Optimizing Fourth-Generation Cephalosporin Production: A Technical Analysis of Cefpirome Sulfate Synthesis
Optimizing Fourth-Generation Cephalosporin Production: A Technical Analysis of Cefpirome Sulfate Synthesis
The pharmaceutical landscape for broad-spectrum antibiotics continues to evolve, driven by the urgent need for effective treatments against resistant bacterial strains. Patent CN101284840A introduces a refined synthetic methodology for Cefpirome Sulfate, a potent fourth-generation cephalosporin known for its zwitterionic structure and superior penetration of bacterial outer membranes. This technical disclosure marks a significant departure from traditional routes that rely on costly iodine-based silylating agents, proposing instead the use of trimethylbromosilane (TMSBr) in conjunction with readily available Cefotaxime. For R&D directors and procurement strategists, this shift represents a pivotal opportunity to enhance process efficiency while mitigating raw material volatility. The protocol outlines a robust pathway that not only simplifies the operational complexity but also ensures the high purity standards required for parenteral administration, addressing both the economic and regulatory challenges inherent in modern antibiotic manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cefpirome has been hindered by the reliance on Iodotrimethylsilane (TMSI) as the primary protecting agent for the carboxyl group during the quaternization step. While TMSI is highly reactive, its commercial application is severely constrained by its exorbitant cost and the complexities associated with handling iodine byproducts. Furthermore, alternative routes attempting to bypass this step often involve harsh reaction conditions or multi-step sequences that introduce 7-amino-cephalosporanic acid (7-ACA) derivatives late in the process, leading to lower overall yields and difficult purification profiles. These conventional methods frequently struggle with the removal of residual heavy metals or halogenated impurities, necessitating extensive downstream processing that erodes profit margins and extends production lead times. The economic burden of these inefficiencies is compounded by the fluctuating global supply of specialized iodine reagents, creating a fragile supply chain vulnerable to geopolitical and market disruptions.
The Novel Approach
The innovative strategy detailed in the patent data circumvents these bottlenecks by utilizing Trimethylbromosilane (TMSBr) as a highly effective yet cost-efficient alternative to TMSI. This approach capitalizes on the superior nucleophilicity of the bromide ion compared to chloride, while avoiding the premium pricing of iodine reagents. By initiating the reaction with Cefotaxime, a commercially mature intermediate with stable supply dynamics, the process achieves a molar conversion efficiency that significantly outperforms legacy methods. The substitution reaction is conducted in an optimized solvent system, typically anhydrous tetrahydrofuran (THF), which enhances the solubility of the reactants and minimizes side reactions such as beta-lactam ring opening. This methodological pivot not only reduces the direct cost of goods sold (COGS) but also streamlines the workflow, allowing for a more direct transition from the dihydrobromide intermediate to the final sulfate salt with minimal loss of potency or stereochemical integrity.
Mechanistic Insights into Silylation-Protection and Quaternization
The core chemical transformation in this synthesis involves the in situ silylation of the Cefotaxime carboxyl group, followed by a nucleophilic substitution at the C-3' position of the cephem nucleus. When Trimethylbromosilane is introduced to the reaction mixture containing Cefotaxime, it rapidly forms a silyl ester, effectively masking the acidic carboxyl group and increasing the lipophilicity of the molecule. This activation is crucial for facilitating the subsequent attack by 2,3-cyclopentapyridine, a bulky heterocyclic amine that serves as the quaternizing agent. The reaction proceeds through a concerted mechanism where the silyl group stabilizes the leaving acetate moiety, allowing the pyridine nitrogen to displace it and form the stable quaternary ammonium salt characteristic of fourth-generation cephalosporins. The use of TMSBr ensures that the silylation equilibrium favors the protected species without requiring excessive temperatures that could degrade the sensitive beta-lactam ring, thereby preserving the biological activity of the final API.
