Advanced Asymmetric Michael Addition for High-Purity Pharmaceutical Intermediates Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to synthesize chiral building blocks, particularly optically active nitro compounds and cyano compounds which serve as critical precursors for high-value APIs. Patent CN100354249C discloses a groundbreaking process for preparing these compounds represented by general formula (C) through an asymmetric Michael reaction. This technology utilizes a novel metal complex derived from the interaction of an optically active nitrogenous compound and a Periodic Table Group VIII metal complex, specifically focusing on Ruthenium-based systems. The significance of this invention lies in its ability to produce essential intermediates for drugs such as Baclofen (a GABA-B receptor agonist) and Endothelin A antagonists with superior efficiency and stereoselectivity. Unlike previous methods that struggled with stability and condition severity, this approach offers a robust solution for the commercial scale-up of complex pharmaceutical intermediates. By leveraging a chiral ruthenium catalyst, manufacturers can access high-purity products that were previously difficult to obtain economically.

The structural versatility of the Michael acceptor, as shown in the general formula (A) above, allows for a broad substrate scope including aromatic, heteroaromatic, and aliphatic substitutions. This flexibility is crucial for R&D teams aiming to diversify their pipeline of high-purity API intermediates without being constrained by narrow reaction tolerances. The patent explicitly details how varying the substituents R1, R2, and R3 enables the creation of diverse chiral centers, making this technology a cornerstone for modern asymmetric synthesis strategies in the production of specialty chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active nitro and cyano compounds has been plagued by significant technical hurdles that impact both cost and operational feasibility. Traditional methods often relied on rhodium complexes which necessitated the use of alkylboronic acids as nucleophilic reagents; these reagents are notoriously unstable and require pre-modulation or excessive addition, complicating the workflow. Furthermore, copper-based systems reported in non-patent literature frequently utilized unstable dialkyl zinc reagents that are hazardous in air and demanded cryogenic reaction temperatures ranging from -30°C to -78°C to achieve acceptable enantioselectivity. Magnesium complex approaches, while avoiding some unstable reagents, typically required high catalyst loadings of approximately 5 mol% along with additives like molecular sieves or amine promoters to drive the reaction, indicating a lack of intrinsic catalytic efficiency. These factors collectively result in high operational expenditures, complex safety protocols, and difficult cost reduction in pharmaceutical intermediates manufacturing due to the energy intensity and reagent instability associated with legacy processes.
The Novel Approach
The methodology described in CN100354249C represents a paradigm shift by employing a chiral ruthenium complex that functions effectively under much milder conditions, typically between -30°C and 40°C, and often at room temperature. This novel approach eliminates the dependency on unstable organometallic reagents like dialkyl zinc or alkylboronic acids, thereby simplifying the supply chain and reducing safety risks. The catalyst system demonstrates high turnover numbers with substrate-to-catalyst ratios (S/C) ranging from 50 to 2000, significantly lowering the amount of precious metal required per batch compared to the 5 mol% loading of older magnesium methods. By achieving high yields and excellent optical purity without the need for excessive promoters or molecular sieves, this process streamlines the downstream purification steps. For procurement managers, this translates to a more reliable sourcing strategy for fine chemical intermediates, as the raw materials are stable and the process conditions are easily manageable in standard reactor setups, facilitating a smoother transition from laboratory discovery to industrial production.
Mechanistic Insights into Ruthenium-Catalyzed Asymmetric Michael Addition
The core of this technological advancement lies in the unique structure and reactivity of the asymmetric metal complex, specifically the ruthenium-amide or ruthenium-hydride species formed in situ. The catalyst is generated by reacting a Group VIII metal precursor, such as [RuCl2(p-cymene)]2, with a chiral diamine ligand defined by general formula (D). This interaction creates a chiral environment around the metal center that is capable of simultaneously activating the Michael donor (the 1,3-dicarbonyl compound) and directing the approach of the Michael acceptor (the nitro or cyano alkene). The ruthenium center acts as a Lewis acid to activate the carbonyl oxygen, while the basic nitrogen of the ligand assists in the deprotonation of the active methylene group of the donor. This dual activation mechanism ensures that the nucleophilic attack occurs with high facial selectivity, leading to the formation of the desired enantiomer with minimal racemic byproduct formation.
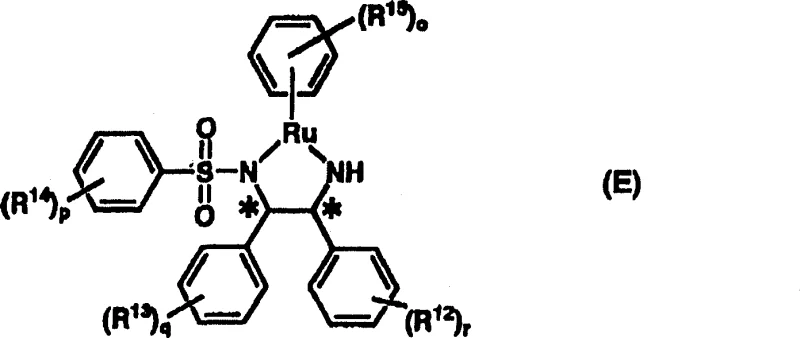
As illustrated in the catalyst structure (Formula E) above, the steric bulk provided by the sulfonyl groups on the diamine ligand plays a critical role in enforcing stereocontrol. The specific arrangement of the phenyl rings and the sulfonyl substituents creates a rigid chiral pocket that discriminates between the prochiral faces of the incoming alkene. This precise geometric control is what allows the reaction to proceed with enantiomeric excess values often exceeding 90%, and in optimized cases reaching up to 98% ee. Understanding this mechanistic nuance is vital for R&D directors, as it highlights the importance of ligand design in achieving reducing lead time for high-purity intermediates by minimizing the need for extensive chiral resolution steps post-reaction. The stability of this Ru-complex also contributes to a cleaner impurity profile, as the catalyst does not decompose readily into species that could contaminate the final API.
