Industrial Scale Synthesis of Cefpirome Sulfate via Optimized Acylation
Introduction to Advanced Cefpirome Sulfate Manufacturing
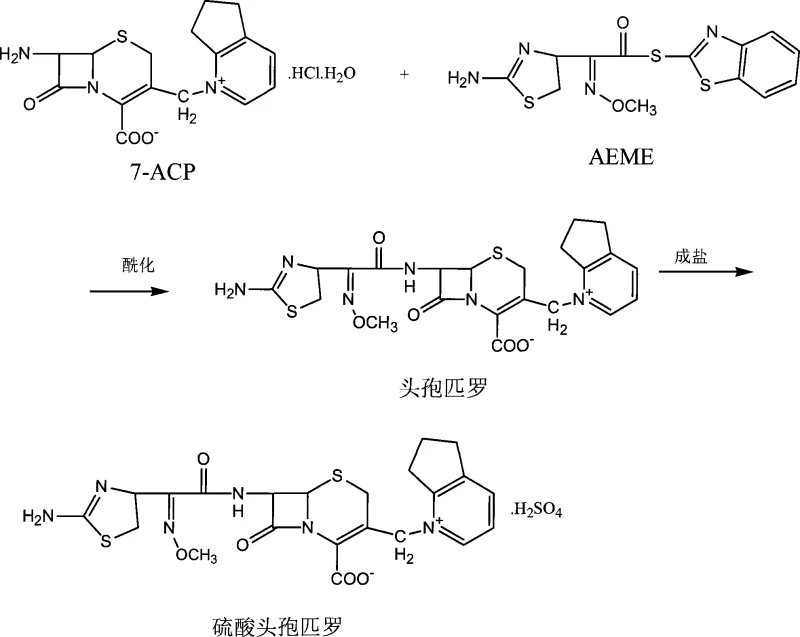
The global demand for fourth-generation cephalosporins continues to rise due to their broad-spectrum efficacy against resistant bacterial strains, necessitating robust and scalable manufacturing processes. Patent CN101066974A introduces a pivotal advancement in the synthesis of cefpirome sulfate, addressing critical bottlenecks found in traditional methodologies. This innovation leverages a direct acylation strategy between 7-amino-3-[(2,3-cyclopentenopyridine)-1-methyl] cephalosporanic acid hydrochloride (7-ACP) and the active ester AEME. By bypassing the cumbersome protection and deprotection sequences associated with earlier generations of synthesis, this method offers a streamlined pathway that is inherently more compatible with continuous industrial operations. For pharmaceutical manufacturers, this represents a significant opportunity to enhance production throughput while maintaining the rigorous purity profiles required for parenteral antibiotics.
The technical significance of this patent lies not only in the reaction itself but in the integrated purification protocol that follows. Traditional methods often struggle with the removal of colored impurities and residual solvents, which can compromise the stability and shelf-life of the final drug substance. The disclosed process incorporates a sophisticated work-up involving solvent extraction, activated carbon treatment, and a unique gamma-alumina column chromatography step. This multi-stage purification ensures that the resulting cefpirome sulfate possesses superior physicochemical properties, including consistent melting points and elemental composition, thereby reducing the risk of batch rejection during quality control testing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing cefpirome have largely relied on starting materials such as 7-amino-cephalosporanic acid (7-ACA) or GCLE, which introduce substantial inefficiencies into the production line. When utilizing 7-ACA, the synthetic route requires an initial silanization step to protect reactive functional groups before the side chain can be introduced at the C3 position. This protection-deprotection cycle not only extends the overall processing time but also necessitates the use of hazardous silylating agents and additional solvent volumes for washing and isolation. Furthermore, the reaction conditions for these older methods are often severe, requiring strict anhydrous environments and extreme temperature controls that increase energy consumption and equipment wear.
