nitro alkenes in the presence of simple bases. Furthermore, the stability of the ruthenium-hydride or amide complex ensures that the catalytic cycle remains intact over extended reaction times, preventing the release of free metal ions that could catalyze decomposition pathways. This robust mechanistic framework allows for the production of intermediates with optical purities exceeding 90% ee in many cases, drastically reducing the burden on downstream purification steps like chiral HPLC or recrystallization, which are often cost-prohibitive at scale.
How to Synthesize Optically Active Nitro Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the ease of handling and reproducibility of the method. The process begins with the in situ generation or pre-formation of the asymmetric ruthenium complex, followed by the addition of the nitro alkene and the dicarbonyl nucleophile in a standard organic solvent like toluene. Reaction monitoring is straightforward, typically requiring stirring for periods ranging from several hours to a few days depending on the specific substrate reactivity and temperature. The workup involves standard techniques such as flash column chromatography, avoiding complex quenching procedures associated with reactive organometallics. For detailed operational parameters and specific stoichiometric ratios optimized for different substrate classes, please refer to the standardized synthesis steps provided below.
- Prepare the asymmetric metal complex by reacting an optically active nitrogenous compound (chiral diamine) with a Periodic Table Group VIII metal complex (preferably Ruthenium) in the presence of a base.
- Mix the prepared asymmetric metal complex with a nitro or cyano alkene substrate (General Formula A) and a 1,3-dicarbonyl compound (General Formula B) in a suitable organic solvent such as toluene.
- Stir the reaction mixture at temperatures ranging from -30°C to 40°C for several hours to days, then purify the resulting optically active product using column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ruthenium-catalyzed process translates directly into enhanced operational resilience and economic efficiency. The elimination of cryogenic conditions represents a major infrastructure advantage, as it removes the dependency on specialized low-temperature reactors and the high energy costs associated with maintaining sub-zero environments. This shift allows for the utilization of standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, significantly lowering the barrier to entry for contract manufacturing organizations. Moreover, the avoidance of pyrophoric reagents like dialkyl zinc reduces safety risks and insurance premiums, while simplifying logistics and storage requirements for raw materials. The robustness of the catalyst system also implies longer campaign runs with less frequent catalyst replenishment, contributing to substantial cost savings in manufacturing overhead.
- Cost Reduction in Manufacturing: The process achieves high yields and selectivity with lower catalyst loadings compared to traditional magnesium or copper methods, directly reducing the cost of goods sold. By operating at mild temperatures, the facility saves significantly on energy consumption for cooling, and the simplified workup reduces solvent usage and waste disposal costs. The high optical purity achieved minimizes the need for expensive chiral resolution steps, further driving down the total production cost.
- Enhanced Supply Chain Reliability: The reagents required, such as dimethyl malonate and various nitro styrenes, are commercially available and stable, ensuring a consistent supply of raw materials without the volatility associated with unstable organometallics. The robust nature of the ruthenium catalyst allows for flexible scheduling and batch sizing, enabling suppliers to respond more quickly to fluctuating demand from pharmaceutical clients. This reliability is critical for maintaining continuous production lines for essential API intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of hazardous byproducts make this process highly scalable from kilogram to multi-ton production levels. The reduced use of toxic promoters and the ability to recycle solvents align with modern green chemistry principles, facilitating easier regulatory approval and environmental compliance. This scalability ensures that the supply chain can grow alongside the clinical and commercial needs of the drug development pipeline without requiring process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this asymmetric synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for technical teams evaluating this route. Understanding these nuances is essential for assessing the feasibility of integrating this method into existing production workflows.
Q: What are the advantages of the ruthenium-diamine catalyst over traditional rhodium or copper catalysts?
A: Unlike traditional rhodium or copper catalysts that often require unstable reagents like dialkyl zinc or extremely low temperatures (-78°C), the ruthenium-diamine complex described in CN1626501A operates efficiently at mild temperatures (-20°C to 30°C) with high enantioselectivity and does not require excessive catalyst loading.
Q: What types of substrates are compatible with this asymmetric Michael addition process?
A: The process demonstrates broad substrate scope, successfully reacting various nitro alkenes and cyano alkenes (including those with aromatic, heteroaromatic, and aliphatic substituents) with 1,3-dicarbonyl compounds like dimethyl malonate and methyl acetoacetate.
Q: Is this process scalable for commercial production of API intermediates?
A: Yes, the process utilizes stable catalysts and mild reaction conditions without the need for cryogenic equipment or highly pyrophoric reagents, making it highly suitable for commercial scale-up and consistent supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Nitro Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to meeting stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify enantiomeric excess and chemical purity for every batch. Our expertise in asymmetric catalysis allows us to optimize this ruthenium-mediated process for your specific target molecules, guaranteeing a supply of intermediates that meet the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline and reduce your overall manufacturing costs.
