Advanced Biocatalytic Synthesis for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
Advanced Biocatalytic Synthesis for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
The pharmaceutical industry constantly seeks more efficient pathways to produce chiral intermediates, and the technology disclosed in patent CN100510093C represents a significant leap forward in this domain. This patent details a robust method for preparing enantiomerically enriched 3-heteroaryl-3-hydroxypropionic acid derivatives and 3-heteroaryl-1-aminopropan-3-ols, which are critical precursors for serotonin and norepinephrine reuptake inhibitors. Unlike traditional chemical synthesis routes that often struggle with stereoselectivity and environmental impact, this biocatalytic approach leverages specific microorganisms to achieve high enantiomeric excess under mild conditions. For R&D directors and procurement specialists, understanding the nuances of this technology is essential for evaluating potential supply chain partners who can deliver high-purity pharmaceutical intermediates with consistent quality. The ability to produce these complex molecules without the need for expensive chiral catalysts or resolution steps marks a pivotal shift towards more sustainable and cost-effective manufacturing processes in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral 3-heteroaryl-3-hydroxypropionic acid derivatives relied heavily on the resolution of racemic mixtures, a process inherently limited by a maximum theoretical yield of 50%. As noted in prior art such as Chirality 2000, enzymatic resolution of racemates often results in the discard of the unwanted enantiomer, leading to significant material waste and increased raw material costs. Furthermore, chemical reduction methods using stoichiometric chiral reagents can be prohibitively expensive and generate substantial amounts of hazardous waste, complicating environmental compliance and disposal protocols. The reliance on transition metal catalysts in some conventional routes also introduces the risk of heavy metal contamination, necessitating additional purification steps that extend lead times and reduce overall process efficiency. These limitations create bottlenecks in the supply chain, making it difficult for manufacturers to scale production without incurring exponential cost increases. Consequently, the industry has long required a method that bypasses these inefficiencies to ensure a reliable supply of high-purity pharmaceutical intermediates.
The Novel Approach
The novel approach described in the patent utilizes a biocatalytic reduction strategy that overcomes the yield barriers of traditional resolution methods by directly synthesizing the desired enantiomer with high selectivity. By employing specific strains of yeast or fungi, such as Saccharomyces cerevisiae or Geotrichum candidum, the process achieves enantiomeric excess values often exceeding 90%, effectively doubling the potential yield compared to resolution techniques. This method operates under mild aqueous conditions, typically at temperatures between 20°C and 40°C, which significantly reduces energy consumption and enhances safety profiles in the manufacturing facility. The versatility of this biocatalytic system allows for the reduction of various heteroaryl ketones, including thiophene, furan, and pyridine derivatives, providing a flexible platform for synthesizing a wide range of analogues. This adaptability is crucial for cost reduction in pharmaceutical intermediates manufacturing, as it allows producers to utilize a single core technology for multiple product lines. The elimination of harsh chemical reagents and the use of renewable biocatalysts align with modern green chemistry principles, offering substantial long-term advantages for supply chain sustainability.
