Advanced Oxidation Strategy for High-Purity EXP-3174: Scaling Commercial Production
Advanced Oxidation Strategy for High-Purity EXP-3174: Scaling Commercial Production
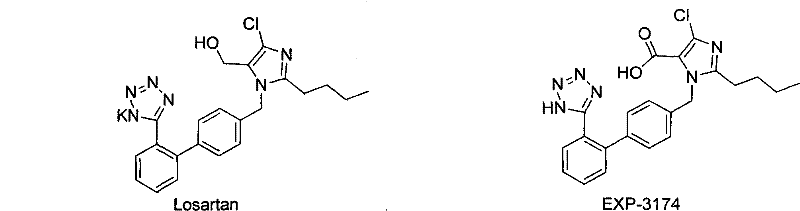
The pharmaceutical industry continuously seeks robust synthetic routes for critical Angiotensin II receptor antagonists, specifically focusing on the active metabolites that drive therapeutic efficacy. Patent CN102190652A introduces a groundbreaking preparation method for EXP-3174, the primary active metabolite of Losartan, which exhibits significantly higher potency than the parent compound. This technical disclosure outlines a sophisticated oxidation protocol transforming the aldehyde precursor into the corresponding carboxylic acid with exceptional efficiency. By leveraging a sodium chlorite-antichlor system within a buffered polar solvent environment, the process achieves yields exceeding 85% and purity levels greater than 99.0%. For R&D directors and procurement specialists, this represents a pivotal shift away from legacy methods plagued by heavy metal contamination and low throughput. The following analysis dissects the mechanistic advantages and commercial viability of this route, positioning it as a cornerstone for reliable pharmaceutical intermediate supplier strategies in the cardiovascular sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Losartan metabolites has been hindered by aggressive oxidation protocols that compromise both environmental safety and product quality. Traditional approaches, such as the direct oxidation of Losartan potassium using potassium permanganate, necessitate fierce reaction conditions that generate substantial quantities of manganese dioxide byproducts. These inorganic sludges create severe downstream processing bottlenecks, requiring complex filtration and purification steps that drastically reduce overall recovery rates. Furthermore, literature indicates that alternative microwave-assisted oxidation methods often struggle with incomplete conversion, leaving significant amounts of the intermediate aldehyde (EXP-3179) unreacted. This results in crude products with impurity profiles that demand preparative chromatography, a technique that is economically prohibitive and technically challenging to implement at an industrial scale. Consequently, legacy methods frequently report yields hovering between 51% and 71%, failing to meet the stringent economic and purity demands of modern API manufacturing.
The Novel Approach
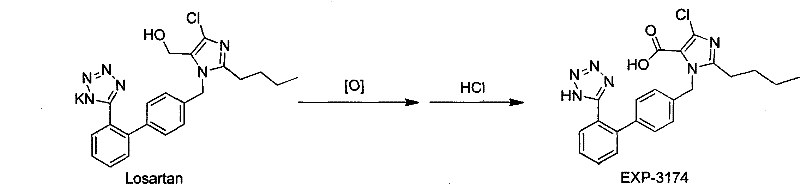
In stark contrast, the methodology disclosed in CN102190652A utilizes a controlled sodium chlorite oxidation system that operates under remarkably mild conditions, typically around 5°C. By employing a buffer solution to maintain the pH between 1 and 7, preferably at 4.5, the reaction selectively oxidizes the aldehyde group to the carboxylic acid without degrading the sensitive tetrazole or imidazole rings. This precision eliminates the formation of complex byproduct mixtures associated with permanganate oxidation, thereby simplifying the isolation process to a straightforward liquid-liquid extraction. The use of hydrogen peroxide in conjunction with sodium chlorite ensures a clean reaction profile where the primary byproduct is water, aligning perfectly with green chemistry principles. This novel approach not only enhances the crude product purity to over 99% but also facilitates easy recrystallization, effectively bypassing the need for costly chromatographic separation and enabling seamless transition from laboratory bench to commercial reactor.
Mechanistic Insights into Sodium Chlorite-Antichlor Oxidation
The core innovation of this synthesis lies in the generation of chlorous acid in situ, which acts as the active oxidizing species for the aldehyde functionality. In the presence of a buffered medium, sodium chlorite reacts to form the reactive intermediate that selectively targets the carbonyl carbon of the EXP-3179 precursor. The addition of hydrogen peroxide serves a dual purpose: it aids in the activation of the oxidant and helps manage the reaction kinetics to prevent runaway exotherms. Crucially, the buffer system stabilizes the pH, preventing the acidic degradation of the tetrazole moiety which is susceptible to harsh acidic environments. This mechanistic control ensures that the oxidation proceeds cleanly to the carboxylic acid stage without over-oxidizing other parts of the biphenyl-imidazole scaffold. The result is a highly specific transformation that preserves the stereochemical and structural integrity of the molecule, which is paramount for maintaining the biological activity required for AT1 receptor antagonism.
