Advanced Synthesis of 1-Hydroxycyclohexyl Phenyl Ketone for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high yield with stringent environmental compliance. Patent CN102267887A introduces a significant technological advancement in the production of 1-hydroxycyclohexyl phenyl ketone, a valuable intermediate for various bioactive molecules. Unlike conventional methods that rely on harsh sulfuring agents, this novel process utilizes cyclohexanecarboxylic acid as the primary starting material, undergoing a sequence of acylation, Friedel-Crafts reaction, and alkaline hydrolysis. The strategic replacement of thionyl chloride with phosphorus trichloride fundamentally alters the impurity profile, removing the generation of sulfur dioxide gas entirely. This shift not only enhances operator safety but also simplifies the downstream purification protocols, resulting in a crude product that is more amenable to refined distillation and crystallization. For R&D directors and process engineers, this represents a pivotal opportunity to optimize supply chains for high-purity pharmaceutical intermediates while adhering to increasingly strict global emission standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-hydroxycyclohexyl phenyl ketone has been plagued by significant environmental and operational inefficiencies associated with traditional halogenation techniques. The prior art predominantly relied on the use of thionyl chloride (SOCl2) for the initial activation of the carboxylic acid, a reaction that inevitably releases large volumes of toxic sulfur dioxide (SO2) gas. Managing these emissions requires expensive scrubbing infrastructure and poses severe regulatory hurdles for manufacturing facilities. Furthermore, the subsequent functionalization steps often involved bromination followed by hydrolysis, which generated substantial quantities of sodium bromide (NaBr) waste. This saline waste stream is difficult to treat and contributes heavily to the total dissolved solids (TDS) load in wastewater, driving up disposal costs and environmental liability. The cumulative effect of these factors is a process with lower overall yields and a prohibitively high ecological footprint, making it unsustainable for modern green chemistry initiatives.
The Novel Approach
The innovative pathway detailed in the patent data offers a transformative solution by restructuring the synthetic logic to prioritize atom economy and waste minimization. By employing phosphorus trichloride (PCl3) for the acylation step, the process generates phosphorous acid derivatives instead of sulfur gases, effectively neutralizing the risk of SO2 emissions at the source. The subsequent introduction of a chlorination-alkaline hydrolysis sequence replaces the problematic bromination step, yielding sodium chloride (NaCl) as the primary inorganic byproduct. Sodium chloride is far less environmentally burdensome than sodium bromide and can often be managed through standard effluent treatment protocols. This methodological shift results in a cleaner reaction profile with higher product yields, as the absence of sulfur contaminants reduces the formation of complex, hard-to-remove side products. Consequently, this approach provides a reliable pharma intermediate supplier with a distinct competitive advantage in terms of both cost-efficiency and regulatory compliance.
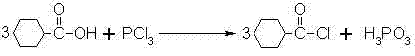
Mechanistic Insights into PCl3-Mediated Acylation and Friedel-Crafts Reaction
The core of this synthesis lies in the efficient activation of cyclohexanecarboxylic acid using phosphorus trichloride, a reaction that proceeds through a nucleophilic acyl substitution mechanism. In this step, the hydroxyl group of the carboxylic acid attacks the phosphorus atom of PCl3, displacing a chloride ion and forming an unstable mixed anhydride intermediate. This intermediate rapidly collapses to release the desired cyclohexanecarbonyl chloride along with phosphorous acid species. The reaction conditions are meticulously controlled, typically maintaining temperatures around 35°C to 60°C to ensure complete conversion while minimizing thermal degradation of the acid chloride. The stoichiometry is critical, with a weight ratio of acid to PCl3 optimized around 2:1 to ensure full consumption of the starting material without excessive reagent waste. This precise control over the activation step is fundamental to achieving the high purity required for downstream pharmaceutical applications.
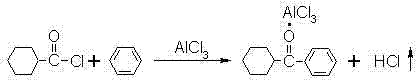
Following the formation of the acid chloride, the process advances to the Friedel-Crafts acylation, a cornerstone reaction for constructing the carbon-carbon bond between the cyclohexane ring and the phenyl group. In the presence of a Lewis acid catalyst such as aluminum chloride (AlCl3), the carbonyl carbon of the acid chloride becomes highly electrophilic, facilitating attack by the electron-rich benzene ring. The reaction forms a stable complex between the resulting ketone and the aluminum chloride, which must be carefully hydrolyzed in a subsequent step using dilute hydrochloric acid to liberate the free ketone. Temperature control during this exothermic process is paramount; maintaining the reaction mixture between 5°C and 15°C prevents poly-acylation and ensures the regioselectivity of the substitution. The careful management of the AlCl3 complex formation and its subsequent decomposition is what allows for the isolation of cyclohexyl phenyl ketone with minimal impurities, setting the stage for the final functionalization steps.
