Advanced Synthetic Route for Dezocine Impurity 3 Ensuring High Purity and Commercial Scalability
Advanced Synthetic Route for Dezocine Impurity 3 Ensuring High Purity and Commercial Scalability
The pharmaceutical industry continuously demands rigorous control over impurity profiles to ensure patient safety, particularly for potent opioid analgesics like dezocine. Patent CN111170886A discloses a groundbreaking preparation method for a specific oxidative degradation impurity of dezocine, identified as (5R,11S)-3-hydroxy-5-methyl-5,6,7,8,9,10,11,12-octahydro-5,11-methylenebenzocyclodecene-13-imine. This technical breakthrough addresses a critical gap in the market where standard reference substances were previously unavailable or difficult to synthesize due to the instability of the unsubstituted imine moiety. By establishing a robust, two-step synthetic pathway involving selective oxidation followed by mild demethylation, this innovation provides a reliable source for high-purity reference standards essential for regulatory compliance and quality control in API manufacturing. For a reliable pharmaceutical intermediate supplier, mastering such complex transformations is key to supporting global drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of related benzocyclodecene derivatives often relied on harsh conditions that compromised product integrity and operational safety. Prior art, such as US 39397666, utilized Raney nickel-catalyzed hydrogenation to generate imine intermediates, a process fraught with difficulties including the risk of over-reduction to amines and the inherent dangers of handling pyrophoric catalysts under pressure. Furthermore, traditional demethylation strategies frequently employed strong Lewis acids like boron tribromide at cryogenic temperatures (e.g., -78°C), which unfortunately promoted the hydrolysis of the sensitive imine bond into a ketone, drastically reducing yield and generating complex impurity profiles that are difficult to separate. These conventional limitations not only inflated production costs but also created significant bottlenecks in the supply chain for high-purity reference materials needed for clinical safety assessments.
The Novel Approach
In stark contrast, the methodology described in CN111170886A introduces a温和 (mild) yet highly efficient strategy that circumvents these historical pitfalls through careful reagent selection and condition optimization. The novel route employs N-halosuccinimides (such as N-chlorosuccinimide or N-bromosuccinimide) in the presence of organic bases to selectively oxidize the primary amine precursor to the desired imine without over-oxidation. Subsequently, the demethylation step utilizes aluminum chloride in conjunction with mercaptans at moderate temperatures (30-40°C), effectively cleaving the methyl ether while preserving the fragile imine functionality. This approach eliminates the need for cryogenic cooling and high-pressure equipment, thereby simplifying the operational workflow and enhancing the overall safety profile of the manufacturing process.
Mechanistic Insights into NXS-Mediated Oxidation and Lewis Acid Demethylation
The core of this synthetic achievement lies in the precise control of chemoselectivity during the oxidation phase. The reaction initiates with the interaction between the primary amine of the starting material (Formula 1) and the N-halosuccinimide oxidant, facilitated by a non-nucleophilic base like DBU or triethylamine. This environment promotes the formation of the nitrogen-halogen intermediate which subsequently eliminates to form the carbon-nitrogen double bond of the imine (Formula 2). Crucially, the reaction temperature is maintained between 45°C and 60°C, providing sufficient energy for conversion while preventing thermal degradation. The choice of solvent, typically 1,2-dichloroethane, ensures optimal solubility of the polycyclic substrate and stabilizes the transition states, allowing the reaction to proceed to completion with minimal formation of hydroxylamine or nitrosamine byproducts.
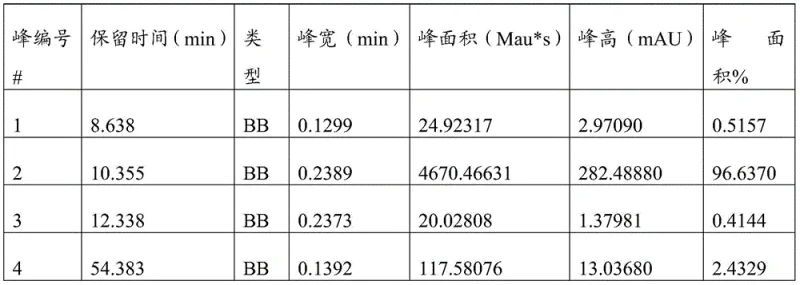
Following the oxidation, the demethylation mechanism leverages the soft Lewis acidity of aluminum chloride coordinated with a thiol scavenger. Unlike hard Lewis acids that aggressively attack the imine nitrogen leading to hydrolysis, the aluminum-thiolate complex specifically targets the oxygen atom of the methoxy group. This coordination weakens the carbon-oxygen bond, allowing for nucleophilic attack by the thiolate or chloride species to release the phenolic hydroxyl group. The presence of the mercaptan acts as a trap for the generated methyl group, driving the equilibrium forward and preventing reverse reactions. This delicate balance allows for the successful isolation of the target impurity (Formula 3) with exceptional purity levels, often exceeding 96.6% as confirmed by HPLC analysis, demonstrating the robustness of this mechanistic design for producing high-purity opioid analgesic intermediates.