How to Synthesize Optically Active Nitro Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a production environment. The process begins with the preparation of the active catalyst species, followed by the addition of the substrates in a suitable solvent system such as toluene, THF, or alcohols. The reaction is typically conducted under an inert atmosphere to prevent oxidation of the sensitive metal center, although the robustness of the Ru-system offers some tolerance. Reaction times can vary from several minutes to 100 hours depending on the specific substrate combination and temperature, but optimization data suggests that high conversions are achievable within 24 to 48 hours at moderate temperatures. For detailed operational parameters and specific stoichiometric ratios tailored to your specific target molecule, please refer to the standardized synthesis guide below.
- Preparation of the Asymmetric Metal Complex: React a Periodic Table Group VIII metal precursor (preferably Ruthenium) with an optically active nitrogenous compound (chiral diamine) in the presence of a base to form the active amide or hydride catalyst species.
- Asymmetric Michael Reaction: Mix the prepared catalyst with a Michael acceptor (nitro or cyano alkene, Formula A) and a Michael donor (1,3-dicarbonyl compound, Formula B) in a suitable organic solvent such as toluene or THF.
- Reaction Control and Isolation: Maintain the reaction temperature between -30°C and 40°C for several hours to days, then isolate the high-purity product via standard purification techniques like column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this ruthenium-catalyzed process offers substantial strategic benefits for supply chain management and cost engineering within the pharmaceutical sector. The elimination of cryogenic requirements (-78°C) drastically reduces energy consumption and allows for the use of standard cooling equipment rather than specialized low-temperature reactors, which directly impacts capital expenditure and operational overhead. Furthermore, the use of stable, commercially available starting materials like trans-beta-nitrostyrene and dimethyl malonate ensures a consistent supply flow, mitigating the risks associated with sourcing unstable reagents like dialkyl zinc. This stability enhances the overall reliability of the manufacturing schedule, ensuring that delivery timelines for critical pharmaceutical intermediates are met without unexpected delays caused by reagent degradation or complex handling procedures.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by lowering the catalyst loading requirements; whereas older magnesium methods needed roughly 5 mol% of catalyst plus promoters, this Ru-system operates efficiently at S/C ratios of 50 to 2000. This reduction in precious metal usage, combined with the removal of expensive additives like molecular sieves and the avoidance of unstable boronic acids, leads to a leaner bill of materials. Additionally, the milder reaction conditions reduce utility costs associated with heating and cooling, contributing to a lower cost of goods sold (COGS) for the final chiral intermediate.
- Enhanced Supply Chain Reliability: By utilizing robust reagents that do not require special storage conditions or immediate preparation prior to use, the supply chain becomes more resilient. The ability to run reactions at temperatures as high as 30°C or as low as -20°C without compromising selectivity provides operational flexibility, allowing production to continue even if specific utility conditions fluctuate. This reliability is crucial for maintaining continuous manufacturing campaigns for high-demand APIs, ensuring that downstream formulation teams receive their materials on schedule without quality deviations.
- Scalability and Environmental Compliance: The simplified workup procedure, which often involves standard column chromatography or recrystallization without the need for complex quenches of reactive organometallics, facilitates easier scale-up from kilogram to tonne levels. The reduction in hazardous waste generation, particularly from avoiding pyrophoric reagents and excessive promoter use, aligns with modern green chemistry principles and environmental regulations. This makes the process not only economically attractive but also environmentally sustainable, reducing the burden on waste treatment facilities and improving the overall safety profile of the chemical plant.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this asymmetric synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the process suitability for your specific application. Understanding these nuances helps in making informed decisions about process adoption and technology transfer.
Q: What are the primary advantages of the Ruthenium-diamine catalyst over traditional Rhodium or Magnesium systems?
A: The Ruthenium-diamine system eliminates the need for unstable reagents like alkylboronic acids or dialkyl zinc, operates at significantly milder temperatures (-20°C to 30°C vs -78°C), and achieves high enantioselectivity with lower catalyst loading compared to conventional magnesium complexes which often require promoters and molecular sieves.
Q: What types of substrates are compatible with this asymmetric Michael addition process?
A: The process is highly versatile, accepting a wide range of Michael acceptors including trans-beta-nitrostyrene derivatives and cinnamyl nitriles with various substituents (halogens, alkoxy groups), and Michael donors such as malonates, acetoacetates, and cyclic 1,3-diketones.
Q: What level of optical purity can be achieved using this patented methodology?
A: Experimental data within the patent demonstrates that this method can achieve exceptional enantiomeric excess (ee), with specific examples reaching up to 98% ee for thiophene and furan derivatives, and consistently above 90% ee for many phenyl-substituted nitro compounds under optimized conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Nitro Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of optically active nitro or cyano compound meets the highest industry standards for enantiomeric excess and chemical purity. Our commitment to excellence makes us a trusted partner for global pharmaceutical companies seeking to secure their supply chains with reliable, high-performance ingredients.
We invite you to engage with our technical procurement team to discuss how this advanced ruthenium-catalyzed technology can be integrated into your production portfolio. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits specific to your target molecule. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver cost-effective and scalable solutions for your complex synthetic challenges.
