Advanced Chiral Resolution Technology for High-Purity Phencynonate Intermediates and Commercial Scalability
The pharmaceutical landscape for anti-motion sickness agents is undergoing a significant transformation driven by the demand for higher efficacy and reduced side effect profiles. Central to this evolution is the development of optically pure intermediates for Phencynonate, a selective anticholinergic drug widely used for preventing motion sickness and treating vertigo. Patent CN100341850C introduces a groundbreaking methodology for the preparation of demethylphencynonate optical isomer salts, specifically utilizing N-p-toluenesulfonyl glutamic acid as a superior chiral resolving agent. This technology addresses the critical bottleneck in synthesizing single-enantiomer Phencynonate, offering a robust pathway to achieve optical purity exceeding 98%. For R&D directors and procurement specialists, this patent represents a pivotal shift from traditional racemic synthesis to precision chiral manufacturing, ensuring that the final API delivers maximum therapeutic potential with minimized dosage requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
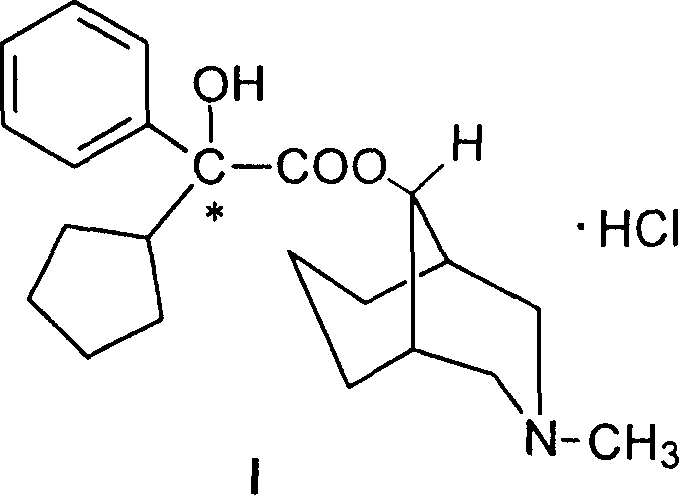
Historically, the industrial production of Phencynonate has relied heavily on the synthesis of its racemic form, primarily because direct optical resolution of the final drug molecule presents formidable chemical challenges. The molecular structure of Phencynonate hydrochloride features a tertiary nitrogen atom within a bridged azabicyclo system, which inherently possesses weak basicity. This chemical characteristic severely hampers the formation of stable diastereomeric salts when attempting to use conventional chiral acids such as tartaric acid or mandelic acid. Without stable salt formation, the fundamental principle of resolution—exploiting solubility differences between diastereomers—cannot be effectively applied. Consequently, manufacturers have been forced to produce and administer the racemate, despite evidence suggesting that the biological activity is concentrated almost exclusively in one enantiomer. This inefficiency not only doubles the metabolic load on the patient but also complicates the regulatory pathway for next-generation generic approvals that demand higher purity standards.
The Novel Approach
The innovative strategy outlined in the patent data circumvents the basicity issue through a clever structural modification prior to the resolution step. By first converting Phencynonate into its N-demethylated derivative, the chemical environment around the nitrogen atom is altered, facilitating more effective interaction with chiral acids. The core of this novelty lies in the selection of N-p-toluenesulfonyl glutamic acid as the resolving agent. Unlike simple organic acids, this sulfonamide-derived amino acid offers a rigid chiral framework and multiple hydrogen-bonding sites that interact selectively with the demethylphencynonate enantiomers. This specific pairing results in diastereomeric salts, designated as (-)III·(+)IV and (+)III·(-)IV, which exhibit drastically different solubility profiles in alcoholic solvents. This allows for a highly efficient separation via recrystallization, a unit operation that is far more scalable and cost-effective than preparative chiral chromatography, thereby solving the long-standing supply chain bottleneck for high-purity Phencynonate intermediates.
