Advanced Manufacturing of Benzopyrone Derivatives for Next-Generation LCD Applications
The landscape of electronic chemical manufacturing is constantly evolving, driven by the demand for high-performance liquid crystal materials with superior thermal and optical stability. A significant breakthrough in this domain is documented in patent CN116283876A, which discloses a robust and efficient preparation method for benzopyrone derivatives. These compounds are critical building blocks for liquid crystal monomers exhibiting negative dielectric anisotropy, a property essential for advanced display technologies. The invention addresses long-standing challenges in the industry by providing a synthetic route that bypasses the need for expensive and hazardous reagents while delivering exceptional yields. For R&D directors and procurement specialists seeking a reliable electronic chemical supplier, understanding the nuances of this technology is paramount for securing a competitive supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzopyrone compounds has been fraught with inefficiencies that hinder large-scale production. As illustrated in the prior art, traditional Route 1 typically begins with 2-fluoro-4-bromophenol, undergoing a series of steps including etherification and LDA-mediated formylation. This pathway is not only chemically complex but also suffers from abysmal overall yields, reported to be approximately 16% for alkyl-substituted derivatives. Furthermore, Route 2 relies on 3-fluoro-4-ethoxyphenylacetaldehyde, necessitating the use of aggressive lithium reagents under harsh conditions. These conventional methods often result in process yields hovering between 30% and 40%, creating significant bottlenecks in cost reduction in electronic chemical manufacturing. The reliance on cryogenic conditions and sensitive organometallic reagents also introduces substantial safety risks and operational complexities that are undesirable for continuous commercial operations.
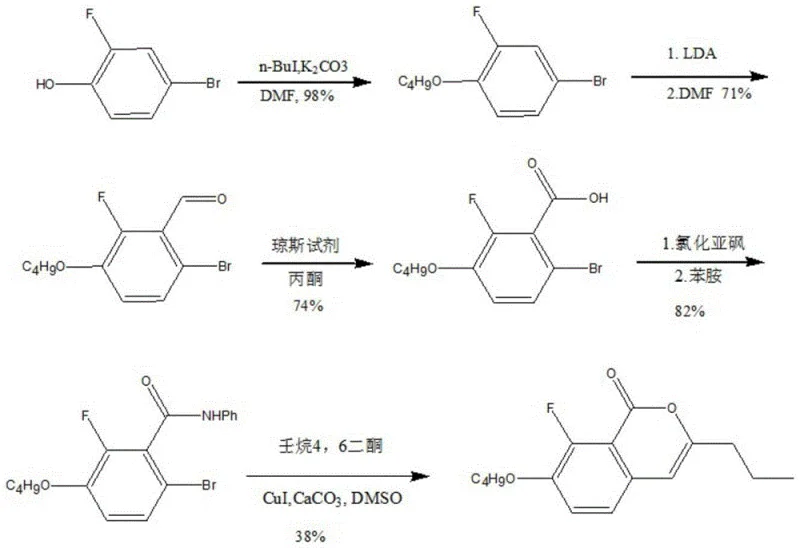
The Novel Approach
In stark contrast, the methodology outlined in patent CN116283876A offers a paradigm shift by utilizing trifluoronitrobenzene as a readily accessible starting material. This novel approach streamlines the synthesis into four distinct stages: ortho-substitution with nitromethane, para-substitution with alkoxides, a Nef reaction to convert nitromethyl groups to carboxyls, and a sophisticated sequence involving reduction and iron-catalyzed cyclization. By eliminating the need for LDA and reducing the dependency on extreme temperatures, this route significantly enhances process safety and operational simplicity. The strategic design of this synthesis allows for the production of high-purity OLED material precursors and liquid crystal intermediates with much greater efficiency, directly addressing the scalability issues plaguing older technologies.
Mechanistic Insights into Iron-Catalyzed Cyclization and Nef Reaction
The core of this technological advancement lies in its clever manipulation of functional groups through well-defined mechanistic steps. The process initiates with an ortho-substitution reaction where trifluoronitrobenzene reacts with nitromethane at temperatures between -10°C and -30°C. This is followed by a para-substitution with alkoxides to install the necessary ether linkage. A critical transformation occurs during the Nef reaction, where the nitromethyl group is converted into a carboxyl group using catalysts such as tetrabutylammonium iodide and zinc acetate in an aqueous medium at 80°C to 100°C. This step is pivotal for establishing the carbon skeleton required for the final lactone ring. Subsequently, the nitro group on the aromatic ring undergoes a reduction to an amine, followed by oxidation and denitrogenation rearrangement to form an aldehyde hydrazine intermediate. This intricate sequence ensures that the reactive handles are perfectly positioned for the final coupling.
The culmination of the synthesis involves a highly selective nucleophilic addition catalyzed by an iron complex, specifically Fe(dmpe)2Cl2. This catalyst facilitates the coupling of the aldehyde hydrazine intermediate with a specific aldehyde substrate to generate a secondary alcohol. The final ring closure is achieved via a Mitsunobu reaction, utilizing triphenylphosphine and diethyl azodicarboxylate at mild temperatures of 0°C to 10°C. This mechanistic precision is crucial for impurity control, as it minimizes side reactions that could compromise the optical properties of the final liquid crystal material. The ability to achieve GC purity levels of 99.9% demonstrates the efficacy of this catalytic system in producing commercial scale-up of complex polymer additives and electronic chemicals.
