Advanced Synthesis of Dezocine Oxidative Impurities for Global Pharmaceutical Quality Assurance
Advanced Synthesis of Dezocine Oxidative Impurities for Global Pharmaceutical Quality Assurance
The pharmaceutical industry faces continuous challenges in ensuring the safety and efficacy of potent analgesics like dezocine, a mu and kappa opioid receptor agonist widely used for postoperative and cancer pain management. As regulatory bodies demand stricter control over degradation products, the ability to synthesize specific impurity reference standards becomes a critical bottleneck in drug development and quality control. Patent CN111170886B addresses this critical gap by disclosing a robust preparation method for a specific oxidative degradation impurity of dezocine, known chemically as (5R,11S)-3-hydroxy-5-methyl-5,6,7,8,9,10,11,12-octahydro-5,11-methylenebenzocyclodecene-13-imine. This compound, often referred to as Impurity 3 in technical literature, possesses a phenolic hydroxyl group and a primary amino group in the parent structure, making it susceptible to oxidation during storage and formulation. The ability to reliably produce this specific degradation product allows manufacturers to establish precise quantitative limits, thereby enhancing patient safety and meeting stringent ICH guidelines for impurity profiling.
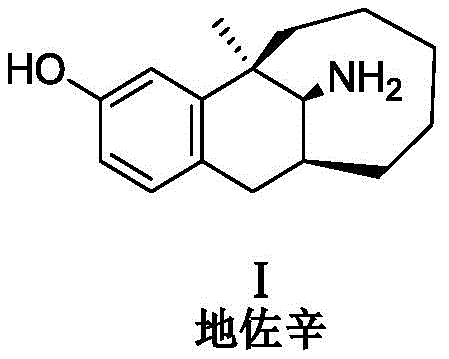
Understanding the structural vulnerability of dezocine is paramount for any reliable pharmaceutical intermediate supplier aiming to support global compliance. The parent molecule contains functional groups that are inherently reactive, particularly under oxidative stress conditions encountered during long-term storage or exposure to light. The impurity generated through this oxidative pathway retains the complex bridged ring system of the parent drug but alters the electronic properties of the A-ring and the nitrogen functionality. By mastering the synthesis of this specific degradant, chemical manufacturers provide essential tools for analytical laboratories to validate their HPLC methods. This ensures that every batch of dezocine injection released to the market is free from harmful levels of oxidative byproducts, maintaining the therapeutic index and minimizing potential adverse effects associated with unknown impurities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of related benzomorphan derivatives and imine intermediates has relied on harsh and hazardous methodologies that are ill-suited for modern GMP environments. One prominent prior art method, disclosed in US 39397666, utilizes a hydrogenation reduction strategy catalyzed by Raney nickel to convert oxime precursors into the desired imine or amine structures. However, this approach presents significant operational risks and selectivity challenges. The use of Raney nickel requires high-pressure hydrogenation equipment, introducing severe safety hazards related to flammability and explosion risks in a production setting. Furthermore, the catalytic activity of nickel is difficult to control precisely; it frequently leads to over-reduction, where the delicate imine double bond is fully reduced to a saturated amine, destroying the target molecular architecture. Additionally, alternative demethylation strategies often employ aggressive Lewis acids like boron tribromide (BBr3) at cryogenic temperatures.
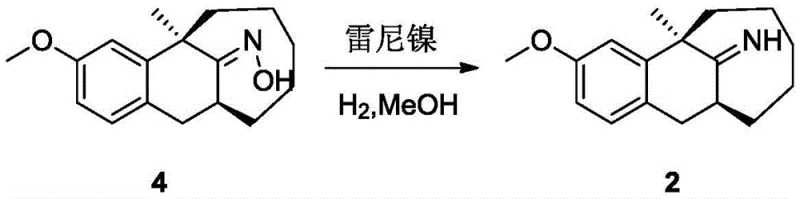
The reliance on boron tribromide for cleaving methyl ethers in sensitive substrates creates further complications for process scalability. As noted in comparative studies within the patent data, the use of BBr3 on imine-containing substrates often triggers unwanted hydrolysis. The strong Lewis acidity activates the imine nitrogen, making it susceptible to nucleophilic attack by water during the quench phase, converting the valuable imine into a ketone byproduct. This side reaction drastically reduces the overall yield, with comparative examples showing two-step yields dropping to as low as 53.8% and HPLC purity plummeting to roughly 64.4%. Such low purity profiles necessitate extensive and costly purification steps, such as preparative chromatography, which are economically unviable for large-scale production of reference standards. These technical barriers have historically limited the availability of high-quality dezocine impurities in the global market.