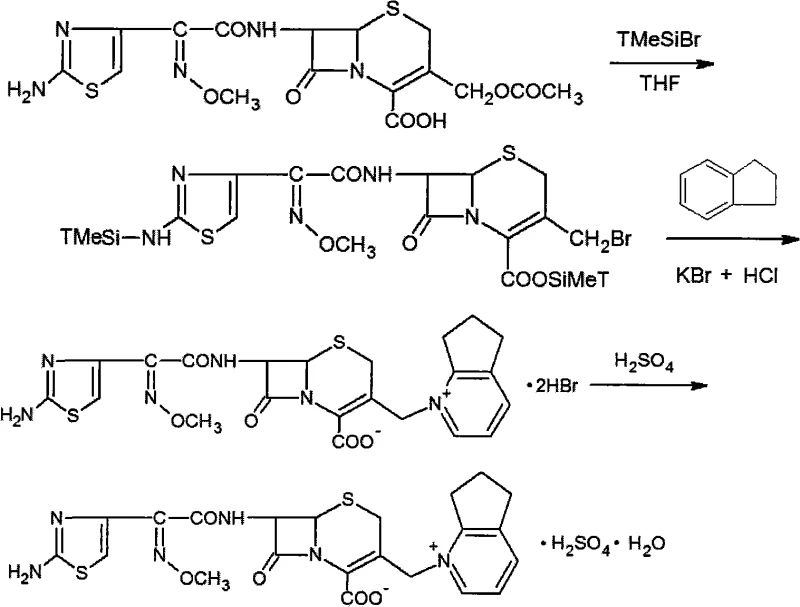
Following the formation of the quaternary intermediate, the process employs a sophisticated workup procedure to isolate the Cefpirome Dihydrobromide with high fidelity. The addition of potassium bromide and hydrochloric acid serves to protonate any unreacted amines and precipitate the product as a stable hydrobromide salt, which is less hygroscopic and easier to handle than the free base. Crucially, the patent emphasizes the importance of temperature control during this phase, maintaining the system near 0°C to prevent hydrolysis of the beta-lactam ring. The subsequent purification steps, including activated carbon treatment and gamma-alumina chromatography, are designed to remove trace organic impurities and potential pyrogens. This rigorous attention to impurity profiling ensures that the final Cefpirome Sulfate meets the stringent pharmacopoeial requirements for injectable antibiotics, demonstrating a deep understanding of the relationship between process parameters and product safety.
How to Synthesize Cefpirome Sulfate Efficiently
Executing this synthesis requires precise adherence to the stoichiometric ratios and environmental controls outlined in the technical data to maximize yield and purity. The process begins with the preparation of an anhydrous reaction environment, where Cefotaxime and 2,3-cyclopentapyridine are mixed in a molar ratio ranging from 1:7 to 1:12, with a preferred optimization at 1:8.5 to drive the reaction to completion. The detailed standardized synthesis steps involve careful addition of reagents under inert atmosphere, followed by controlled crystallization and ion-exchange purification. Operators must ensure that the solvent system remains free of moisture to prevent premature desilylation, which could lead to polymerization or degradation of the cephem core. The following guide summarizes the critical operational phases derived from the patent embodiments.
- Prepare the reaction solvent by mixing anhydrous THF with trimethylbromosilane under nitrogen protection at low temperature.
- Add 2,3-cyclopentapyridine and Cefotaxime to the mixture, reflux, then cool and treat with KBr/HCl to precipitate Cefpirome Dihydrobromide.
- Dissolve the dihydrobromide intermediate, purify via ion-exchange resin and chromatography, then crystallize with sulfuric acid to obtain Cefpirome Sulfate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond simple unit cost reductions. By shifting the raw material base to Cefotaxime and Trimethylbromosilane, manufacturers can leverage the extensive existing production capacity for third-generation cephalosporins, ensuring a stable and predictable supply of starting materials. This reduces the dependency on niche reagents that are often subject to volatile pricing and long lead times. Furthermore, the simplified purification protocol, which utilizes standard ion-exchange resins and common chromatographic media rather than exotic catalysts, lowers the barrier to entry for scale-up and reduces the capital expenditure required for specialized equipment. The overall process design prioritizes operational simplicity, which translates directly into reduced labor costs and shorter batch cycle times, enhancing the agility of the manufacturing facility to respond to market demand fluctuations.
