Advanced Electrochemical Catalysis for Scalable Aziridine Compound Manufacturing
Advanced Electrochemical Catalysis for Scalable Aziridine Compound Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, cost-effective methodologies for constructing strained nitrogen heterocycles, particularly aziridines, which serve as pivotal building blocks for bioactive molecules. A groundbreaking approach detailed in patent CN103436911A introduces a novel electrochemical catalytic synthesis method that fundamentally redefines the production landscape for these valuable intermediates. Unlike traditional chemical oxidation methods that often rely on stoichiometric amounts of hazardous oxidants or expensive transition metal catalysts, this invention leverages electrons as the primary oxidant in a streamlined single-chamber setup. By shifting from complex dual-chamber systems to a simplified undivided cell architecture and replacing costly platinum anodes with accessible glassy carbon electrodes, this technology offers a compelling value proposition for manufacturers aiming to optimize their supply chains. The process operates under mild conditions, typically between 0°C and 40°C, utilizing common halide salts as electrocatalysts to facilitate the indirect electrolysis of N-aminophthalimide and various olefinic substrates. This strategic pivot not only enhances operational safety by eliminating strong chemical oxidants but also drastically simplifies the engineering requirements for reactor design, making it an ideal candidate for modern, green chemistry initiatives in API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those reported by Yudin et al., have historically relied on sophisticated and capital-intensive electrochemical setups that pose significant barriers to industrial adoption. These conventional processes typically necessitate the use of a two-chamber electrolytic cell separated by a membrane or diaphragm, a configuration that inherently increases the internal resistance of the system and consequently elevates the energy consumption required to drive the reaction. Furthermore, the reliance on platinized platinum as the working electrode introduces a substantial material cost burden, as platinum is a precious metal with volatile market pricing and limited availability. From an operational perspective, these older methods often employ potentiostatic electrolysis, which requires precise potential control via expensive three-electrode systems and specialized electrochemical workstations. This level of complexity not only inflates the initial capital investment but also complicates process control and maintenance, rendering such techniques less suitable for the rigorous demands of large-scale commercial production where simplicity and reliability are paramount. Additionally, the limited lifespan of the barrier films in divided cells necessitates periodic replacement, adding to the operational downtime and recurring maintenance costs that erode profit margins in high-volume manufacturing environments.
The Novel Approach
The innovative method described in the patent data overcomes these historical bottlenecks by implementing a direct, constant-current electrolysis protocol within a simple single-chamber cell. This architectural simplification eliminates the need for ion-exchange membranes, thereby reducing the cell's internal resistance and allowing for operation at lower decomposition voltages, which translates directly into significant energy savings. Crucially, the substitution of the expensive platinum anode with a glassy carbon electrode represents a major cost reduction strategy, as glassy carbon is not only far more affordable but also exhibits excellent stability and conductivity in the specified electrolytic media. The transition to constant current electrolysis further democratizes the technology, as it removes the dependency on sophisticated potentiostats, allowing the process to be run with standard, industrial-grade rectifiers that are commonplace in electrochemical plants. This approach utilizes inexpensive tetraalkylammonium halides or alkali metal halides as redox mediators to facilitate the indirect oxidation of the nitrogen source, ensuring high selectivity while maintaining a gentle reaction environment. By integrating these engineering and chemical improvements, the new method achieves a level of operational simplicity and economic efficiency that was previously unattainable, paving the way for the widespread commercialization of electrochemically synthesized aziridines.
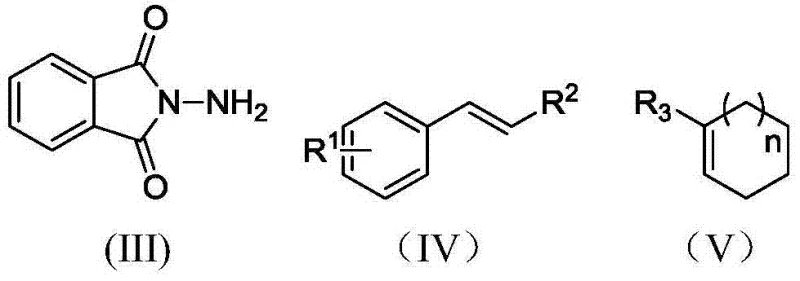
The versatility of this electrochemical platform is further demonstrated by its broad substrate scope, accommodating both linear styrenes and cyclic olefins with high efficacy. As illustrated in the reaction schemes, the method successfully converts N-aminophthalimide and various substituted styrenes into the corresponding N-phthalimido aziridines, which are versatile intermediates for ring-opening reactions to generate beta-amino acids and other pharmacophores. The ability to tolerate diverse functional groups on the aromatic ring, including electron-donating methyl groups and electron-withdrawing halogens like fluorine, chlorine, and bromine, underscores the robustness of the catalytic system. Moreover, the extension of this methodology to cycloolefins allows for the synthesis of fused bicyclic aziridine structures, expanding the chemical space accessible to medicinal chemists. This adaptability ensures that the process can be tailored to produce a wide array of high-purity pharmaceutical intermediates, meeting the stringent quality requirements of downstream drug synthesis without the need for extensive process redevelopment for each new substrate.