How to Synthesize 1-Hydroxycyclohexyl Phenyl Ketone Efficiently
The synthesis of this high-value intermediate requires a disciplined approach to reaction engineering, particularly regarding the sequential addition of reagents and temperature modulation. The process begins with the activation of the acid, followed immediately by the Friedel-Crafts coupling to minimize the storage time of the reactive acid chloride. Once the ketone skeleton is established, the alpha-position is functionalized via chlorination using chlorine gas at elevated temperatures, introducing a leaving group that is subsequently displaced by a hydroxyl ion. The detailed standardized synthesis steps, including specific molar ratios, stirring rates, and workup procedures, are outlined below to ensure reproducibility and safety in a pilot or production environment.
- Activate cyclohexanecarboxylic acid using phosphorus trichloride (PCl3) at controlled temperatures to form cyclohexanecarbonyl chloride.
- Perform Friedel-Crafts acylation with benzene and aluminum chloride catalyst to construct the cyclohexyl phenyl ketone skeleton.
- Execute alpha-chlorination followed by alkaline hydrolysis to substitute the chlorine atom with a hydroxyl group, yielding the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates directly into tangible operational benefits and risk mitigation. The elimination of sulfur dioxide emissions removes the need for specialized corrosion-resistant equipment and complex gas scrubbing systems, leading to significant capital expenditure savings and reduced maintenance overheads. Furthermore, the switch from bromine-based chemistry to chlorine-based chemistry leverages more abundant and cost-effective raw materials, stabilizing the supply chain against volatility in halogen markets. The generation of sodium chloride as a byproduct, rather than sodium bromide, drastically simplifies wastewater treatment requirements, allowing facilities to operate with lower environmental compliance costs and reduced risk of regulatory fines. These factors combine to create a manufacturing process that is not only chemically superior but also economically resilient in the face of tightening global environmental regulations.
- Cost Reduction in Manufacturing: The removal of thionyl chloride and bromine reagents eliminates the high costs associated with handling hazardous sulfur and bromine waste streams. By utilizing phosphorus trichloride and chlorine gas, the process relies on commodity chemicals that are generally cheaper and more readily available in bulk quantities. Additionally, the higher yield reported in the patent implies better raw material utilization, meaning less starting material is wasted per kilogram of finished product. This efficiency gain directly improves the gross margin for the manufacturer, allowing for more competitive pricing strategies in the global market without sacrificing quality or profitability.
- Enhanced Supply Chain Reliability: Dependence on specialized reagents like thionyl chloride can introduce bottlenecks, especially when environmental restrictions limit their production or transport. This new route utilizes widely available industrial chemicals, ensuring a steady and reliable flow of inputs even during market disruptions. The simplified waste profile also means that third-party waste disposal vendors are more willing and able to handle the effluent, reducing the risk of production stoppages due to waste tank capacity issues. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery expectations of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, as the reaction conditions (temperatures and pressures) are well within the range of standard industrial reactors. The absence of toxic sulfur gases makes it easier to obtain environmental permits for expansion or new facility construction. Moreover, the recycling of solvents like toluene and petroleum ether, as described in the refining step, further enhances the sustainability profile of the operation. This alignment with green chemistry principles positions the manufacturer favorably for partnerships with multinational corporations that have strict carbon footprint and sustainability mandates for their suppliers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and procedural details outlined in the patent documentation, providing clarity on how this method outperforms legacy processes. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: How does this new synthesis route improve environmental compliance compared to traditional methods?
A: The patented process replaces thionyl chloride (SOCl2) with phosphorus trichloride (PCl3), completely eliminating the emission of toxic sulfur dioxide (SO2) gas. Additionally, the final byproduct is sodium chloride (NaCl) rather than sodium bromide (NaBr), significantly reducing the burden on wastewater treatment systems and lowering hazardous waste disposal costs.
Q: What are the critical control points for ensuring high purity in the Friedel-Crafts step?
A: Precise temperature control between 5°C and 15°C is essential during the addition of cyclohexanecarbonyl chloride to the benzene-AlCl3 mixture to prevent poly-acylation and thermal degradation. Furthermore, the subsequent hydrolysis must be carefully managed with dilute hydrochloric acid to ensure complete decomposition of the aluminum complex without promoting side reactions, followed by rigorous distillation to remove unreacted benzene.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It utilizes common industrial reagents like chlorine gas and caustic soda, and incorporates efficient gas absorption systems for hydrogen chloride recovery. The elimination of corrosive sulfur gases simplifies reactor material requirements, making it highly viable for metric-ton scale manufacturing in standard chemical plants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Hydroxycyclohexyl Phenyl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity 1-hydroxycyclohexyl phenyl ketone that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging the efficiencies of the PCl3-based route, we can offer a product that is not only chemically superior but also produced with a significantly reduced environmental footprint.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. Whether you require a Customized Cost-Saving Analysis or need to review specific COA data and route feasibility assessments for your upcoming projects, we are ready to provide the data-driven insights you need. Contact us today to secure a reliable supply of this key intermediate and leverage our expertise in sustainable chemical manufacturing to drive your business forward.
