Advanced Immobilized Catalysts for Scalable Production of Chiral Hydroxy Esters
Advanced Immobilized Catalysts for Scalable Production of Chiral Hydroxy Esters
The landscape of fine chemical manufacturing is undergoing a transformative shift driven by the urgent need for sustainable, cost-effective, and highly selective catalytic processes. Patent CN1530367A introduces a groundbreaking methodology for the preparation of enantiomerically enriched alpha- and beta-hydroxycarboxylic esters, which serve as pivotal building blocks in the synthesis of complex active pharmaceutical ingredients (APIs). This technology leverages novel immobilized transition metal complexes that overcome the historical limitations of traditional homogeneous catalysis, particularly the reliance on expensive and oxidation-sensitive chiral phosphine ligands. By anchoring chiral nitrogen compounds onto micro- or mesoporous carriers like MCM-41, this invention delivers a robust heterogeneous system capable of achieving high conversions and exceptional enantioselectivities under mild hydrogenation conditions. For global procurement and R&D teams, this represents a strategic opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-value chiral synthons with improved process economics and reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial production of enantiomerically enriched hydroxy esters has relied heavily on catalytic hydrogenation using transition metal complexes modified with chiral phosphines. While effective in laboratory settings, these homogeneous systems present severe drawbacks for large-scale manufacturing, primarily due to the exorbitant cost and extreme sensitivity to oxidation inherent in chiral phosphine ligands. Furthermore, the separation of the catalyst from the product stream in homogeneous processes is notoriously difficult, often requiring energy-intensive distillation or chromatography steps that degrade overall yield and purity. Alternative methods utilizing palladium or nickel catalysts modified by cinchona alkaloids have been explored, yet they frequently suffer from moderate enantiomeric excess, failing to meet the stringent stereochemical purity requirements demanded by modern regulatory bodies for drug substances. Consequently, the industry has long sought a catalytic solution that combines the high selectivity of homogeneous systems with the operational simplicity of heterogeneous catalysis without compromising on performance metrics.
The Novel Approach
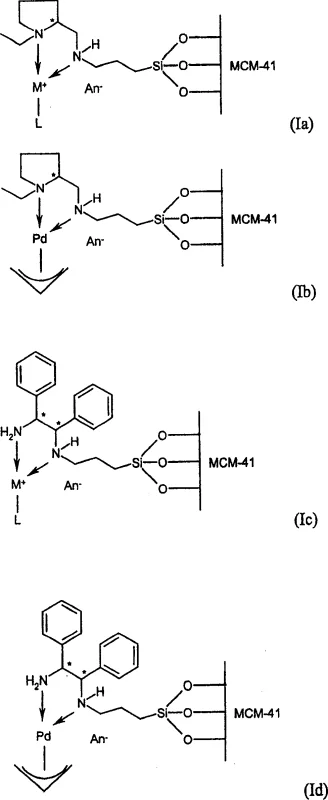
The innovative process disclosed in the patent data fundamentally reimagines catalyst design by covalently immobilizing enantiomerically enriched chiral nitrogen compounds onto solid support materials. Unlike previous attempts where heterogenization often led to a drastic drop in activity or selectivity, this specific architecture surprisingly enhances enantioselectivity. The method utilizes a linker group to bridge the chiral nitrogen ligand—such as (S)-2-aminomethyl-1-ethylpyrrolidine or (1R,2R)-1,2-diphenylethylenediamine—and a porous carrier like MCM-41. This creates a rigid, well-defined chiral environment around the active metal center, typically rhodium, iridium, or palladium. Comparative data within the patent reveals that while the homogeneous analogues of these specific complexes yielded virtually zero enantiomeric excess, the immobilized versions achieved values exceeding 90% ee. This paradigm shift allows manufacturers to utilize robust, filterable catalysts that maintain high performance, effectively solving the dual challenges of cost reduction in API manufacturing and simplified downstream processing.
Mechanistic Insights into Immobilized Transition Metal Catalysis
The core of this technological advancement lies in the precise molecular engineering of the catalyst surface. The process begins with the activation of the support material, where surface hydroxyl groups are reacted with chlorosilanes to create reactive halogenated sites. These sites then undergo nucleophilic substitution with the chiral amine, firmly locking the stereocenter onto the solid matrix. Once the chiral framework is established, a transition metal precursor, such as a rhodium cyclooctadiene complex, is introduced to coordinate with the nitrogen atoms. This coordination generates the active catalytic species directly on the pore surface. The mesoporous structure of carriers like MCM-41 is critical, as it ensures high surface area and accessibility for the substrate molecules while preventing the aggregation of metal centers. The resulting catalyst operates through a standard asymmetric hydrogenation cycle, where the prochiral ketone substrate coordinates to the metal, followed by the migratory insertion of hydride and subsequent reductive elimination to release the chiral alcohol product. The rigidity imposed by the support appears to restrict the conformational freedom of the ligand, favoring the transition state that leads to the desired enantiomer.
Furthermore, the immobilization strategy provides a unique mechanism for impurity control that is absent in homogeneous systems. In traditional liquid-phase catalysis, trace metal residues often persist in the final product, necessitating costly scavenging steps to meet ppm-level specifications required for pharmaceuticals. With this heterogeneous system, the metal is physically bound to the insoluble support, allowing for near-quantitative removal of the catalyst simply by filtration. This drastically reduces the risk of heavy metal contamination in the final hydroxycarboxylic ester product. Additionally, the covalent nature of the linkage prevents the leaching of the chiral ligand into the reaction medium, ensuring that the optical purity of the product remains consistent throughout the reaction lifecycle. This mechanistic stability is crucial for maintaining batch-to-batch consistency, a key metric for supply chain reliability in the production of high-purity OLED material or pharmaceutical intermediates where stereochemical integrity is non-negotiable.
