Scalable Biocatalytic Synthesis of Chiral 3-Heteroaryl-3-Hydroxypropionic Acid Derivatives for Pharma
Scalable Biocatalytic Synthesis of Chiral 3-Heteroaryl-3-Hydroxypropionic Acid Derivatives for Pharma
The pharmaceutical industry's relentless pursuit of enantiomerically pure active pharmaceutical ingredients (APIs) has driven significant innovation in asymmetric synthesis, particularly for intermediates used in treating neurological disorders. Patent CN1497048A introduces a groundbreaking biocatalytic process for preparing enantiomer-enriched 3-heteroaryl-3-hydroxypropionic acid derivatives, which serve as critical precursors for serotonin and norepinephrine uptake inhibitors. Unlike traditional chemical resolutions that inherently cap yields at 50%, this microbial reduction strategy leverages the stereoselective power of yeasts and fungi to deliver high-value chiral building blocks with exceptional purity. For R&D directors and procurement specialists, this technology represents a paradigm shift towards more sustainable and economically viable manufacturing routes for complex heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 3-heteroaryl-1-aminopropan-3-ols and their hydroxy-acid precursors relied heavily on chemical resolution of racemates or the use of stoichiometric chiral auxiliaries. A notable prior art method described in Chirality 2000 involves the enzymatic resolution of racemic 3-chloro-1-(2-thienyl)-1-propanol, a process fundamentally flawed by its maximum theoretical yield of only 50% for the desired enantiomer. Furthermore, conventional chemical reductions often require expensive transition metal catalysts, stringent anhydrous conditions, and generate significant heavy metal waste, complicating downstream purification and environmental compliance. These factors collectively inflate the cost of goods sold (COGS) and introduce supply chain vulnerabilities associated with the sourcing of precious metal catalysts and specialized reagents.
The Novel Approach
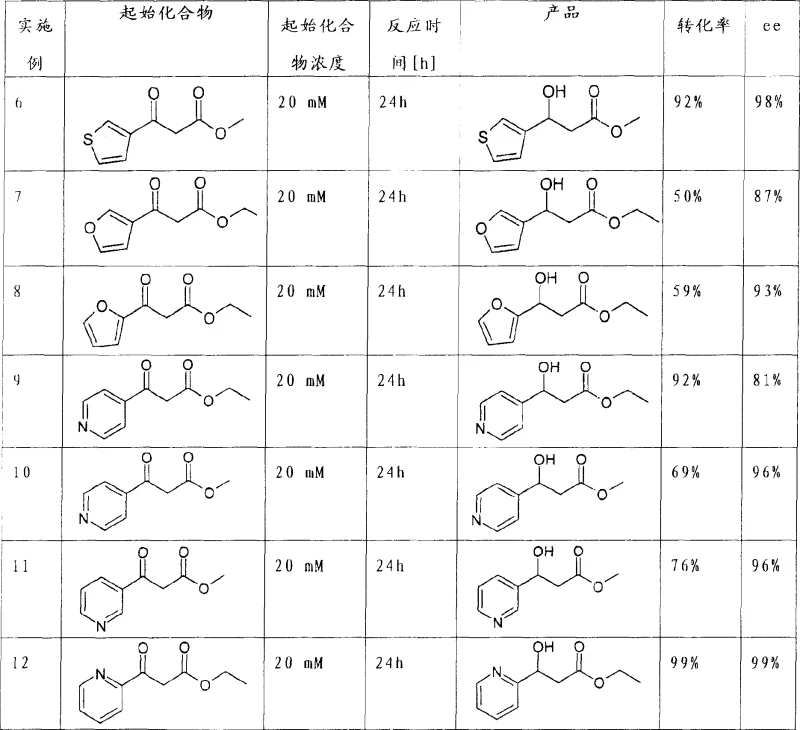
The novel approach disclosed in the patent utilizes whole-cell biocatalysis, specifically employing strains such as Saccharomyces cerevisiae and Geotrichum candidum, to effect the enantioselective reduction of 3-oxocarboxylic acid derivatives. This method operates under mild aqueous conditions, typically at pH 6 to 8 and temperatures around 25°C to 30°C, eliminating the need for hazardous organic solvents and extreme pressure. By bypassing the resolution step entirely, the process theoretically allows for 100% conversion of the starting ketone into the desired (S)-alcohol, effectively doubling the material efficiency compared to resolution strategies. The robustness of this biological system is evidenced by its tolerance to various heteroaryl substituents, including thiophene, furan, and pyridine rings, making it a versatile platform technology for diverse medicinal chemistry programs.
Mechanistic Insights into Microbial Ketoreduction
The core mechanism driving this transformation involves intracellular ketoreductases (KREDs) present within the yeast or fungal cells, which utilize cofactors like NADPH to transfer a hydride ion to the prochiral ketone carbonyl. The enzyme's active site imposes strict steric constraints, ensuring that hydride delivery occurs exclusively from one face of the planar carbonyl group, thereby establishing the (S)-configuration at the newly formed stereocenter. As demonstrated in the experimental data, substrates such as methyl 3-oxo-3-(2-thienyl)propionate are reduced with remarkable fidelity, achieving enantiomeric excess (ee) values greater than 97%. This high stereocontrol is maintained even when the electronic nature of the heteroaryl ring is varied, as seen with electron-rich furans and electron-deficient pyridines, indicating a broad and adaptable catalytic pocket within the microorganism.