Mechanistic Insights into Chiral Recognition and Salt Formation
The success of this resolution process is rooted in the precise stereoelectronic complementarity between the demethylphencynonate substrate and the N-p-toluenesulfonyl glutamic acid resolver. When the racemic demethylphencynonate interacts with a single enantiomer of the chiral acid, two distinct diastereomeric salts are formed. The crystal lattice energy of these salts differs significantly due to the spatial arrangement of the bulky cyclopentyl and phenyl groups relative to the sulfonamide moiety of the acid. In the preferred solvent system, typically ethanol or methanol at elevated temperatures (50-80°C), one diastereomer reaches its saturation point and precipitates out of the solution much earlier than the other. This thermodynamic preference is amplified through iterative recrystallization cycles, typically performed 3 to 5 times, which progressively enriches the solid phase with the desired enantiomer while leaving the unwanted isomer in the mother liquor. The patent data explicitly notes that this method yields optical isomers with purity greater than 98%, a benchmark that is critical for meeting modern pharmacopoeial standards.
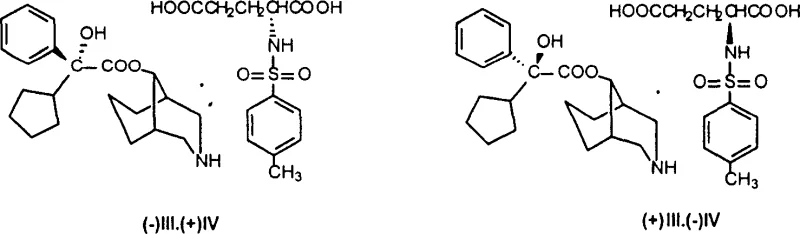
Furthermore, the process incorporates a robust mechanism for maximizing yield through mother liquor recycling. After the primary precipitation of the first enantiomeric salt, the remaining mother liquor contains an enriched concentration of the opposite enantiomer. By evaporating the solvent and treating the residue with the opposite enantiomer of the chiral acid (e.g., switching from (+)IV to (-)IV), the second isomer can be recovered and purified with similar efficiency. This dual-recovery approach ensures that the overall atom economy of the process is optimized, minimizing waste generation. Following the isolation of the optically pure salt, a straightforward basification step using dilute alkali solutions (5-10% KOH or NaOH) liberates the free base of the demethylphencynonate. This free base is then subjected to a final methylation step, typically using methyl iodide in the presence of a base like potassium carbonate, to regenerate the tertiary amine functionality, thus completing the transformation back to the therapeutically active Phencynonate structure but now in its optically pure form.
How to Synthesize Demethylphencynonate Salts Efficiently
The synthesis of these critical intermediates requires strict adherence to the reaction parameters defined in the patent to ensure consistent optical purity and yield. The process begins with the protection and subsequent deprotection of the amine to facilitate the resolution, followed by the critical salt formation step. Operators must maintain precise temperature controls during the recrystallization phases, as slight deviations can impact the enantiomeric excess of the precipitate. The detailed standardized synthetic steps, including specific molar ratios, solvent volumes, and agitation speeds required for GMP-compliant manufacturing, are outlined in the technical guide below.
- Demethylation of Phencynonate Racemate: React phencynonate hydrochloride with 2,2,2-trichloroethoxycarbonyl chloride followed by reductive cleavage with zinc powder to obtain demethylphencynonate.
- Diastereomeric Salt Formation: React the demethylphencynonate racemate with a single enantiomer of N-p-toluenesulfonyl glutamic acid in ethanol to form insoluble diastereomeric salts.
- Purification and Recovery: Perform repeated recrystallization (3-5 times) to isolate the optically pure salt, followed by basification to recover the free base intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this resolution technology offers substantial strategic advantages over traditional racemic synthesis or chromatographic separation methods. The primary value driver is the elimination of expensive and low-throughput chiral HPLC columns, which are often the bottleneck in scaling up chiral drug production. By relying on crystallization-induced diastereomeric resolution, the process utilizes standard stainless steel reactors and filtration equipment that are ubiquitous in fine chemical manufacturing facilities. This compatibility with existing infrastructure significantly lowers the barrier to entry for commercial scale-up, allowing for rapid transition from pilot plant batches to multi-ton annual production without requiring capital-intensive equipment upgrades. Furthermore, the starting materials, including glutamic acid and p-toluenesulfonyl chloride, are commodity chemicals with stable global supply chains, mitigating the risk of raw material shortages that often plague specialized chiral pool syntheses.