Impurity control is inherently built into this mechanism through the use of a scavenging agent, specifically sodium sulfite, added post-reaction. This step effectively quenches any residual oxidative species, preventing secondary reactions during the workup phase that could lead to chlorinated impurities or ring-opening degradation. Unlike permanganate methods where manganese ions can coordinate with the tetrazole nitrogen atoms leading to difficult-to-remove complexes, the sodium chlorite system leaves behind soluble salts that are easily washed away during the aqueous extraction phases. This chemical orthogonality allows for a much cleaner impurity profile, reducing the burden on analytical QC labs to identify and quantify trace metal contaminants. For process chemists, this means a more predictable crystallization behavior and a final drug substance that consistently meets pharmacopeial standards for heavy metals and related substances without extensive reprocessing.
How to Synthesize EXP-3174 Efficiently
The operational protocol for this synthesis is designed for reproducibility and safety, utilizing standard glassware or stainless steel reactors commonly found in fine chemical facilities. The process begins with the dissolution of the aldehyde starting material in a polar solvent such as isopropanol or dimethyl sulfoxide, ensuring a homogeneous reaction mixture. Critical control points include the slow addition of the sodium chlorite solution to manage heat evolution and the strict maintenance of the pH window using phosphate or acetate buffers. Following the oxidation period, the reaction is quenched, and the product is isolated through solvent exchange and extraction, yielding a white solid that requires minimal further purification. The detailed standardized synthesis steps, including specific molar ratios and agitation speeds, are outlined in the technical guide below to ensure consistent batch-to-batch quality.
- Dissolve the aldehyde precursor (EXP-3179) in a polar solvent such as isopropanol or DMSO under stirring to form a clear solution.
- Adjust the pH to between 1.0 and 7.0 using a buffer system, then add hydrogen peroxide and slowly drip sodium chlorite solution while maintaining temperature between -10°C and 30°C.
- Quench excess oxidant with sodium sulfite, remove solvent via distillation, and extract the product with ethyl acetate to obtain high-purity EXP-3174.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this oxidation technology translates directly into enhanced operational resilience and significant cost optimization opportunities. By eliminating the reliance on heavy metal oxidants like potassium permanganate, manufacturers can avoid the substantial costs associated with hazardous waste disposal and environmental compliance reporting. The simplified workup procedure, which replaces complex chromatography with standard extraction and crystallization, drastically reduces the consumption of expensive silica gel and organic solvents. This streamlining of the downstream process shortens the overall production cycle time, allowing facilities to increase throughput without requiring additional capital investment in specialized equipment. Furthermore, the high yield and purity achieved reduce the need for re-processing batches, thereby minimizing material loss and ensuring a more stable supply of critical intermediates for downstream API formulation.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the subsequent removal steps significantly lowers the raw material and waste treatment expenses. Since the reaction byproducts are primarily water and soluble salts, the effluent treatment load is substantially decreased compared to traditional methods. This reduction in chemical intensity and waste volume leads to a leaner cost structure, making the production of high-purity Losartan metabolite more economically viable in competitive markets. Additionally, the ability to use common polar solvents like isopropanol or acetone, rather than exotic reagents, further stabilizes the supply chain against price volatility.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent output even when scaling from pilot plants to multi-tonne production lines. Because the reaction conditions are mild and do not require high-pressure or high-temperature equipment, the risk of unplanned downtime due to equipment failure is minimized. The use of readily available reagents such as sodium chlorite and hydrogen peroxide means that sourcing is not dependent on single-source suppliers of niche catalysts. This diversification of the supply base mitigates the risk of shortages and ensures that delivery schedules for key pharmaceutical intermediates can be met reliably, supporting continuous API manufacturing operations globally.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations that are standard in the fine chemical industry. The absence of toxic heavy metals simplifies the regulatory filing process for new drug master files, as residual metal limits are easily met without complex scavenging resins. This environmental friendliness aligns with increasingly stringent global regulations regarding pharmaceutical manufacturing emissions. Facilities adopting this method can achieve higher production capacities with a smaller environmental footprint, satisfying both corporate sustainability goals and regulatory requirements for green chemistry practices in the synthesis of cardiovascular therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this oxidation process. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, focusing on practical aspects of quality control and process adaptation. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing manufacturing workflows. The responses highlight the specific advantages in terms of purity profiles and operational simplicity that distinguish this method from prior art.
Q: How does this sodium chlorite method compare to traditional potassium permanganate oxidation?
A: Unlike the harsh potassium permanganate method which generates manganese dioxide sludge and requires difficult purification, this novel approach uses mild conditions (5°C, pH 4.5) yielding over 85% purity with simple extraction workups.
Q: What are the critical process parameters for maximizing yield in EXP-3174 synthesis?
A: Maintaining the reaction temperature at approximately 5°C and controlling the pH within the 4.5 range using a phosphate or acetate buffer are essential to prevent over-oxidation and ensure yields exceed 85%.
Q: Is this oxidation process suitable for large-scale industrial manufacturing?
A: Yes, the process avoids expensive chromatography and hazardous heavy metals, utilizing standard reactors and simple liquid-liquid extraction, making it highly adaptable for tonne-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable EXP-3174 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving cardiovascular medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market supply is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of EXP-3174 meets the highest international standards. Our commitment to process excellence means we can deliver this complex imidazole derivative with the consistency and reliability required by top-tier pharmaceutical companies.
We invite you to collaborate with us to optimize your supply chain for Losartan metabolites. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced oxidation technology can enhance your production efficiency. Let us be your partner in delivering high-purity pharmaceutical intermediates that drive the success of your generic and innovative drug portfolios.