Impurity control in this biocatalytic system is inherently superior to chemical methods due to the high chemoselectivity of the enzymes. Unlike chemical reducing agents like sodium borohydride or lithium aluminum hydride, which might reduce other sensitive functional groups such as nitriles or esters indiscriminately, the microbial system targets the ketone moiety with precision. For instance, the patent details the successful reduction of 3-oxo-3-(2-thienyl)propionitrile to the corresponding hydroxy-nitrile without affecting the cyano group, a transformation that is challenging to achieve chemoselectively using standard hydride reagents. This selectivity minimizes the formation of side products, simplifying the workup procedure to a straightforward extraction and significantly reducing the burden on analytical quality control teams.
How to Synthesize 3-Heteroaryl-3-Hydroxypropionic Acid Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this biocatalytic route at both laboratory and pilot scales. The process begins with the cultivation of the selected microbial strain in a nutrient-rich medium, followed by harvesting the biomass and resuspending it in a buffered solution supplemented with a co-substrate like glucose to regenerate the necessary cofactors. The starting ketone is then introduced to the cell suspension, where the biotransformation proceeds over a period of 10 to 48 hours. Detailed standardized synthetic steps for optimizing cell density, pH control, and substrate feeding strategies are provided in the guide below to ensure reproducible high-yield outcomes.
- Cultivate microorganisms such as Saccharomyces cerevisiae or Geotrichum candidum in YM or GC medium until an optical density (OD600) of 5 to 300 is reached.
- Harvest cells via centrifugation and resuspend in a buffer solution (e.g., potassium phosphate pH 6-8) containing a co-substrate like glucose.
- Add the 3-heteroaryl-3-oxopropionate substrate (20-50 mM) to the cell suspension and incubate at 25-30°C for 10 to 48 hours with shaking.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic technology offers profound strategic advantages beyond mere technical feasibility. The elimination of stoichiometric chiral resolving agents and precious metal catalysts translates directly into a simplified raw material portfolio, reducing dependency on volatile commodity markets for specialized chemicals. Furthermore, the use of water as the primary reaction medium drastically cuts solvent procurement costs and mitigates the environmental liabilities associated with volatile organic compound (VOC) emissions. This alignment with green chemistry principles not only enhances the corporate sustainability profile but also future-proofs the supply chain against increasingly stringent environmental regulations regarding waste disposal and carbon footprint.
- Cost Reduction in Manufacturing: The transition from a 50% yield resolution process to a dynamic kinetic resolution or direct asymmetric reduction effectively doubles the output per unit of starting material, leading to substantial cost savings in raw material consumption. Additionally, the removal of heavy metal catalysts eliminates the need for expensive scavenging resins and rigorous metal testing, streamlining the purification train and reducing overall processing time. The operational simplicity of running reactions at ambient temperature and pressure further lowers energy consumption compared to high-pressure hydrogenation or cryogenic chemical reductions.
- Enhanced Supply Chain Reliability: The starting materials for this process, such as 3-oxo-3-(2-thienyl)propionic acid esters, are readily accessible via established chemical synthesis routes involving inexpensive reagents like dimethyl carbonate and acetylthiophene. The reliance on robust, commercially available yeast strains ensures that the biocatalyst itself is not a single point of failure; multiple vendors can supply the necessary microbial biomass, preventing supply bottlenecks. This redundancy in the supply base provides procurement teams with greater negotiating leverage and security of supply for long-term API production campaigns.
- Scalability and Environmental Compliance: Biocatalytic processes are inherently scalable, moving seamlessly from shake flasks to multi-liter fermenters as demonstrated in the patent examples using 10-liter setups. The aqueous nature of the waste stream simplifies effluent treatment, as it lacks the toxic halogenated solvents often found in traditional organic synthesis. This ease of waste management reduces the operational overhead for environmental health and safety (EHS) departments and facilitates faster regulatory approval for new drug filings by presenting a cleaner, more controlled manufacturing process to health authorities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic route. These answers are derived directly from the experimental data and claims within patent CN1497048A, providing a factual basis for evaluating the technology's fit for your specific project requirements. Understanding these nuances is crucial for making informed decisions about process development and vendor selection.
Q: What is the enantiomeric excess (ee) achieved in this biocatalytic process?
A: The process described in patent CN1497048A consistently achieves high enantiomeric excess, typically exceeding 97% ee for the (S)-enantiomer when using strains like Saccharomyces cerevisiae NG 247 or Y278.
Q: Can this method be applied to heterocycles other than thiophene?
A: Yes, the method demonstrates broad substrate scope, successfully reducing 3-oxo-derivatives containing furan, pyridine, and pyrrole rings with high conversion rates and stereoselectivity.
Q: What are the typical reaction conditions for the microbial reduction?
A: The reaction is typically conducted in an aqueous buffer at a pH between 6 and 8, at temperatures ranging from 25°C to 30°C, using substrate concentrations between 20 mM and 50 mM.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Heteroaryl-3-Hydroxypropionic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity chiral intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral chromatography capabilities to guarantee that every batch of 3-heteroaryl-3-hydroxypropionic acid derivatives meets the exacting standards required for global pharmaceutical registration.
We invite you to engage with our technical procurement team to discuss how this biocatalytic technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this greener, more efficient route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecule, ensuring a reliable and cost-effective supply of these vital pharmaceutical intermediates.