The Novel Approach
The methodology outlined in CN111170886B represents a paradigm shift towards milder, more selective chemistry that prioritizes atom economy and operational safety. Instead of relying on high-pressure hydrogenation or cryogenic Lewis acid treatments, this novel route employs a two-step sequence starting from a stable amine precursor. The first step involves a controlled oxidation using N-halosuccinimides (such as N-chlorosuccinimide or N-bromosuccinimide) in the presence of a non-nucleophilic base like DBU. This reagent combination allows for the gentle conversion of the primary amine to the corresponding imine without the risk of over-oxidation to nitro or hydroxylamine species. The reaction proceeds efficiently at moderate temperatures between 45°C and 60°C, eliminating the need for energy-intensive cooling or heating systems. This mild oxidative protocol preserves the integrity of the sensitive bridged ring system while establishing the crucial imine functionality required for the final impurity structure.
Following the oxidation, the second step utilizes a sophisticated demethylation strategy that circumvents the hydrolysis issues associated with traditional Lewis acids. By combining aluminum chloride with a mercaptan scavenger, such as thiophenol or ethanethiol, the process achieves selective cleavage of the methyl ether bond. The mercaptan acts as a soft nucleophile that traps the methyl group, preventing the aluminum species from attacking the imine nitrogen. This synergistic effect allows the reaction to proceed at ambient to slightly elevated temperatures (30-40°C) in common solvents like dichloromethane. The result is a dramatic improvement in both yield and purity, with experimental data demonstrating two-step molar yields exceeding 70% and HPLC purity levels reaching 96.6%. This high level of chemical fidelity ensures that the resulting material is suitable for direct use as a certified reference standard without further chromatographic purification.
Mechanistic Insights into Selective Oxidation and Lewis Acid Catalyzed Demethylation
The success of this synthetic route lies in the precise mechanistic control exerted during the oxidation phase. When using N-halosuccinimides (NXS) in the presence of a base like DBU, the reaction likely proceeds through the formation of an N-haloamine intermediate. The base facilitates the elimination of the halogen and a proton from the alpha-carbon, generating the carbon-nitrogen double bond characteristic of the imine. Unlike metal-catalyzed oxidations which can be indiscriminate, this organic oxidation pathway is highly specific to the primary amine functionality. The steric environment of the bridged ring system protects other vulnerable sites from oxidation, ensuring that the phenolic ether remains intact during this stage. This selectivity is crucial because premature oxidation of the aromatic ring would lead to quinone-like byproducts that are difficult to separate. The use of DBU, a strong non-nucleophilic base, ensures that the elimination step is rapid and irreversible, driving the equilibrium towards the desired imine product (Formula 2) while minimizing the formation of chlorinated or brominated side products on the aromatic ring.
In the subsequent demethylation step, the interaction between aluminum chloride and the mercaptan creates a unique reactive species that targets the ether linkage specifically. Aluminum chloride coordinates with the oxygen atom of the methoxy group, weakening the carbon-oxygen bond. Simultaneously, the sulfur atom of the mercaptan acts as a potent nucleophile, attacking the methyl group to form a sulfonium intermediate which eventually yields the demethylated phenol and a methyl sulfide byproduct. The presence of the mercaptan is the key differentiator here; it effectively competes with the imine nitrogen for coordination with the aluminum species. By saturating the Lewis acid with the sulfur scavenger, the electrophilicity of the aluminum towards the imine nitrogen is suppressed. This prevents the hydrolysis pathway that plagues conventional BBr3 methods. Consequently, the imine double bond remains untouched, and the phenolic hydroxyl group is revealed with high fidelity. This mechanistic nuance allows for the isolation of the target hydroxy-imine (Formula 3) in high purity, making it an ideal candidate for analytical calibration.
How to Synthesize (5R,11S)-3-hydroxy-5-methyl-5,6,7,8,9,10,11,12-octahydro-5,11-methylenebenzocyclodecene-13-imine Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of scale-up, making it accessible for both laboratory research and pilot plant operations. The process begins with the dissolution of the methoxy-amine starting material in a chlorinated solvent, followed by the controlled addition of the oxidant. Maintaining the temperature within the specified 45-60°C range is critical to ensure complete conversion without degradation. Once the oxidation is complete, the crude intermediate can be carried forward directly without isolation, streamlining the workflow and reducing material loss. The second stage involves the careful addition of aluminum chloride and the chosen thiol scavenger. Quenching the reaction with aqueous ammonia at low temperatures ensures safe neutralization of the acidic species while precipitating aluminum salts for easy removal.