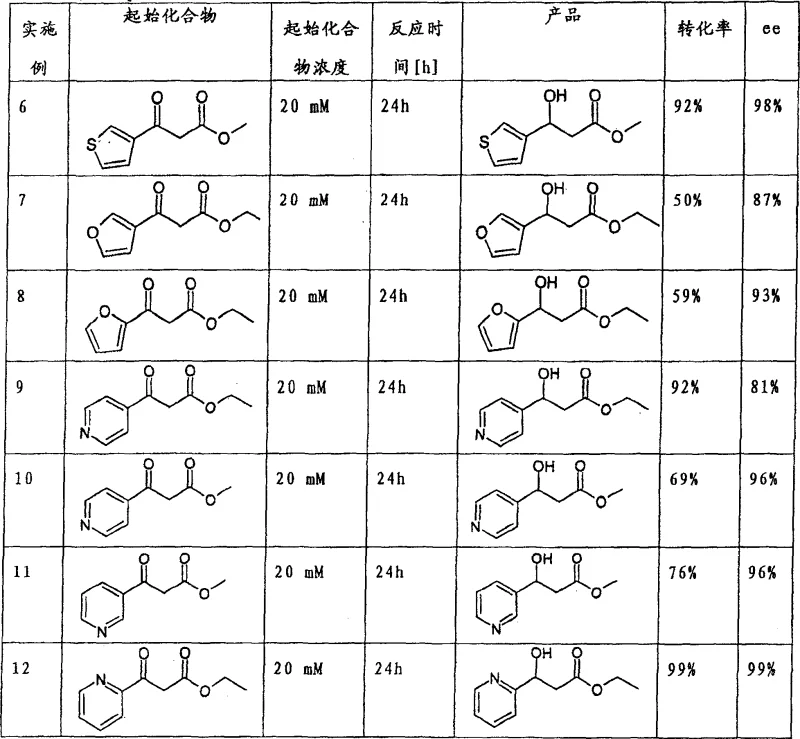
Mechanistic Insights into Biocatalytic Ketone Reduction
The core mechanism of this transformation involves the stereoselective reduction of the ketone carbonyl group in the 3-oxo-carboxylic acid derivative to a hydroxyl group, mediated by oxidoreductase enzymes within the microbial cells. These enzymes, often dependent on cofactors like NADPH, transfer a hydride ion to the carbonyl carbon with precise spatial orientation, ensuring the formation of the (S)-configured alcohol. The microbial environment provides a continuous regeneration system for these cofactors, typically through the metabolism of added co-substrates like glucose, which maintains the catalytic cycle without the need for external cofactor addition. This self-sustaining enzymatic activity is a key factor in the process's economic viability, as it minimizes the consumption of expensive reagents. The specificity of the microbial strain dictates the stereochemical outcome, with certain strains showing a strong preference for the (S)-enantiomer, which is often the pharmacologically active form required for downstream drug synthesis. Understanding this mechanistic detail allows R&D teams to optimize reaction conditions, such as pH and substrate concentration, to maximize both yield and enantiomeric purity.
Impurity control is another critical aspect of this biocatalytic mechanism, as the mild conditions prevent many of the side reactions common in chemical synthesis, such as over-reduction or racemization. The aqueous nature of the reaction medium facilitates the separation of organic products through extraction, while the biological system tends to be highly chemoselective, leaving other functional groups on the heteroaryl ring intact. This high level of chemoselectivity reduces the burden on downstream purification processes, resulting in a cleaner crude product that requires fewer crystallization or distillation steps. For supply chain heads, this translates to reduced processing time and lower solvent consumption, directly impacting the cost of goods sold. The ability to consistently produce material with high optical purity ensures that downstream synthesis steps are not compromised by chiral impurities, which could otherwise lead to failed batches in the final API production. This reliability is paramount for maintaining the integrity of the pharmaceutical supply chain and ensuring regulatory compliance.
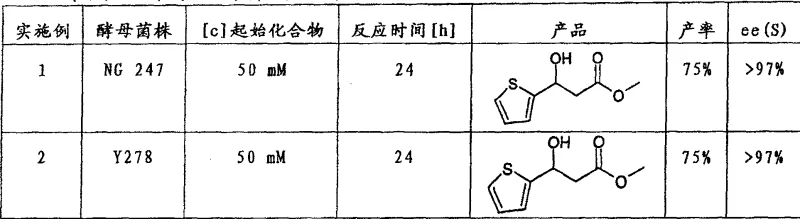
How to Synthesize 3-Heteroaryl-3-Hydroxypropionic Acid Derivatives Efficiently
Implementing this synthesis route requires careful attention to microbial cultivation and reaction parameter control to ensure optimal performance and reproducibility on a commercial scale. The process begins with the growth of the selected microbial strain in a nutrient-rich medium, where parameters such as optical density, temperature, and pH are closely monitored to achieve the desired biocatalytic activity. Once the biomass is prepared, the substrate is introduced under controlled conditions to initiate the reduction, with reaction progress tracked via chromatographic methods to determine endpoint and conversion. Detailed standard operating procedures are essential to maintain consistency between batches, particularly when scaling from laboratory to pilot or production scale. The following guide outlines the critical steps involved in this biocatalytic transformation, serving as a foundational reference for technical teams evaluating the feasibility of this route.
