Advanced Aziridine Synthesis via Ionic Liquid Catalysis for Commercial Scale-Up
Advanced Aziridine Synthesis via Ionic Liquid Catalysis for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for constructing complex heterocyclic scaffolds, particularly aziridine compounds which serve as vital intermediates in drug discovery. Patent CN1206217C introduces a groundbreaking methodology that utilizes room temperature ionic liquids as dual-function catalysts and reaction media for the synthesis of these valuable nitrogen-containing heterocycles. This innovation represents a significant departure from traditional synthetic routes that rely heavily on volatile organic solvents and expensive metal catalysts, addressing critical concerns regarding environmental impact and process safety. By leveraging the unique physicochemical properties of ionic liquids, such as negligible vapor pressure and high thermal stability, this process establishes a truly green chemical protocol that minimizes waste generation while maximizing atom economy. For R&D directors and process chemists, this technology offers a robust platform for generating high-purity aziridine derivatives with exceptional stereocontrol, paving the way for more reliable supply chains in the production of active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aziridine compounds has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up. Traditional methods often involve the ring-closing of 1,2-aminoalcohols or the use of expensive nitrene precursors like PhI=Ts in conjunction with metal complexes, which drastically inflates raw material costs and complicates downstream processing. Furthermore, conventional catalytic systems utilizing Lewis acids or transition metals frequently suffer from poor selectivity, leading to the formation of numerous by-products that require rigorous and costly purification steps to isolate the desired target molecule. The reliance on volatile organic solvents in these legacy processes not only poses serious safety risks due to flammability and toxicity but also creates substantial environmental liabilities associated with solvent disposal and emissions. These factors collectively contribute to extended lead times and reduced overall process efficiency, making it challenging for procurement teams to secure cost-effective supplies of high-quality aziridine intermediates for large-scale manufacturing operations.
The Novel Approach
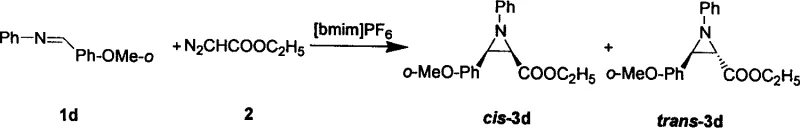
In stark contrast to these outdated methodologies, the novel approach detailed in the patent employs room temperature ionic liquids to facilitate the direct reaction between imine compounds and diazo compounds under mild conditions. This strategy eliminates the need for hazardous organic solvents during the reaction phase, thereby creating a safer working environment and significantly reducing the ecological footprint of the synthesis. The ionic liquid acts as a highly effective promoter that stabilizes the reactive intermediates, enabling the reaction to proceed smoothly at room temperature within a short timeframe of 1 to 5 hours. This simplification of reaction conditions translates directly into operational excellence, as it removes the energy-intensive requirements for heating or cooling that are typical of conventional protocols. Moreover, the system demonstrates remarkable versatility across a wide range of substrates, including various substituted anilines and alkylamines, ensuring that diverse chemical libraries can be accessed without the need for extensive method re-optimization.
Mechanistic Insights into Ionic Liquid-Promoted Aziridination
The core of this technological advancement lies in the unique ability of the ionic liquid to function simultaneously as a solvent and a catalyst, creating a specialized microenvironment that favors the formation of the aziridine ring. The cationic and anionic components of the ionic liquid, such as the 1-butyl-3-methylimidazolium cation and hexafluorophosphate anion, interact with the diazo compound to facilitate the generation of a metal-free carbene or carbenoid species in situ. This activated species then undergoes a highly stereoselective addition to the carbon-nitrogen double bond of the imine substrate, driven by the stabilizing effects of the ionic medium. The result is a reaction pathway that inherently favors the formation of the cis-isomer, with experimental data showing selectivity reaching up to 100 percent in many cases, which is a substantial improvement over the mixed isomer ratios typically observed with Lewis acid catalysts. This high degree of stereocontrol is critical for pharmaceutical applications where the biological activity of the final drug molecule is often dependent on its specific three-dimensional configuration.
Beyond mere catalytic activity, the ionic liquid system plays a pivotal role in impurity control and product isolation, which are key concerns for quality assurance teams. Because the ionic liquid is immiscible with common non-polar extraction solvents like petroleum ether, the product can be easily separated from the reaction mixture through simple liquid-liquid extraction, leaving the catalyst behind in the ionic phase. This physical separation mechanism prevents the contamination of the final product with residual catalyst metals or ligands, a common issue in transition-metal catalyzed reactions that often requires additional scavenging steps. The ease of separation ensures that the resulting aziridine compounds meet stringent purity specifications required for regulatory compliance in drug manufacturing. Additionally, the stability of the ionic liquid allows it to be recovered and reused multiple times without significant degradation in performance, further enhancing the robustness of the process and ensuring consistent batch-to-batch quality.