Mechanistic Insights into Indirect Electrochemical Aziridination
The core of this technological advancement lies in the mechanism of indirect electrochemical oxidation mediated by halide ions, which serves as a highly efficient electron transfer shuttle between the anode surface and the organic substrates. In this catalytic cycle, the halide mediator (such as iodide from tetrabutylammonium iodide) is initially oxidized at the glassy carbon anode to generate a reactive halogen species or radical cation. This electro-generated active species then reacts with the N-aminophthalimide in the bulk solution to form a reactive nitrogen-centered radical or cation intermediate, effectively activating the nitrogen source for subsequent cycloaddition. This indirect pathway is critical because it prevents the direct oxidation of the organic substrates at the electrode surface, which could lead to polymerization, over-oxidation, or electrode fouling, thereby preserving the integrity of the electrode and enhancing the overall Faradaic efficiency of the process. The presence of a base, such as potassium carbonate or 2,6-dichloropyridine, plays a vital role in neutralizing the protons generated during the reaction, driving the equilibrium forward and preventing the acid-catalyzed degradation of the sensitive aziridine ring. By carefully tuning the current density between 4 mA/cm² and 12 mA/cm² and controlling the total charge passed to approximately 3 F/mol, the process ensures complete conversion of the limiting reagent while minimizing side reactions, resulting in a clean impurity profile that simplifies downstream purification.
Impurity control in this electrochemical system is inherently superior to many thermal chemical methods due to the mild reaction temperatures and the specificity of the electro-generated oxidant. Operating at ambient or near-ambient temperatures (0-40°C) suppresses thermal decomposition pathways and minimizes the formation of polymeric byproducts that often plague radical reactions conducted at elevated temperatures. The use of a single-chamber cell without a separating membrane also eliminates the risk of cross-contamination between anolyte and catholyte compartments, which can sometimes lead to complex mixture formation in divided cells. Furthermore, the choice of supporting electrolyte, such as lithium perchlorate or triethylamine/acetic acid buffers, helps maintain a stable ionic environment that supports consistent current flow and prevents localized pH spikes that could degrade the product. The result is a reaction mixture where the desired aziridine product is the dominant species, allowing for straightforward isolation via solvent evaporation and standard chromatographic techniques. This high level of selectivity is essential for pharmaceutical applications, where strict limits on genotoxic impurities and heavy metals must be adhered to, and the absence of transition metal catalysts in this electrochemical route inherently satisfies the demand for metal-free synthetic processes.
How to Synthesize N-Phthalimido Aziridines Efficiently
Implementing this electrochemical synthesis route requires careful attention to the preparation of the electrolytic medium and the precise control of electrical parameters to ensure reproducible high yields. The process begins with the dissolution of the starting materials, specifically N-aminophthalimide and the chosen olefinic substrate, in a suitable solvent system such as 2,2,2-trifluoroethanol or acetonitrile, which provides the necessary solubility and conductivity. A catalytic amount of the halide mediator, typically tetrabutylammonium iodide, is added along with a stoichiometric base to scavenge protons, creating a homogeneous reaction mixture ready for electrolysis. The detailed standardized synthesis steps, including specific molar ratios, stirring rates, and workup procedures, are outlined in the guide below to assist process chemists in replicating the patented results with high fidelity.
- Prepare the electrolytic solution by dissolving N-aminophthalimide and the alkene substrate (styrene or cycloolefin) in trifluoroethanol or acetonitrile with a supporting electrolyte like lithium perchlorate.
- Add a catalytic amount of tetraalkylamine halide (e.g., tetrabutylammonium iodide) and a base such as potassium carbonate to the single-chamber cell equipped with a glassy carbon anode.