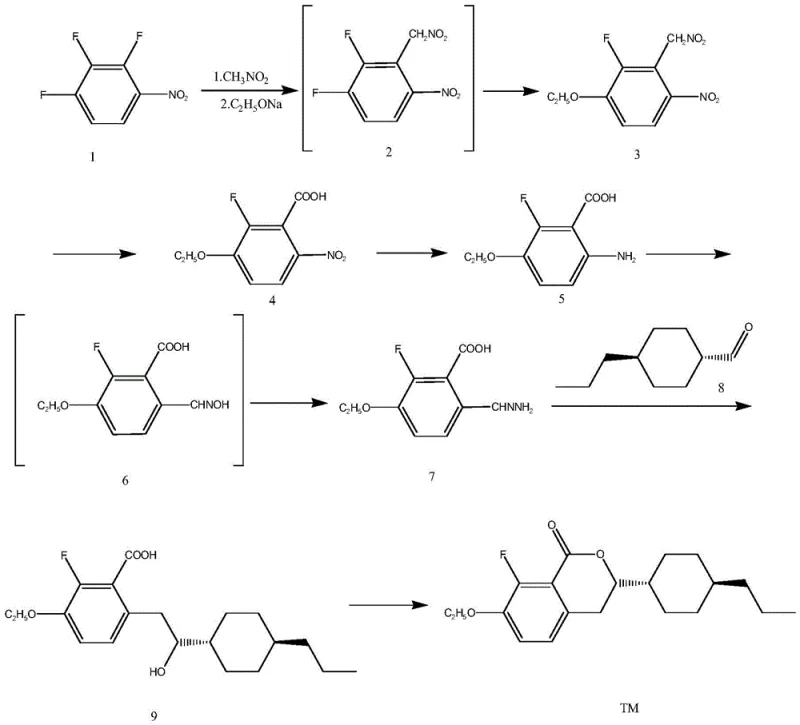
How to Synthesize Benzopyrone Efficiently
Implementing this synthesis requires careful attention to reaction parameters and purification protocols to maximize yield and purity. The process begins with the preparation of the nitromethyl intermediate, followed by the critical Nef oxidation which sets the stage for ring formation. The subsequent conversion to the aldehyde hydrazine must be managed precisely to avoid over-oxidation or incomplete reduction. Finally, the iron-catalyzed coupling and Mitsunobu cyclization demand strict control over stoichiometry and temperature to ensure the formation of the correct stereoisomer. For detailed operational guidelines, the standardized synthesis steps are provided below to assist technical teams in replicating these results.
- Perform ortho-substitution on trifluoronitrobenzene with nitromethane followed by para-substitution with alkoxide to obtain the first intermediate.
- Convert the nitromethyl group to a carboxyl group via a Nef reaction using zinc acetate and tetrabutylammonium iodide catalysts.
- Transform the nitro group into an aldehyde hydrazine intermediate through reduction, oxidation, and denitrogenation rearrangement.
- Execute iron-catalyzed nucleophilic addition with a specific aldehyde followed by Mitsunobu ring-closure to finalize the benzopyrone structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this patented methodology offers profound advantages that extend beyond mere chemical yield. The shift away from scarce and hazardous reagents like LDA towards commodity chemicals such as trifluoronitrobenzene and nitromethane drastically simplifies raw material sourcing. This transition mitigates the risk of supply disruptions and stabilizes input costs, which is a primary concern for any procurement manager overseeing the budget for high-purity electronic chemicals. Furthermore, the milder reaction conditions reduce the energy burden on manufacturing facilities, contributing to a more sustainable and cost-effective production model.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of earth-abundant iron catalysts significantly lowers the direct material costs associated with production. Additionally, the high overall yield observed in the patent examples, with individual steps reaching up to 90% to 95%, means less raw material is wasted per unit of product. This efficiency translates into substantial cost savings without the need for complex recycling loops, making the process economically superior to legacy methods that suffer from yields as low as 16%.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials and avoiding reagents that require special handling or import licenses, the supply chain becomes more resilient. The robustness of the reaction conditions, which tolerate a broader range of operational parameters compared to cryogenic lithiation, ensures consistent batch-to-batch quality. This reliability is essential for reducing lead time for high-purity liquid crystal intermediates, allowing manufacturers to meet tight delivery schedules for downstream display panel producers.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard solvents like ethanol and water which are easier to recover and recycle than exotic organic solvents. The avoidance of heavy metal waste streams, thanks to the iron catalyst, simplifies wastewater treatment and ensures compliance with stringent environmental regulations. This green chemistry approach not only reduces disposal costs but also aligns with the corporate sustainability goals of major electronics manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzopyrone synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of what partners can expect when integrating this route into their production lines.
Q: What are the key advantages of this benzopyrone synthesis method over conventional routes?
A: Unlike conventional routes that rely on harsh lithium reagents and yield only about 16% to 40%, this patented method utilizes widely available raw materials like trifluoronitrobenzene and achieves significantly higher overall yields with milder reaction conditions.
Q: How is high purity ensured in the final liquid crystal intermediate?
A: The process incorporates specific purification steps such as washing with petroleum ether and freeze-washing with ethanol, resulting in target products with GC purity reaching up to 99.9% as demonstrated in the patent examples.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the method avoids extremely low-temperature cryogenic conditions typical of LDA chemistry and uses standard solvents and catalysts, making it highly suitable for commercial scale-up in electronic chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzopyrone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance of final electronic devices. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of benzopyrone derivative meets the exacting standards required for liquid crystal applications. Our infrastructure is designed to handle complex organic syntheses with the precision and safety necessary for the electronics sector.
We invite you to collaborate with us to leverage this advanced technology for your specific product needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how this innovative synthesis method can enhance your supply chain efficiency and product quality.
