Scalable Asymmetric Hydrogenation Technology for High-Purity Chiral Intermediates
Scalable Asymmetric Hydrogenation Technology for High-Purity Chiral Intermediates
The landscape of chiral synthesis is undergoing a significant paradigm shift, driven by the urgent need for more sustainable and cost-effective manufacturing processes in the fine chemical and pharmaceutical sectors. Patent CN1530171A introduces a groundbreaking methodology for the asymmetric hydrogenation of carboxylic esters, specifically targeting the production of enantiomerically enriched alpha- and beta-hydroxycarboxylic esters. These compounds serve as critical building blocks in the synthesis of complex active pharmaceutical ingredients (APIs) and agrochemicals. Unlike traditional homogeneous catalytic systems that rely on expensive and air-sensitive chiral phosphines, this invention leverages a novel class of heterogeneous catalysts comprising transition metals adsorbed onto microporous or mesoporous carrier materials. This technological advancement addresses long-standing industry pain points regarding catalyst recovery, metal contamination, and process scalability, positioning it as a vital asset for any organization seeking a reliable pharmaceutical intermediate supplier capable of delivering high-value chiral synthons.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of enantiomerically enriched hydroxycarboxylic acid esters has relied heavily on homogeneous catalytic hydrogenation using transition metal complexes modified with chiral phosphine ligands. While effective in laboratory settings, these conventional methods suffer from severe drawbacks when translated to industrial manufacturing scales. The primary limitation lies in the inherent instability and high cost of chiral phosphines, which are often sensitive to oxidation and require stringent handling conditions, thereby inflating operational expenditures. Furthermore, the homogeneous nature of these catalysts necessitates complex and energy-intensive downstream processing steps to separate the precious metal residues from the product stream. This often involves multiple chromatographic purifications or the use of expensive metal scavengers, which not only increases waste generation but also poses significant risks of product loss and reduced overall yield. Additionally, alternative methods utilizing cinchona alkaloid-modified platinum or nickel catalysts have been documented, yet they frequently fail to deliver the high levels of enantiomeric excess required for modern regulatory standards, limiting their utility in high-value drug synthesis.
The Novel Approach
The innovative strategy outlined in the patent data circumvents these traditional bottlenecks by employing a heterogeneous catalytic system based on chiral nitrogen compounds coordinated with metals such as rhodium, iridium, or palladium. By immobilizing these active catalytic species onto solid support materials like MCM-41, Davison silica, or various zeolite types, the process achieves a unique combination of high activity and exceptional stereoselectivity. This approach fundamentally transforms the workflow by allowing the catalyst to be easily separated from the reaction mixture through simple filtration or centrifugation, effectively eliminating the need for complex metal removal protocols. The use of chiral diamines and related nitrogen ligands, which are generally more robust and accessible than their phosphine counterparts, further enhances the economic viability of the process. Moreover, the patent data demonstrates that these supported catalysts maintain high conversion rates and enantioselectivity even after multiple recycling runs, proving their durability and suitability for continuous manufacturing environments where supply chain reliability is paramount.
Mechanistic Insights into Supported Transition Metal Catalysis
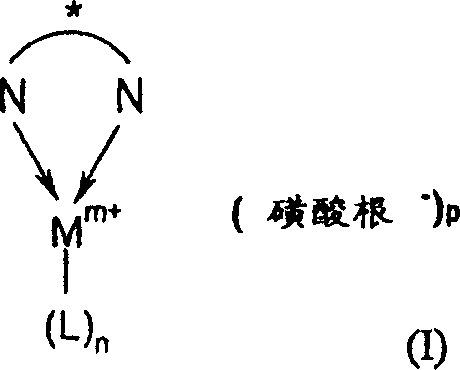
The core of this technological breakthrough lies in the precise architectural design of the catalyst, defined generally by Formula (I) in the patent documentation. This complex features a central transition metal ion, preferably in the +1 oxidation state for rhodium and iridium or +2 for palladium, coordinated with a chiral nitrogen ligand and stabilized by a sulfonate anion. The chiral nitrogen component, depicted in Formula (II), acts as the stereochemical director, creating a rigid chiral environment around the metal center that dictates the facial selectivity of the hydrogen addition to the prochiral ketone substrate. Common ligands include enantiomerically enriched diamines such as (1R,2R)-1,2-diphenylethylenediamine or pyrrolidine derivatives, which form stable chelates with the metal. The presence of the sulfonate anion, such as triflate or mesylate, is crucial as it serves as a weakly coordinating counter-ion that maintains the cationic character of the metal center, thereby enhancing its electrophilicity and catalytic turnover frequency without interfering with the substrate binding site.
Beyond the molecular composition, the physical support matrix plays a pivotal role in the catalyst's performance and longevity. The patent specifies the use of microporous, mesoporous, or macroporous carrier materials with pore sizes ranging from 15 to 250 Angstroms, with MCM-41 and specific silica gels being particularly preferred. This porous architecture ensures high surface area dispersion of the active sites, maximizing the contact between the substrate and the catalyst while preventing the aggregation of metal clusters that could lead to deactivation. The interaction between the metal complex and the support is typically achieved through adsorption, creating a robust heterogeneous system that retains the high enantioselectivity characteristic of homogeneous analogues while gaining the practical benefits of a solid catalyst. This dual optimization of electronic properties via ligand design and physical properties via support engineering results in a catalytic system capable of delivering enantiomeric excess values exceeding 90%, and often reaching up to 99%, which is critical for meeting the stringent purity specifications of the pharmaceutical industry.