- Perform constant current electrolysis at 4-12 mA/cm² until 2.5-3.5 F/mol of electricity is passed, then isolate the product via solvent removal and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical methodology presents a transformative opportunity to reduce manufacturing costs and enhance supply security for critical aziridine intermediates. The shift away from precious metal catalysts and complex divided-cell hardware directly addresses the volatility of raw material costs and the high capital expenditure associated with traditional electrochemical setups. By simplifying the reactor design to a single-chamber configuration using commodity-grade glassy carbon electrodes, manufacturers can significantly lower the barrier to entry for producing these high-value compounds, fostering a more competitive and resilient supply market. The reliance on electricity as the primary reagent also decouples production from the supply chains of hazardous chemical oxidants, reducing logistical risks and storage costs associated with dangerous goods. This operational agility allows suppliers to respond more rapidly to fluctuations in demand, ensuring consistent availability of key intermediates for downstream pharmaceutical clients without the delays often caused by the procurement of specialized reagents.
- Cost Reduction in Manufacturing: The elimination of expensive platinized platinum electrodes in favor of glassy carbon anodes results in a drastic reduction in equipment material costs, while the removal of ion-exchange membranes further decreases consumable expenses. Additionally, the lower internal resistance of the single-chamber cell reduces the voltage required for electrolysis, leading to substantial savings in electrical energy consumption per kilogram of product produced. The avoidance of stoichiometric chemical oxidants and transition metal catalysts also removes the costs associated with purchasing, handling, and disposing of these reagents, contributing to a leaner and more cost-effective production model. These cumulative savings allow for a more competitive pricing structure for the final aziridine intermediates, enhancing the margin potential for both the manufacturer and the end-user in the pharmaceutical value chain.
- Enhanced Supply Chain Reliability: The reagents required for this process, including tetraalkylammonium halides, alkali metal halides, and common organic solvents, are widely available industrial commodities with stable global supply chains. This contrasts sharply with methods relying on specialized ligands or rare earth catalysts that may be subject to geopolitical supply disruptions or long lead times. The simplicity of the equipment, which can be constructed from standard laboratory glassware or scaled using conventional steel reactors lined with inert materials, ensures that production capacity can be easily expanded or replicated across different manufacturing sites without the need for custom-engineered electrochemical workstations. This flexibility strengthens the overall resilience of the supply network, minimizing the risk of production stoppages due to equipment failure or parts shortages.
- Scalability and Environmental Compliance: The use of constant current electrolysis is inherently easier to scale than potentiostatic methods, as it does not require complex feedback loops or reference electrodes that can be difficult to maintain in large vessels. This facilitates a smoother transition from pilot-scale optimization to multi-ton commercial production, accelerating the time-to-market for new drug candidates requiring these intermediates. Furthermore, the process generates minimal waste, as the electrons serve as a clean oxidant and the halide mediator is recycled within the cycle, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The absence of heavy metal residues in the final product simplifies the purification process and reduces the environmental burden of wastewater treatment, making this a truly green manufacturing solution for the modern chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical aziridination technology, providing clarity on its operational benefits and compatibility with existing infrastructure. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, offering a factual basis for decision-making regarding process adoption. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating electrochemical steps into their current synthetic routes for API intermediates.
Q: What are the primary cost advantages of this electrochemical method over traditional platinum-based systems?
A: The method replaces expensive platinized platinum electrodes with cost-effective glassy carbon electrodes and eliminates the need for complex two-chamber cells, significantly reducing both equipment capital expenditure and operational energy costs due to lower internal resistance.
Q: How does the single-chamber design impact energy consumption during synthesis?
A: By utilizing a single-chamber electrolytic cell, the internal resistance is substantially reduced compared to divided cells, which lowers the required decomposition voltage and results in markedly decreased energy consumption per mole of product formed.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of constant current electrolysis rather than potentiostatic control simplifies the equipment requirements, removing the need for expensive electrochemical workstations and making the process highly amenable to industrial scale-up using standard rectifiers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aziridine Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this electrochemical synthesis technology in delivering high-quality aziridine intermediates to the global pharmaceutical market. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art facilities are equipped to handle electrochemical processes safely and efficiently, utilizing rigorous QC labs to verify stringent purity specifications for every batch. We are committed to leveraging our technical expertise to optimize reaction parameters, ensuring maximum yield and minimal impurity levels that meet the exacting standards of international regulatory bodies.
We invite prospective partners to engage with our technical procurement team to discuss how this innovative method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this electrochemical route for your supply chain. We encourage you to contact us today to obtain specific COA data for our electrochemically synthesized aziridines and to receive comprehensive route feasibility assessments that demonstrate our capability to support your long-term production goals with reliability and precision.