How to Synthesize Immobilized Chiral Catalysts Efficiently
The synthesis of these advanced catalytic materials follows a streamlined, three-stage protocol designed for reproducibility and scalability. Initially, the support material is activated under controlled conditions to ensure optimal functional group density. Subsequently, the chiral ligand is coupled to the activated support in a solvent-mediated reaction, followed by thorough washing to remove unreacted amines. Finally, the metallation step introduces the active transition metal species. This modular approach allows for the fine-tuning of catalyst loading and properties by varying the linker length or the specific chiral diamine employed. The detailed standardized synthesis steps see the guide below for specific reaction parameters and purification techniques.
- Activate the porous support material, such as MCM-41, by reacting it with chlorosilanes to introduce functional halogen groups capable of covalent bonding.
- Couple the activated support with an enantiomerically enriched chiral nitrogen compound, such as (S)-2-aminomethyl-1-ethylpyrrolidine, to form the immobilized ligand framework.
- Complex the immobilized ligand with a transition metal precursor, such as a rhodium or palladium salt, to generate the final active heterogeneous catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this immobilized catalyst technology offers profound strategic advantages that extend beyond simple reaction efficiency. The primary value driver is the substantial cost savings achieved by eliminating the need for proprietary, high-cost chiral phosphine ligands, which are often subject to volatile pricing and supply constraints. By utilizing readily available chiral amines and robust support materials, the raw material cost base for the catalytic system is significantly lowered. Moreover, the heterogeneous nature of the catalyst simplifies the manufacturing workflow, removing the need for complex catalyst recovery units or extensive metal scavenging resins. This streamlining of the process flow directly translates to reduced operational expenditures and shorter cycle times, enhancing the overall agility of the supply chain in responding to market demands for critical intermediates.
- Cost Reduction in Manufacturing: The economic impact of switching to this immobilized system is driven by the complete avoidance of expensive noble metal-phosphine complexes that characterize traditional asymmetric hydrogenation. Since the catalyst is heterogeneous, it can theoretically be recovered and reused for multiple cycles, amortizing the initial capital cost of the metal and ligand over a much larger production volume. This reusability factor, combined with the elimination of downstream metal removal steps, results in a drastically simplified cost structure. The process avoids the use of sensitive reagents that require specialized handling or storage conditions, further reducing overhead costs associated with safety and inventory management in the production facility.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on single-source suppliers for specialized homogeneous catalysts. This technology mitigates that risk by utilizing commodity-grade support materials like silica gel or MCM-41 and widely available chiral amines, which are less susceptible to supply chain disruptions. The robustness of the immobilized catalyst also means it has a longer shelf life and is less prone to deactivation during transport or storage compared to air-sensitive homogeneous counterparts. This durability ensures that production schedules can be maintained without unexpected delays caused by catalyst degradation, providing a more predictable and reliable source of high-purity hydroxy esters for downstream synthesis.
- Scalability and Environmental Compliance: Scaling up asymmetric hydrogenation processes is historically challenging due to heat transfer issues and catalyst handling difficulties in large reactors. The solid nature of this immobilized catalyst facilitates easier handling in fixed-bed or slurry reactors, making the transition from pilot scale to commercial production significantly smoother. From an environmental perspective, the ability to filter and reuse the catalyst minimizes the generation of hazardous waste streams containing heavy metals. This aligns with increasingly stringent global environmental regulations regarding waste disposal and solvent usage. The process inherently supports green chemistry principles by reducing the E-factor of the synthesis, making it an attractive option for companies aiming to improve their sustainability profiles while scaling up complex polymer additives or pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this immobilized catalyst technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the operational boundaries and potential applications. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: Why does immobilization improve enantioselectivity in this specific process?
A: According to patent CN1530367A, the immobilized analogues demonstrated significantly higher enantiomeric excess compared to their homogeneous counterparts, which showed negligible selectivity in comparative examples. This suggests that the rigid environment provided by the mesoporous support stabilizes the active chiral conformation.
Q: What types of substrates are suitable for this catalytic system?
A: The technology is specifically optimized for the asymmetric hydrogenation of alpha- and beta-ketocarboxylic acid esters. Preferred substrates include methyl phenylglyoxylate and ethyl chloroacetoacetate, which are critical precursors for various pharmaceutical agents.
Q: Can the catalyst be reused for multiple batches?
A: Yes, the heterogeneous nature of the catalyst allows for easy separation via filtration or centrifugation. The covalent bonding of the chiral ligand to the support prevents leaching, enabling potential reuse and significantly reducing operational costs compared to single-use homogeneous systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxycarboxylic Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of chiral intermediates requires more than just a promising patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We understand the critical importance of stringent purity specifications in the pharmaceutical sector and operate rigorous QC labs equipped with advanced chiral HPLC and GC capabilities to verify enantiomeric excess and chemical purity at every stage of production. Our infrastructure is designed to handle the specific requirements of heterogeneous catalysis, including specialized filtration and drying units tailored for supported metal catalysts.
We invite you to collaborate with us to optimize your supply chain for chiral building blocks. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this immobilized catalyst route for your specific target molecules. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data rather than theoretical projections. Together, we can drive down costs and enhance the reliability of your critical intermediate supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
