Advanced Synthesis of O-Amino-P-Tert-Butylphenol for Commercial Scale-Up
The global demand for high-performance fluorescent whitening agents, such as OB and EFT series, necessitates a robust and safe supply chain for their critical precursors. Patent CN101774933B introduces a transformative methodology for the preparation of o-amino-p-tert-butylphenol, a pivotal intermediate in this value chain. Unlike traditional routes that suffer from safety hazards and environmental burdens, this invention leverages a clever cascade of diazotization, hydrolysis, and nitration starting from p-tert-butylaniline. For R&D directors and procurement strategists, this patent represents a significant opportunity to optimize manufacturing protocols. By shifting the synthetic entry point from phenol derivatives to amine derivatives, the process fundamentally alters the impurity profile and safety landscape. The technical breakthrough lies in the seamless integration of the first three reaction steps into a single vessel, drastically reducing unit operations and associated handling risks. This report analyzes the technical merits and commercial viability of this approach, positioning it as a superior alternative for the reliable o-amino-p-tert-butylphenol supplier market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of o-amino-p-tert-butylphenol has relied on two primary pathways, both of which present significant operational challenges for large-scale manufacturing. The first conventional method involves the direct nitration of p-tert-butylphenol followed by reduction. While conceptually simple, this route is plagued by severe safety issues; the nitration step inevitably generates polynitrated byproducts. During the subsequent vacuum distillation required to isolate the mono-nitro intermediate, the concentration of these unstable polynitrates increases in the still pot, leading to localized overheating and a high risk of explosion. Furthermore, the reduction step in this traditional route often suffers from low yields and generates substantial sulfide waste if alkali sulfides are used. The second conventional pathway utilizes an azo coupling reaction between diazotized aniline and p-tert-butylphenol, followed by reduction. Although this avoids direct nitration of the phenol, the process is operationally cumbersome, involving multiple isolation steps and generating stoichiometric amounts of aniline byproduct that, while recyclable, adds complexity to the workflow. Both methods struggle to meet modern standards for cost reduction in fluorescent whitener intermediate manufacturing due to their inherent inefficiencies and safety liabilities.
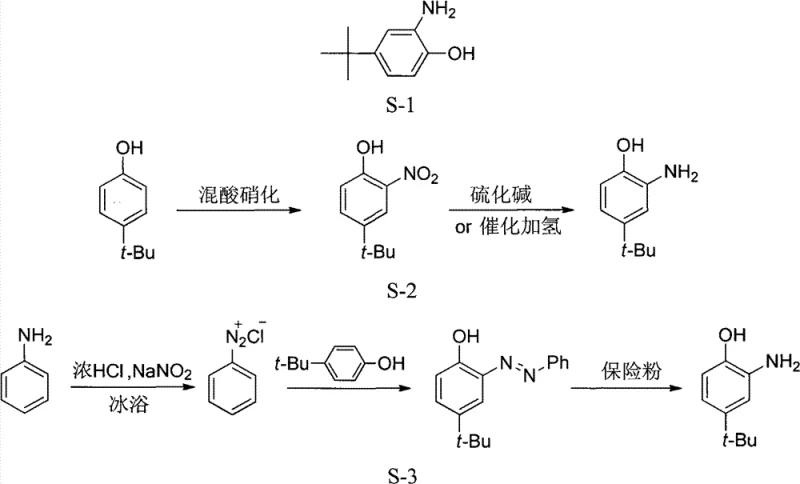
The Novel Approach
The methodology disclosed in CN101774933B circumvents these historical bottlenecks by reversing the synthetic logic, starting with p-tert-butylaniline instead of the phenol. This strategic shift allows for a controlled diazotization followed by a unique one-pot hydrolysis and nitration sequence. By generating the diazonium salt first and then treating it directly with dilute nitric acid under reflux, the process achieves the introduction of the nitro group and the conversion of the amine to a hydroxyl group simultaneously. This eliminates the need for hazardous vacuum distillation of unstable nitro-phenols, as the intermediate o-nitro-p-tert-butylphenol can be cleanly separated via steam distillation. The final reduction step employs hydrazine hydrate catalyzed by transition metal chlorides, a system that operates under atmospheric pressure and avoids the high-pressure hydrogenation equipment typically required for nitro reductions. This novel approach not only enhances safety by removing explosion risks but also simplifies the equipment footprint, making it an ideal candidate for commercial scale-up of complex fine chemical intermediates.
Mechanistic Insights into Diazotization-Hydrolysis-Nitration Cascade
The core innovation of this synthesis lies in the precise control of the diazonium intermediate and its subsequent transformation. The process initiates with the diazotization of p-tert-butylaniline in dilute sulfuric acid at a strictly controlled temperature range of 0-5°C. Maintaining this low temperature is critical to prevent the premature decomposition of the diazonium salt, which would lead to tar formation and loss of yield. Once the diazonium species is fully formed, indicated by a positive starch-iodide test, the reaction mixture is not isolated. Instead, dilute nitric acid is added directly, and the mixture is heated to reflux. This triggers a dual mechanism: the hydrolysis of the diazonium group to form the phenolic hydroxyl moiety and the electrophilic aromatic substitution by the nitronium ion generated in situ. The steric hindrance provided by the tert-butyl group directs the nitration predominantly to the ortho position relative to the newly formed hydroxyl group, ensuring high regioselectivity.