How to Synthesize Enantiomerically Enriched Hydroxy Esters Efficiently
The synthetic protocol described in the patent offers a streamlined pathway for converting readily available ketocarboxylic acid esters into valuable chiral hydroxy esters. The process begins with the preparation of the supported catalyst, where a solution of the pre-formed metal-ligand complex is contacted with the dried porous support material in a suitable organic solvent such as dichloromethane or toluene. Following adsorption and drying, the resulting solid catalyst is charged into a hydrogenation vessel along with the substrate, typically an alpha- or beta-keto ester like methyl benzoylformate or ethyl chloroacetoacetate. The reaction is conducted under a hydrogen atmosphere at moderate temperatures ranging from 0 to 100 degrees Celsius and pressures between 0.1 to 200 bar, although optimal results are often observed at lower pressures around 4 to 30 bar. The use of protic solvents like methanol or ethanol is preferred to facilitate hydrogen transfer and substrate solubility. Upon completion, the reaction mixture is simply filtered to recover the catalyst for reuse, and the filtrate is concentrated to yield the high-purity product, drastically simplifying the isolation procedure compared to traditional methods.
- Preparation of the supported catalyst by adsorbing the chiral metal complex onto a porous carrier material such as MCM-41 or silica gel.
- Loading the ketocarboxylic acid ester substrate and the heterogeneous catalyst into a hydrogenation autoclave with a suitable solvent like methanol.
- Conducting the asymmetric hydrogenation under controlled hydrogen pressure and temperature, followed by simple filtration to recover the catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this heterogeneous asymmetric hydrogenation technology represents a strategic opportunity to optimize manufacturing costs and enhance operational resilience. The shift from homogeneous to heterogeneous catalysis directly addresses the high cost of goods sold associated with precious metal usage and waste disposal. By enabling the efficient recovery and recycling of the catalyst, the process significantly reduces the consumption of expensive rhodium or iridium metals, leading to substantial cost savings over the lifecycle of the product. Furthermore, the elimination of complex metal scavenging and purification steps shortens the overall production cycle time, allowing for faster turnaround and improved responsiveness to market demand fluctuations. This efficiency gain is compounded by the reduced generation of hazardous chemical waste, which lowers environmental compliance costs and aligns with increasingly strict global sustainability mandates.
- Cost Reduction in Manufacturing: The implementation of this supported catalyst system drives down manufacturing expenses primarily through the minimization of raw material waste and the extension of catalyst life. Unlike homogeneous systems where the catalyst is lost in the workup, this technology allows for the physical separation and potential reuse of the catalytic material, as evidenced by the recycling data in the patent examples. This durability translates to a lower cost per kilogram of the final chiral intermediate. Additionally, the simplified downstream processing removes the need for expensive chromatographic columns or specialized metal removal resins, further stripping away non-value-added costs from the production budget and improving the overall margin profile for high-volume commercial campaigns.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness and stability of the heterogeneous catalyst formulation. Solid-supported catalysts are generally less sensitive to air and moisture compared to their homogeneous phosphine-based counterparts, simplifying storage and transportation logistics and reducing the risk of spoilage during transit. This stability ensures that production schedules are not disrupted by catalyst degradation or the need for specialized inert atmosphere handling. Moreover, the use of widely available support materials like silica and standard transition metals mitigates the risk of supply bottlenecks associated with exotic or single-source ligands, providing a more secure and predictable sourcing strategy for critical raw materials essential for uninterrupted API manufacturing.
- Scalability and Environmental Compliance: The transition to a heterogeneous process inherently supports safer and more scalable operations, which is a key consideration for expanding production capacity. The ability to run reactions in standard autoclaves with simple filtration workups facilitates the scale-up from pilot plant to multi-ton commercial production without the engineering challenges posed by handling large volumes of pyrophoric or toxic homogeneous catalysts. From an environmental perspective, the reduction in solvent usage for purification and the decrease in heavy metal discharge into waste streams significantly lower the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only ensures compliance with current regulations but also future-proofs the supply chain against tightening environmental laws, making it a sustainable choice for long-term partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and claims within the patent specification, providing a factual basis for evaluating the process feasibility. Understanding these details is crucial for technical teams assessing the integration of this methodology into existing production lines or for procurement specialists evaluating supplier capabilities. The answers highlight the versatility of the catalyst system across different substrates and its proven track record in delivering high optical purity.
Q: What are the advantages of using supported catalysts over homogeneous systems for asymmetric hydrogenation?
A: Supported catalysts offer superior ease of separation via filtration, allowing for catalyst recycling and significantly reducing residual metal contamination in the final API intermediate, which simplifies downstream purification.
Q: Which metals and ligands are preferred for this hydrogenation process?
A: The patent specifies Rhodium (I), Iridium (I), and Palladium (II) complexes coordinated with chiral nitrogen ligands such as chiral diamines or pyrrolidine derivatives, stabilized by sulfonate anions.
Q: Can this process be scaled for industrial production of chiral intermediates?
A: Yes, the use of heterogeneous catalysts in autoclave systems facilitates commercial scale-up by improving handling safety, enabling continuous processing options, and ensuring consistent enantioselectivity across batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxycarboxylic Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like those described in CN1530171A for the production of high-value chiral intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this heterogeneous hydrogenation process are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of hydroxycarboxylic esters meets the exacting standards required by global pharmaceutical clients. We combine deep technical expertise in asymmetric synthesis with a commitment to quality assurance, making us the ideal partner for bringing complex chiral molecules from the lab bench to the marketplace.
We invite you to engage with our technical procurement team to discuss how this innovative catalytic route can be tailored to your specific project needs. By leveraging our capabilities, you can access a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this supported catalyst system for your target molecule. We encourage you to request specific COA data and route feasibility assessments to validate the performance metrics and ensure a seamless transition to a more cost-effective and sustainable supply chain solution. Let us collaborate to optimize your synthesis and secure a competitive advantage in the global market.