How to Synthesize Aziridine Compounds Efficiently
The implementation of this ionic liquid-mediated synthesis route offers a streamlined protocol that is readily adaptable for both laboratory-scale optimization and pilot-plant production. The general procedure involves mixing the imine substrate and the diazo compound in a stoichiometric ratio within the ionic liquid medium, followed by stirring at ambient temperature until conversion is complete. This straightforward operational sequence reduces the complexity of the manufacturing process, minimizing the potential for human error and equipment failure during scale-up. For detailed standard operating procedures and specific parameter optimizations regarding different substrate classes, please refer to the comprehensive guide provided below which outlines the precise steps for execution.
- Prepare the reaction mixture by combining the imine substrate and diazo compound (such as ethyl diazoacetate) in a molar ratio of 1: 1.
- Add the reactants to a room temperature ionic liquid, such as [Bmim]PF6, which serves as both the catalyst and the reaction medium.
- Stir the mixture at room temperature for 1 to 5 hours, then separate the product via extraction with petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic liquid technology presents a compelling value proposition centered around cost reduction and supply reliability. By eliminating the need for expensive transition metal catalysts and precious metal ligands, the raw material costs associated with the synthesis are drastically lowered, allowing for more competitive pricing structures in the final supply contracts. The removal of volatile organic solvents from the reaction step also leads to significant savings in waste disposal fees and regulatory compliance costs, as the process generates less hazardous waste that requires special handling. Furthermore, the ability to recycle the ionic liquid catalyst multiple times without loss of activity means that the effective cost per kilogram of the catalyst approaches zero over the lifecycle of the production campaign, providing a substantial long-term economic advantage over single-use catalytic systems.
- Cost Reduction in Manufacturing: The elimination of expensive metal catalysts and the reduction in solvent consumption directly translate to lower variable costs per unit of production. Since the ionic liquid serves as both the reaction medium and the catalyst, there is no need to purchase separate solvent grades or invest in complex catalyst recovery infrastructure. This simplification of the bill of materials allows manufacturers to offer more aggressive pricing to their clients while maintaining healthy profit margins. Additionally, the high selectivity of the reaction reduces the loss of valuable starting materials to by-products, improving the overall yield and further driving down the cost of goods sold for these critical pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the ionic liquid system ensures consistent production output regardless of minor fluctuations in ambient conditions, which is vital for maintaining steady supply flows to downstream customers. Because the catalyst can be reused extensively, the supply chain is less vulnerable to disruptions caused by shortages of specialized catalytic reagents or fluctuations in the market price of precious metals. The simplified workup procedure also shortens the overall cycle time for each batch, enabling manufacturers to respond more quickly to urgent orders and reduce lead times for high-purity aziridine compounds. This agility is a key differentiator in a market where speed to market for new drug candidates is increasingly critical.
- Scalability and Environmental Compliance: The green nature of this process aligns perfectly with modern environmental regulations and corporate sustainability goals, reducing the risk of future regulatory shutdowns or fines. The non-volatile nature of the ionic liquid minimizes atmospheric emissions, making it easier to obtain necessary environmental permits for large-scale production facilities. As production scales from kilograms to tons, the safety benefits of operating without flammable organic solvents become even more pronounced, lowering insurance premiums and improving the overall safety profile of the manufacturing site. This forward-looking approach ensures that the supply of these intermediates remains secure and compliant with evolving global standards for green chemistry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details helps stakeholders make informed decisions about integrating this method into their existing supply chains.
Q: What are the advantages of using ionic liquids over traditional organic solvents for aziridine synthesis?
A: Ionic liquids eliminate the need for volatile organic solvents, significantly reducing environmental pollution and safety hazards. Furthermore, they act as both catalyst and medium, simplifying the workup process and allowing for catalyst reuse without loss of activity.
Q: How does this method improve stereoselectivity compared to metal-catalyzed routes?
A: The ionic liquid environment promotes high cis-selectivity, often achieving up to 100% cis-isomer formation, whereas traditional metal complex or Lewis acid catalysis often results in complex mixtures of by-products that are difficult to purify.
Q: Can the ionic liquid catalyst be recycled for industrial production?
A: Yes, the patent data demonstrates that the ionic liquid can be recovered and reused multiple times (at least five cycles shown) while maintaining consistent catalytic activity and yield, which is crucial for cost-effective large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aziridine Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this ionic liquid catalysis technology for the production of high-value aziridine intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of aziridine compound meets the exacting standards required for pharmaceutical applications. We are committed to leveraging our technical expertise to optimize this green synthesis route for your specific needs, delivering superior quality and consistency.
We invite you to contact our technical procurement team to discuss how we can support your upcoming projects with a Customized Cost-Saving Analysis tailored to your volume requirements. By partnering with us, you gain access to specific COA data and route feasibility assessments that demonstrate the viability of this advanced manufacturing method. Let us help you secure a stable, cost-effective, and environmentally responsible supply of these critical building blocks for your drug development pipeline.
