Scaling High-Purity o-Amino-p-tert-butylphenol Production via Safe Normal-Pressure Catalytic Hydrogenation
The chemical industry's demand for high-purity intermediates used in optical brighteners and specialty polymers is constantly evolving, driven by the need for safer and more sustainable manufacturing processes. Patent CN101948393B introduces a transformative normal-pressure production method for o-amino-p-tert-butylphenol, a critical precursor for the synthesis of fluorescent whitening agent OB. This technology addresses the longstanding safety hazards and environmental burdens associated with traditional high-pressure hydrogenation and wet chemical reduction methods. By leveraging a specific catalytic system involving skeleton nickel and lower fatty alcohols, the process achieves exceptional conversion rates at pressures below 0.5 MPa. For R&D directors and procurement strategists, this represents a significant opportunity to optimize supply chains for fine chemical intermediates while adhering to stricter environmental regulations. The ability to produce this key intermediate under near-atmospheric conditions fundamentally alters the economic and safety profile of the manufacturing landscape.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of o-amino-p-tert-butylphenol has been plagued by severe safety and environmental challenges that hinder scalable production. Traditional routes often rely on iron powder reduction in acidic media, which generates substantial quantities of iron sludge that are costly and difficult to dispose of, creating a heavy environmental burden. Alternatively, sulfide reduction methods produce large volumes of toxic, brown-black wastewater containing sulfur compounds, necessitating complex and expensive treatment facilities before discharge. Furthermore, conventional catalytic hydrogenation processes typically operate at elevated pressures ranging from 1.0 to 1.5 MPa to ensure adequate reaction kinetics. These high-pressure conditions require specialized, expensive reactor vessels and pose significant safety risks regarding hydrogen leakage at dynamic seals, which can lead to catastrophic accidents. The combination of hazardous waste generation and high-pressure operational requirements makes these legacy methods increasingly untenable in a modern regulatory environment focused on green chemistry and worker safety.
The Novel Approach
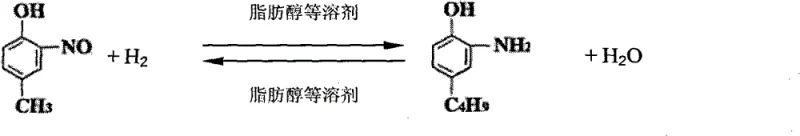
The innovative method described in the patent data circumvents these issues by utilizing a low-pressure catalytic hydrogenation strategy that operates effectively at pressures below 0.5 MPa, and in many embodiments, at normal atmospheric pressure. This approach employs a skeleton nickel catalyst in conjunction with lower fatty alcohol solvents, such as n-butanol or n-amyl alcohol, which serve a dual purpose. Not only do they dissolve the reactants, but they also act as water-carrying agents that continuously remove the water byproduct formed during the reduction of the nitro group. This in-situ water removal drives the reaction equilibrium towards completion without the need for high hydrogen pressure to force conversion. Consequently, the process eliminates the need for high-pressure equipment, drastically reducing capital investment and operational risk. The result is a cleaner, safer, and more economically viable pathway that yields white crystalline o-amino-p-tert-butylphenol with purity levels exceeding 98%, free from the heavy metal contamination associated with iron powder methods.
Mechanistic Insights into Skeletal Nickel-Catalyzed Hydrogenation
The core of this technological breakthrough lies in the synergistic interaction between the skeletal nickel catalyst and the specific choice of fatty alcohol solvents. In this catalytic cycle, molecular hydrogen is activated on the surface of the nickel catalyst, facilitating the reduction of the nitro group (-NO2) on the o-nitro-p-tert-butylphenol substrate to an amino group (-NH2). Unlike standard hydrogenations where water accumulation can inhibit catalyst activity or reverse the reaction, this system leverages the physical properties of C4-C10 fatty alcohols. These solvents form an azeotrope with the water generated during the reaction, allowing for continuous separation of water as the reaction proceeds. This mechanism effectively shifts the thermodynamic equilibrium towards the product side according to Le Chatelier's principle, ensuring high conversion rates even at low hydrogen partial pressures. The use of skeletal nickel provides a high surface area for catalysis, offering a cost-effective alternative to precious metal catalysts like palladium while maintaining robust activity and selectivity for the nitro-to-amino transformation.
Furthermore, the impurity profile of the final product is meticulously controlled through this specific mechanistic pathway. Traditional reduction methods often suffer from over-reduction or side reactions that generate complex organic impurities difficult to separate. In contrast, the selective nature of the nickel-catalyzed hydrogenation under these mild thermal conditions (80-200°C) minimizes side reactions such as de-alkylation of the tert-butyl group or ring hydrogenation. The subsequent purification steps, involving suction filtration, activated carbon decolorization, and recrystallization from the reaction solvent, further refine the product quality. The removal of water during the reaction also prevents hydrolysis side reactions that might occur in aqueous acidic environments typical of iron powder reductions. This results in a final product with a melting point of 164-166°C and a purity suitable for high-end applications in fluorescent whitening agents, where color stability and optical performance are paramount.
How to Synthesize o-Amino-p-tert-butylphenol Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety while minimizing waste. The process begins with the charging of the reactor with the nitro precursor, the fatty alcohol solvent, and the catalyst, followed by a rigorous purging sequence to eliminate oxygen. The detailed standardized synthesis steps below outline the critical operational parameters, including temperature ramping, pressure maintenance, and the crucial water separation phase that defines the success of this low-pressure methodology. Adhering to these protocols ensures that the reaction proceeds smoothly to completion within a short timeframe, typically between 0.5 to 5.0 hours, delivering consistent batch quality.