- Cost Reduction in Manufacturing: The substitution of Iodotrimethylsilane with Trimethylbromosilane represents a significant downward pressure on raw material expenses without compromising reaction kinetics. Since TMSBr is chemically more active than chlorosilanes yet substantially cheaper than iodosilanes, the process achieves an optimal balance between reagent cost and performance. Additionally, the high yield reported in the patent embodiments, reaching up to 84% for the intermediate and 80% for the final sulfate salt, minimizes waste generation and maximizes the throughput of valuable API per batch. This efficiency gain eliminates the need for expensive recycling loops or complex recovery systems for precious metal catalysts, further driving down the operational expenditure associated with antibiotic production.
- Enhanced Supply Chain Reliability: Sourcing Cefotaxime as the primary precursor aligns the supply chain with one of the most widely manufactured beta-lactam intermediates globally, particularly within the Asian market where production capacity is robust. This abundance ensures that manufacturers are not held hostage by single-source suppliers or limited production slots for specialized starting materials. The use of common organic solvents like THF and ethanol, along with standard inorganic salts like potassium bromide, further de-risks the supply chain by utilizing commodities that are readily available from multiple vendors. This diversification of the supply base creates a resilient procurement strategy that can withstand regional disruptions or logistical bottlenecks, ensuring continuous production schedules.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, moving seamlessly from laboratory gram-scale experiments to multi-ton commercial production without requiring fundamental changes to the reaction engineering. The avoidance of heavy metal catalysts simplifies the environmental compliance profile, as there is no need for rigorous testing and removal of trace metals like palladium or platinum from the final drug substance. The waste streams generated are primarily organic solvents and aqueous salt solutions, which can be managed through standard distillation and wastewater treatment protocols. This alignment with green chemistry principles not only reduces the environmental footprint but also facilitates faster regulatory approvals in markets with strict ecological guidelines, accelerating time-to-market for generic or biosimilar versions of the drug.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Cefpirome Sulfate synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the practical implications for industrial adoption. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the competitive positioning of the resulting product in the global marketplace.
Q: Why is Trimethylbromosilane preferred over Iodotrimethylsilane in this synthesis?
A: Trimethylbromosilane offers a superior balance of reactivity and cost. While Iodotrimethylsilane is highly reactive, it is significantly more expensive. Trimethylbromosilane is more active than Trimethylchlorosilane, ensuring efficient silylation protection of the carboxyl group without the prohibitive costs associated with iodine-based reagents, thereby optimizing the overall production economics.
Q: What represents the critical quality attribute in the final Cefpirome Sulfate product?
A: The critical quality attribute is the high chemical purity, specifically achieving levels above 99.6% as demonstrated in the patent examples. This is accomplished through a rigorous purification sequence involving activated carbon decolorization, gamma-alumina chromatography, and controlled crystallization, which effectively removes pyrogens, intracellular toxins, and colored impurities common in beta-lactam synthesis.
Q: How does the use of Cefotaxime as a starting material impact supply chain stability?
A: Utilizing Cefotaxime as the primary raw material leverages an established and mature supply chain, particularly within the Chinese market where capacity is abundant. Unlike specialized precursors that may face bottlenecks, Cefotaxime is a widely produced third-generation cephalosporin, ensuring consistent availability and reducing the risk of raw material shortages for the manufacturing of fourth-generation derivatives like Cefpirome.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefpirome Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent concept to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of silylation and quaternization chemistry is maintained at every scale. We operate stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art HPLC and spectroscopic instrumentation to verify that every batch of Cefpirome Sulfate meets the highest international standards. Our commitment to quality assurance means that we do not just supply chemicals; we deliver validated solutions that integrate seamlessly into your downstream formulation processes.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this TMSBr-mediated process. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence and our proven track record in the cephalosporin sector.