Another critical drawback of conventional routes is the difficulty in controlling impurity profiles during the final stages of synthesis. The byproducts generated from silanization and subsequent coupling reactions are often structurally similar to the target molecule, making them challenging to separate via standard crystallization techniques. This leads to lower overall yields and necessitates repeated recrystallization steps, which further erodes the mass balance and increases the environmental footprint of the manufacturing process. For supply chain managers, these complexities translate into longer lead times and higher vulnerability to raw material shortages, as the supply chain for specialized protected intermediates is often less resilient than that for bulk commodity chemicals.
The Novel Approach
The methodology outlined in the patent fundamentally restructures the synthesis by employing 7-ACP as the starting nucleus, which already contains the requisite cyclopentenopyridine moiety at the C3 position. This strategic choice eliminates the need for C3 modification entirely, collapsing what was previously a multi-step sequence into a single, highly efficient acylation reaction. The reaction is conducted in a biphasic system comprising water and polar aprotic solvents like DMF or DMAC, allowing for excellent solubility of the reactants while facilitating easy pH control. By maintaining the pH between 6.0 and 9.5, the process ensures optimal nucleophilic attack of the 7-amino group on the active ester without promoting hydrolysis of the sensitive beta-lactam ring.
Furthermore, the novel approach integrates a highly effective purification strategy that addresses the color and purity issues plaguing earlier methods. Instead of relying solely on crystallization, the process employs a combination of liquid-liquid extraction to remove the mercaptobenzothiazole byproduct, followed by a polishing step using gamma-alumina chromatography. This specific adsorption technique is capable of removing trace chromophores and polar impurities that activated carbon alone cannot capture. The result is a high-quality cefpirome solution that, upon salt formation with sulfuric acid and precipitation with acetone, yields a product with exceptional clarity and stability, suitable for direct formulation into injectable dosage forms.
Mechanistic Insights into Acylation and Purification Dynamics
The core chemical transformation in this process is the nucleophilic acyl substitution at the 7-position of the cephalosporin nucleus. The 7-ACP molecule acts as the nucleophile, where the lone pair electrons on the primary amine attack the carbonyl carbon of the AEME active ester. This reaction is facilitated by the presence of a base, such as triethylamine, which scavenges the proton released during the bond formation, driving the equilibrium towards the product. The Z-configuration of the methoxyimino group in the AEME side chain is critical, as it confers the necessary steric and electronic properties to resist beta-lactamase enzymes, a key feature of fourth-generation cephalosporins. Maintaining the reaction temperature between -10°C and 25°C is essential to preserve the stereochemical integrity of the double bond in the dihydrothiazine ring and prevent epimerization.
Following the acylation, the mechanistic focus shifts to the separation science employed to isolate the pure antibiotic. The extraction step utilizes immiscible organic solvents to selectively partition the lipophilic mercaptobenzothiazole byproduct away from the aqueous phase containing the zwitterionic cefpirome. Subsequent passage through the gamma-alumina column exploits the amphoteric nature of the alumina surface to adsorb polar impurities and colored degradation products. Unlike silica gel, gamma-alumina offers a specific surface chemistry that interacts favorably with the cephalosporin structure, allowing the target molecule to elute while retaining contaminants. This selective retention mechanism is vital for achieving the high purity levels required for regulatory approval, ensuring that the final sulfate salt is free from genotoxic impurities and heavy metals.
How to Synthesize Cefpirome Sulfate Efficiently
The synthesis of cefpirome sulfate via this patented route offers a clear roadmap for process chemists aiming to optimize production efficiency. The procedure begins with the precise preparation of the reaction medium, where the ratio of organic solvent to water is carefully tuned to maximize reactant solubility while minimizing side reactions. Operators must monitor the pH closely during the addition of the base to ensure it remains within the narrow window that favors acylation over hydrolysis. Once the reaction reaches completion, indicated by the depletion of the starting 7-ACP, the work-up phase commences immediately to prevent product degradation. The following guide outlines the standardized operational steps derived from the patent examples to ensure reproducible high-yield outcomes.