How to Synthesize Dezocine Impurity 3 Efficiently
Implementing this synthesis requires strict adherence to the optimized stoichiometry and temperature profiles outlined in the patent embodiments to maximize yield and minimize side reactions. The process begins with the dissolution of the amine precursor in a chlorinated solvent, followed by the controlled addition of the oxidant and base, ensuring exotherms are managed effectively. After the oxidation is complete, the intermediate can be carried forward crude, eliminating time-consuming purification steps and reducing solvent waste. The subsequent demethylation requires careful addition of aluminum chloride and mercaptan under inert atmosphere to prevent moisture ingress, followed by a controlled quench with ammonia water to neutralize the acidic species without degrading the product. For detailed standardized operating procedures and specific safety protocols regarding reagent handling, please refer to the technical guide below.
- Oxidize (5R,11S,13S)-3-methoxy-5-methyl-5,6,7,8,9,10,11,12-octahydro-5,11-methylenebenzocyclodecen-13-amine using N-chlorosuccinimide or N-bromosuccinimide with a base like DBU at 45-60°C.
- Isolate the intermediate imine compound (Formula 2) via filtration and washing, proceeding directly to the next step without column chromatography.
- React the intermediate with aluminum chloride and a mercaptan (e.g., thiophenol) in dichloromethane at 30-40°C to effect demethylation, followed by aqueous quenching and extraction.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial strategic benefits for procurement managers and supply chain directors focused on cost reduction in API manufacturing and operational efficiency. By replacing hazardous high-pressure hydrogenation and cryogenic demethylation steps with ambient pressure and moderate temperature reactions, the process significantly lowers capital expenditure requirements for specialized reactor equipment and cooling infrastructure. The elimination of column chromatography for the intermediate further reduces solvent consumption and processing time, directly translating to lower variable costs per kilogram of produced material. Additionally, the use of commercially abundant reagents like N-halosuccinimides and aluminum chloride mitigates supply risks associated with exotic or regulated catalysts, ensuring a more resilient and continuous supply chain for critical reference standards.
- Cost Reduction in Manufacturing: The avoidance of expensive noble metal catalysts like Raney nickel and the removal of energy-intensive cryogenic cooling steps result in a drastically simplified cost structure. The ability to telescope the reaction steps without isolating the unstable intermediate via chromatography reduces labor hours and solvent disposal costs, offering substantial cost savings compared to traditional multi-step purifications.
- Enhanced Supply Chain Reliability: Utilizing widely available commodity chemicals such as N-bromosuccinimide and thiophenol ensures that raw material sourcing is not subject to the volatility often seen with specialized organometallic reagents. This stability in the supply base allows for more accurate forecasting and inventory planning, reducing lead time for high-purity pharmaceutical intermediates and preventing production delays due to material shortages.
- Scalability and Environmental Compliance: The mild reaction conditions and simplified workup procedures, involving basic filtration and extraction, make this process inherently easier to scale from laboratory benchtop to multi-ton commercial production. Furthermore, the reduction in hazardous waste generation, particularly the avoidance of heavy metal residues and large volumes of cryogenic solvents, aligns with increasingly stringent environmental regulations and sustainability goals within the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the scalability, purity, and specific reaction conditions of this novel dezocine impurity synthesis. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this route for their own reference standard production or quality control laboratories. The answers provided are derived directly from the experimental data and comparative examples presented in the patent documentation to ensure accuracy and reliability.
Q: Why is Aluminum Chloride preferred over Boron Tribromide for this demethylation?
A: Aluminum chloride, when used with mercaptans, prevents the hydrolysis of the sensitive imine structure that typically occurs with strong Lewis acids like boron tribromide, resulting in significantly higher yields (up to 72.4% vs 53.8%) and purity (96.6% vs 64.4%).
Q: Is this synthesis method suitable for large-scale manufacturing?
A: Yes, the process utilizes mild reaction temperatures (30-60°C), avoids high-pressure hydrogenation, and employs readily available reagents, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the typical purity achieved with this novel route?
A: The optimized protocol consistently delivers the target dezocine impurity with an HPLC purity exceeding 96.6%, meeting the stringent requirements for qualitative and quantitative analysis in drug safety testing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dezocine Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having access to well-characterized impurities for ensuring the safety and efficacy of opioid analgesics like dezocine. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. We operate stringent purity specifications and utilize rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of dezocine impurity meets the highest industry standards for identity and assay, providing you with the confidence needed for regulatory submissions.
We invite you to collaborate with us to optimize your supply chain for these critical reference materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our efficient synthesis route can reduce your overall procurement costs. Please contact us today to request specific COA data and route feasibility assessments, and let us support your commitment to medication safety and quality excellence.