- Cost Reduction in Manufacturing: The economic model of this process is highly favorable due to the reliance on recrystallization rather than chromatography. Chromatographic separation typically involves high solvent consumption, expensive stationary phases, and low throughput, all of which drive up the cost per kilogram of the API. In contrast, the salt formation method described in CN100341850C utilizes inexpensive solvents like ethanol and benzene derivatives, and the resolving agent can potentially be recovered and recycled from the mother liquors after basification. The ability to recover both enantiomers from the racemic starting material further enhances the effective yield, ensuring that nearly 100% of the input material is converted into a valuable product stream rather than being discarded as waste. This comprehensive utilization of feedstock translates directly into a lower cost of goods sold (COGS) for the final pharmaceutical product.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for pharmaceutical buyers, and this synthetic route offers superior robustness. The reaction conditions are mild, typically ranging from ambient temperature to 100°C, and do not require exotic catalysts or sensitive organometallic reagents that might be subject to export controls or supply volatility. The use of zinc powder for the reductive deprotection step is a well-established industrial practice with a secure supply base. Additionally, the intermediate salts are crystalline solids with defined melting points (e.g., 130-132°C for the acid, 138-139°C for the ester salts), which facilitates easy handling, storage, and transportation compared to unstable oils or liquids. This physical stability reduces the risk of degradation during logistics, ensuring that the material arrives at the formulation site with its quality attributes intact.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process is designed for scalability. While the initial demethylation step involves chlorinated reagents, the subsequent workup and purification steps rely on aqueous washes and alcohol-based recrystallizations, which simplify waste stream management. The high optical purity achieved (>98%) reduces the burden on downstream purification, meaning less solvent and energy are required in the final API finishing stages. The ability to recycle the mother liquor to recover the counter-enantiomer aligns with green chemistry principles by maximizing atom economy. For manufacturers aiming to reduce their carbon footprint and meet stringent environmental regulations, this resolution pathway offers a cleaner alternative to less efficient separation technologies, supporting long-term sustainability goals while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these chiral intermediates. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for feasibility assessments. Understanding these nuances is essential for technical teams evaluating the integration of this pathway into their existing manufacturing portfolios.
Q: Why is direct resolution of phencynonate hydrochloride difficult?
A: Phencynonate contains a tertiary nitrogen structure which exhibits weak basicity, making it difficult to form stable salts directly with common acidic resolution reagents like tartaric acid or mandelic acid.
Q: What is the optical purity achievable with this method?
A: The patented process utilizing N-p-toluenesulfonyl glutamic acid allows for the production of phencynonate optical isomers with an optical purity greater than 98%.
Q: How does the L-isomer compare to the racemate in efficacy?
A: Preliminary studies indicate that the L-phencynonate isomer possesses significantly higher affinity for rat brain M receptors, exhibiting anticholinergic activity approximately 18 to 21 times stronger than the D-isomer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phencynonate Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from racemic to chiral drugs requires a partner with deep technical expertise and proven manufacturing capabilities. Our team has extensively analyzed the pathways described in CN100341850C and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand the critical nature of maintaining stringent purity specifications, particularly when dealing with chiral intermediates where minor impurities can compromise the efficacy of the final anti-motion sickness medication. Our rigorous QC labs are equipped with advanced chiral HPLC and polarimetry tools to ensure that every batch of demethylphencynonate salt meets the >98% optical purity benchmark before it leaves our facility, guaranteeing consistency for your downstream synthesis.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced resolution technology for your next-generation anti-vertigo formulations. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can reduce your overall manufacturing expenses. We encourage you to contact our technical procurement team today to request specific COA data for our chiral intermediates and to discuss route feasibility assessments for your project timelines. Let us help you secure a reliable supply of high-quality Phencynonate intermediates that drive both clinical efficacy and commercial success.