- Mix 7-ACP and AEME in an organic solvent/water mixture, adjusting pH to 6.0-9.5 to initiate acylation at controlled temperatures.
- Extract the reaction mixture to remove byproducts, followed by decolorization with activated carbon and purification through a gamma-alumina column.
- Add sulfuric acid solution to the purified cefpirome solution to form the salt, then induce crystallization using acetone to isolate the final solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method translates into tangible operational improvements and risk mitigation. The simplification of the synthetic route directly correlates with a reduction in the number of unit operations required, which in turn lowers the capital expenditure needed for reactor trains and purification equipment. By eliminating the need for specialized protected intermediates like silylated 7-ACA, manufacturers can source raw materials from a broader and more competitive supplier base, enhancing supply security. The ability to recycle solvents such as DMF and acetone further contributes to a leaner cost structure, reducing both raw material procurement costs and waste disposal fees associated with volatile organic compounds.
- Cost Reduction in Manufacturing: The elimination of protection and deprotection steps significantly reduces the consumption of expensive reagents and solvents, leading to a substantial decrease in the overall cost of goods sold. The high conversion efficiency of the acylation reaction minimizes the loss of valuable starting materials, ensuring that the theoretical yield is closely approached in practical settings. Additionally, the regeneration capability of the gamma-alumina stationary phase means that the cost of chromatographic media is amortized over many batches, providing long-term economic benefits that compound over the lifecycle of the product.
- Enhanced Supply Chain Reliability: Utilizing 7-ACP and AEME as starting materials leverages established supply chains for cephalosporin intermediates, reducing the risk of disruptions caused by niche reagent shortages. The robustness of the reaction conditions, which do not require cryogenic temperatures or ultra-high vacuum, allows for production in a wider range of manufacturing facilities, increasing geographic flexibility. This resilience is crucial for maintaining continuous supply to global markets, especially in the face of logistical challenges or regional regulatory changes that might impact the availability of more complex precursors.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard agitation and filtration equipment that can be easily scaled from pilot plant to commercial tonnage without significant re-engineering. The efficient recovery and recycling of solvents align with increasingly stringent environmental regulations, reducing the facility's carbon footprint and potential liability associated with hazardous waste emissions. This alignment with green chemistry principles not only ensures regulatory compliance but also enhances the corporate sustainability profile, which is becoming a key factor in vendor selection for major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cefpirome sulfate synthesis method. These answers are derived directly from the experimental data and process descriptions provided in the patent literature, offering clarity on critical process parameters and quality attributes. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for procurement professionals assessing the reliability of the supply source.
Q: What are the key advantages of using 7-ACP over 7-ACA for Cefpirome synthesis?
A: Using 7-ACP eliminates the need for complex silanization protection and deprotection steps required when starting from 7-ACA. This significantly shortens the synthetic route, reduces solvent consumption, and lowers the overall operational complexity, making it more suitable for large-scale industrial production.
Q: How does the gamma-alumina purification step improve product quality?
A: While activated carbon removes bulk impurities, passing the solution through a gamma-alumina column specifically targets trace colored impurities and residual solvents that affect the visual quality and stability of the final antibiotic. This step ensures the product meets stringent pharmacopeial standards for color and purity without requiring recrystallization.
Q: Is the solvent system used in this process environmentally sustainable?
A: Yes, the process utilizes a recoverable solvent system. The organic solvents such as DMF or DMAC and the anti-solvent acetone can be distilled and recycled efficiently. Additionally, the gamma-alumina stationary phase can be regenerated, minimizing solid waste generation and aligning with green chemistry principles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefpirome Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in patent examples are realized in full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against international pharmacopeial standards. Our commitment to quality assurance guarantees that the cefpirome sulfate we deliver meets the exacting requirements of your formulation teams.
We invite you to collaborate with us to leverage this advanced synthesis technology for your antibiotic portfolio. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your product quality and your bottom line.