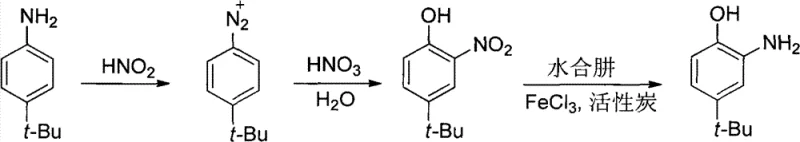
Following the nitration, the crude o-nitro-p-tert-butylphenol is isolated via steam distillation, a technique that effectively separates the volatile product from non-volatile tarry byproducts and inorganic salts. The final transformation involves the reduction of the nitro group to an amine using hydrazine hydrate. This reduction is catalyzed by ferric chloride (FeCl3) in the presence of activated carbon. The mechanism likely involves the formation of a metal-hydrazine complex that facilitates electron transfer to the nitro group. Crucially, the addition of a small amount of sodium dithionite acts as an antioxidant, preventing the oxidation of the sensitive amino-phenol product during the workup. This multi-faceted catalytic system ensures that the final product achieves a chromatographic purity of greater than 99%, meeting the stringent requirements for high-purity OLED material and fluorescent whitener applications without the need for extensive chromatographic purification.
How to Synthesize O-Amino-P-Tert-Butylphenol Efficiently
Implementing this synthesis requires careful attention to the stoichiometry and thermal profiles described in the patent examples. The process is designed to be telescoped, meaning the intermediate diazonium salt does not need to be isolated, which reduces solvent usage and processing time. Operators must ensure that the molar ratio of sulfuric acid to amine is maintained between 1.2:1 and 2.5:1 to ensure complete salt formation without excessive acidity that might complicate downstream neutralization. Similarly, the nitric acid addition must be controlled to balance the hydrolysis and nitration rates. For detailed operational parameters, including specific stirring rates and addition times that maximize yield, please refer to the standardized protocol below.
- Diazotization of p-tert-butylaniline in dilute sulfuric acid at 0-5°C using sodium nitrite.
- Direct addition of dilute nitric acid for simultaneous hydrolysis and nitration, followed by steam distillation.
- Catalytic reduction of the nitro-intermediate using hydrazine hydrate and ferric chloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible strategic benefits beyond mere technical elegance. The primary advantage is the drastic simplification of the production workflow. By combining diazotization, hydrolysis, and nitration into a single reactor train, the requirement for intermediate storage tanks and transfer pumps is eliminated. This reduction in unit operations translates directly to lower capital expenditure (CAPEX) for new facilities and reduced maintenance costs for existing ones. Furthermore, the use of atmospheric pressure throughout the entire sequence removes the need for expensive high-pressure autoclaves typically associated with catalytic hydrogenation, further lowering the barrier to entry for production.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the elimination of costly separation steps. Traditional methods often require energy-intensive vacuum distillation or complex extraction protocols to remove polynitrated impurities. In contrast, the steam distillation step in this novel route is energy-efficient and highly effective at purification. Additionally, the catalyst system utilizes inexpensive iron salts rather than precious metals like palladium or platinum, which are subject to volatile market pricing. The ability to recycle solvents like ethanol or methanol, as demonstrated in the patent examples, further drives down the variable cost per kilogram, ensuring substantial cost savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialized reagents or hazardous intermediates that are difficult to transport. This synthesis relies on commodity chemicals such as sulfuric acid, nitric acid, and hydrazine hydrate, which are readily available from multiple global suppliers. The robustness of the process against minor fluctuations in reaction conditions means that batch-to-batch variability is minimized, reducing the risk of off-spec material that could disrupt downstream customer production. By mitigating the safety risks associated with polynitrate accumulation, the process also reduces the likelihood of unplanned shutdowns due to safety incidents, thereby ensuring a more stable and predictable delivery schedule for clients.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions intensify, the environmental profile of a chemical process becomes a key differentiator. This method generates significantly less hazardous waste compared to sulfide reduction routes, which produce toxic sulfur-containing effluents. The byproducts of the diazotization-hydrolysis step are primarily nitrogen gas, water, and inorganic salts, which are easier to treat in standard wastewater facilities. The absence of heavy metal contamination in the final product simplifies the disposal of mother liquors. This green chemistry profile facilitates easier permitting for plant expansions and aligns with the sustainability goals of major multinational corporations seeking responsible partners for their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of technology transfer or contract manufacturing agreements.
Q: Why is the diazotization route safer than direct nitration?
A: Direct nitration of p-tert-butylphenol often leads to polynitrated byproducts which accumulate during distillation, creating severe explosion hazards. The diazotization route avoids this accumulation entirely.
Q: What are the purity specifications achievable with this method?
A: The patented process utilizes steam distillation and specific recrystallization steps to achieve chromatographic purity greater than 99%, suitable for high-grade fluorescent whitener synthesis.
Q: Does this process require high-pressure equipment?
A: No, the entire reaction sequence, including the reduction step using hydrazine hydrate, operates under atmospheric pressure, significantly lowering capital expenditure for reactor vessels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-Amino-P-Tert-Butylphenol Supplier
The technical potential of the diazotization-hydrolysis-nitration route is immense, offering a pathway to high-quality intermediates with a superior safety and environmental profile. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this patented chemistry to life. Our facility is equipped with state-of-the-art reactors capable of handling exothermic diazotization reactions safely, along with rigorous QC labs that enforce stringent purity specifications to ensure every batch meets the >99% purity benchmark. We understand that consistency is key for your downstream applications, whether in fluorescent whitening or advanced material synthesis.
We invite you to collaborate with us to leverage this advanced manufacturing capability. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your total landed cost. Please contact our technical procurement team today to request specific COA data from our pilot batches and comprehensive route feasibility assessments. Let us partner with you to secure a sustainable and efficient supply of this critical chemical building block.