- Charge the reactor with o-nitro-p-tert-butylphenol, a lower fatty alcohol solvent (such as n-butanol), and a hydrogenation catalyst like skeleton nickel.
- Purge the system with nitrogen to remove air, then introduce hydrogen gas to maintain a pressure below 0.5 MPa while heating to 80-200°C.
- Continuously separate the water generated during the reaction using the solvent as a water carrier, then filter, decolorize, and recrystallize the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this normal-pressure production method offers substantial strategic advantages beyond mere technical feasibility. The shift from high-pressure to low-pressure processing fundamentally changes the cost structure of manufacturing this intermediate. By eliminating the requirement for high-pressure autoclaves and the associated safety infrastructure, the capital expenditure (CAPEX) for new production lines is significantly reduced. Additionally, the operational expenditure (OPEX) is lowered due to the reduced energy consumption required for compression and the elimination of costly waste treatment processes associated with iron sludge or sulfide wastewater. This creates a more resilient supply chain capable of withstanding regulatory shocks related to environmental compliance. The simplicity of the process also enhances scalability, allowing manufacturers to respond more agilely to market demand fluctuations without the long lead times associated with constructing high-pressure facilities.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of expensive and hazardous reagents with commodity chemicals. Traditional methods utilizing hydrazine hydrate involve high raw material costs and significant safety handling expenses, while iron powder reduction incurs hidden costs in waste disposal and environmental remediation. This catalytic hydrogenation method utilizes hydrogen gas and skeleton nickel, which are relatively inexpensive and widely available. Moreover, the ability to operate at normal pressure removes the need for specialized high-pressure vessel certifications and maintenance, leading to substantial long-term savings in equipment lifecycle costs. The high yield reported in the patent data, often exceeding 96%, further optimizes material utilization, reducing the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by environmental shutdowns or safety incidents in chemical manufacturing. This process mitigates those risks by adopting a green chemistry approach that generates minimal hazardous waste. The absence of toxic sulfide byproducts or iron sludge means the facility is less likely to face regulatory interruptions or fines. Furthermore, the raw materials required, such as n-butanol and hydrogen, are bulk commodities with stable global supply chains, reducing the risk of raw material shortages. The robustness of the skeleton nickel catalyst also contributes to reliability, as it is less sensitive to poisoning compared to some precious metal catalysts, ensuring consistent batch-to-batch performance and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: As global regulations tighten around industrial emissions, the scalability of a chemical process is inextricably linked to its environmental footprint. This method is inherently scalable because it avoids the engineering complexities of high-pressure scale-up, where heat transfer and mixing become critical failure points. The "green" nature of the reaction, producing only water as a byproduct alongside the desired amine, simplifies the permitting process for new plants or expansion projects. This ease of compliance facilitates faster time-to-market for capacity expansions. Additionally, the solvent system allows for potential recycling of the fatty alcohol after product crystallization, further enhancing the sustainability profile and reducing the overall volume of chemical waste requiring treatment, aligning perfectly with corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this normal-pressure hydrogenation technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy synthesis routes. Understanding these distinctions is crucial for technical teams evaluating process transfers or new vendor qualifications.
Q: What is the primary advantage of this normal-pressure method over traditional high-pressure hydrogenation?
A: The primary advantage is significantly enhanced operational safety and reduced capital expenditure. Traditional methods often require pressures of 1.0-1.5 MPa, necessitating expensive high-pressure vessels and posing leakage risks. This patented process operates effectively at pressures below 0.5 MPa, even approaching atmospheric pressure, which drastically lowers equipment investment and eliminates the risk of high-pressure hydrogen leaks.
Q: How does the solvent system contribute to the high yield of the reaction?
A: The process utilizes lower fatty alcohols (C4-C10), such as n-butanol or n-amyl alcohol, which function not only as solvents but also as water-carrying agents. By continuously separating the water produced during the hydrogenation reaction via azeotropic distillation principles, the chemical equilibrium is driven towards the product side. This ensures complete conversion of the nitro group to the amino group without requiring extreme pressure conditions.
Q: Why is this method considered more environmentally friendly than iron powder or sulfide reduction?
A: Unlike iron powder reduction which generates massive amounts of difficult-to-treat iron sludge, or sulfide reduction which produces toxic brown-black wastewater, this catalytic hydrogenation method is clean. The only byproduct is water, which is removed during the process. This eliminates the 'three wastes' pollution associated with older reduction technologies, aligning with modern green chemistry standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable o-Amino-p-tert-butylphenol Supplier
The transition to safer, low-pressure manufacturing methodologies requires a partner with deep technical expertise and proven scale-up capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, ensuring that every batch of o-amino-p-tert-butylphenol meets the exacting standards required for fluorescent whitening agent synthesis. We understand that consistency in melting point and color stability is non-negotiable for your downstream applications, and our advanced process controls are designed to deliver exactly that level of precision reliably.
We invite you to collaborate with us to leverage these advanced manufacturing efficiencies for your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this normal-pressure technology can reduce your total landed cost. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, high-quality supply of this critical intermediate while optimizing your production economics.