- Oxidize (5R,11S,13S)-3-methoxy-5-methyl-5,6,7,8,9,10,11,12-octahydro-5,11-methylenebenzocyclodecen-13-amine (Formula 1) using N-chlorosuccinimide or N-bromosuccinimide in the presence of a base like DBU at 45-60°C to form the methoxy-imine intermediate (Formula 2).
- React the crude Formula 2 intermediate with aluminum chloride and a mercaptan scavenger (e.g., thiophenol or ethanethiol) in dichloromethane or 1,2-dichloroethane at 30-40°C.
- Quench the reaction with ammonia water, extract with organic solvent, and purify via slurry with n-hexane to obtain the target hydroxy-imine impurity with >96% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from high-pressure hydrogenation to this ambient pressure oxidative route offers substantial strategic benefits. The elimination of Raney nickel and high-pressure reactors removes a significant category of operational risk and capital expenditure. Facilities no longer need to maintain specialized hydrogenation suites or manage the disposal of pyrophoric nickel catalysts, which are classified as hazardous waste. This simplification of the infrastructure requirements translates directly into lower overhead costs and faster turnaround times for production batches. Furthermore, the reagents utilized in this novel method, such as N-halosuccinimides and aluminum chloride, are commodity chemicals available from multiple global suppliers. This diversifies the supply chain, reducing the risk of bottlenecks associated with single-source specialty catalysts. The robustness of the process also means that batch-to-batch variability is minimized, ensuring a consistent supply of high-purity material for downstream analytical applications.
- Cost Reduction in Manufacturing: The avoidance of cryogenic conditions and expensive boron tribromide reagents significantly lowers the utility and raw material costs per kilogram. By operating at near-ambient temperatures, the process reduces energy consumption for heating and cooling, contributing to a smaller carbon footprint and lower operational expenses. The high yield and purity achieved eliminate the need for costly preparative HPLC purification, allowing for simpler crystallization or slurry techniques that are far more economical at scale.
- Enhanced Supply Chain Reliability: The use of stable, shelf-stable reagents ensures that production can be scheduled with greater certainty, unaffected by the logistical challenges of transporting hazardous gases or pyrophoric solids. The simplified workup procedure, which involves standard aqueous extractions and filtration, reduces the processing time per batch. This efficiency allows manufacturers to respond more rapidly to urgent requests for reference standards from regulatory bodies or quality control laboratories, shortening the overall lead time for critical pharmaceutical testing materials.
- Scalability and Environmental Compliance: The process generates less hazardous waste compared to metal-catalyzed routes, as there are no heavy metal residues to remove from the final product or wastewater streams. The solvents used, primarily dichloromethane or dichloroethane, can be efficiently recovered and recycled using standard distillation units. This alignment with green chemistry principles facilitates easier regulatory approval for manufacturing sites and supports the sustainability goals of modern pharmaceutical companies seeking eco-friendly supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of dezocine impurities. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on why this specific synthetic route is superior for generating reference standards. Understanding these nuances helps stakeholders make informed decisions about sourcing and quality specifications.
Q: Why is the synthesis of dezocine oxidative impurities technically challenging?
A: The target impurity contains a rare unsubstituted imine structure which is highly unstable. Conventional strong Lewis acids like boron tribromide used for demethylation often cause hydrolysis of the imine into a ketone, leading to low yields and poor purity profiles unsuitable for reference standards.
Q: How does the new method improve upon prior art Raney Nickel reduction routes?
A: Prior art methods utilizing Raney Nickel hydrogenation require high-pressure equipment and suffer from over-reduction, where the imine is inadvertently reduced to an amine. The new ambient pressure oxidation route eliminates these safety hazards and selectivity issues.
Q: What represent the key commercial advantages of this synthesis route?
A: The process utilizes readily available reagents like N-halosuccinimides and aluminum chloride under mild thermal conditions. This avoids the need for specialized high-pressure reactors and complex cryogenic workups, significantly simplifying supply chain logistics and reducing operational costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dezocine Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize that the availability of high-purity impurity standards is the cornerstone of drug safety and regulatory compliance. Our technical team has extensively analyzed the pathways described in CN111170886B and integrated these advanced oxidative and demethylation techniques into our own process development pipeline. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need milligrams for method validation or kilograms for stability studies, we can deliver. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of dezocine impurity we supply meets the exacting standards required by global pharmacopoeias.
We invite pharmaceutical quality assurance teams and procurement specialists to collaborate with us on their impurity profiling needs. By leveraging our optimized synthesis routes, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure your supply chain with reliable, high-quality chemical intermediates that support the safety and efficacy of life-saving medications worldwide.