- Cultivate selected microorganisms such as Saccharomyces cerevisiae or Geotrichum candidum in a nutrient medium until optimal optical density is reached.
- Introduce the 3-oxo-carboxylic acid derivative substrate into the microbial suspension under controlled pH and temperature conditions.
- Isolate the resulting enantiomerically enriched 3-hydroxy product via extraction and purification methods such as distillation or crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this biocatalytic technology offers profound advantages that address key pain points in the procurement and management of complex pharmaceutical intermediates. The shift from resolution-based methods to direct asymmetric synthesis eliminates the inherent 50% yield loss, effectively doubling the output from the same amount of raw materials and significantly lowering the unit cost of production. This efficiency gain is compounded by the reduction in waste disposal costs and the simplification of the purification workflow, which together contribute to a more lean and agile manufacturing operation. For procurement managers, this means access to a more cost-competitive supply base without compromising on the stringent quality standards required for pharmaceutical applications. The use of readily available starting materials and common fermentation equipment further enhances the economic attractiveness of this route, making it a viable option for large-scale commercialization.
- Cost Reduction in Manufacturing: The elimination of expensive chiral catalysts and the reduction in solvent usage directly lower the variable costs associated with production. By avoiding the need for stoichiometric resolving agents, the process reduces raw material expenses and minimizes the environmental footprint associated with chemical waste. This qualitative improvement in process efficiency allows for more competitive pricing structures, enabling partners to achieve substantial cost savings over the lifecycle of the product. The streamlined workflow also reduces labor and overhead costs, as fewer unit operations are required to achieve the final purity specifications.
- Enhanced Supply Chain Reliability: The use of robust microbial strains and standard fermentation technology ensures a stable and scalable supply source that is less susceptible to the volatility of specialized chemical reagent markets. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, as production can be ramped up quickly to meet demand fluctuations without the need for complex supply chain adjustments. The ability to produce material in-house using established bioprocessing infrastructure mitigates the risk of supply disruptions, providing a secure foundation for long-term procurement strategies.
- Scalability and Environmental Compliance: The aqueous nature of the reaction and the use of biological catalysts align perfectly with increasingly stringent environmental regulations, reducing the burden of compliance and permitting. This process is inherently scalable, as fermentation technology is well-understood and easily transferred from laboratory to industrial scale, facilitating the commercial scale-up of complex pharmaceutical intermediates. The reduced generation of hazardous waste simplifies disposal protocols and lowers the risk of environmental incidents, enhancing the overall sustainability profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this biocatalytic synthesis route. These answers are derived directly from the technical disclosures within the patent documentation, providing a clear understanding of the process capabilities and limitations. For stakeholders evaluating this technology, these insights offer a practical perspective on how it can be integrated into existing manufacturing frameworks to drive value. Understanding these details is essential for making informed decisions about process adoption and supplier selection.
Q: What is the primary advantage of this biocatalytic method over traditional chemical resolution?
A: The primary advantage is the ability to achieve high enantiomeric excess without the theoretical 50% yield limit associated with traditional racemic resolution, significantly improving overall process efficiency.
Q: Which microorganisms are preferred for this reduction process?
A: Yeasts and fungi are preferred, specifically strains such as Saccharomyces cerevisiae and Geotrichum candidum, which demonstrate high stereoselectivity for the (S)-enantiomer.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent describes fermentation and cultivation methods that are adaptable to industrial fermenters, ensuring scalability and consistent supply for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Heteroaryl-3-Hydroxypropionic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthesis routes for complex pharmaceutical intermediates, and we possess the technical expertise to bring this biocatalytic technology to commercial fruition. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by global pharmaceutical clients. Our commitment to quality and reliability makes us an ideal partner for companies seeking to secure a stable supply of high-value chiral intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your project requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make data-driven decisions about your sourcing strategy. Our goal is to collaborate closely with you to engineer solutions that enhance your competitive advantage in the marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
