Advanced Green Synthesis of Photoinitiator 184 for Industrial Scale-Up
Advanced Green Synthesis of Photoinitiator 184 for Industrial Scale-Up
The global demand for high-performance UV curing materials continues to surge, driving the need for more efficient and environmentally sustainable manufacturing processes for key components like Photoinitiator 184. A significant technological breakthrough in this sector is detailed in patent CN108911959B, which outlines a novel preparation process for 1-hydroxycyclohexyl phenyl ketone. This invention fundamentally restructures the synthetic pathway, moving away from the traditional, hazard-laden four-step sequence toward a streamlined, two-stage green chemistry approach. By leveraging high-temperature catalytic ketonization and advanced phase transfer catalysis, this method addresses critical pain points regarding safety, waste generation, and operational complexity that have long plagued the industry. For R&D directors and procurement strategists, understanding this shift is vital for securing a competitive edge in the supply of high-purity photoinitiators.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Photoinitiator 184 has been burdened by a cumbersome four-step synthesis that relies heavily on hazardous reagents and generates substantial toxic waste. The traditional route begins with the acyl chlorination of cyclohexanecarboxylic acid using phosphorus trichloride, followed by a Friedel-Crafts acylation with benzene and aluminum trichloride. This is subsequently followed by a dangerous chlorination step using chlorine gas and a final alkaline hydrolysis. This legacy process creates severe environmental and safety liabilities, including the generation of high-phosphorus wastewater, aluminum-containing sludge, and corrosive hydrogen chloride gas. Furthermore, the reliance on benzene, a known carcinogen, and the handling of toxic chlorine gas impose rigorous safety protocols and high capital expenditures for waste treatment facilities, drastically inflating the cost of goods sold and complicating regulatory compliance for manufacturers.
The Novel Approach
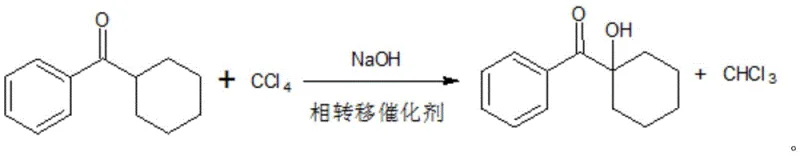
In stark contrast, the innovative process disclosed in the patent simplifies the synthesis into two distinct, highly efficient sections that eliminate the most problematic reagents. The first section involves a direct high-temperature ketone making step where benzoic acid and cyclohexanecarboxylic acid are liquefied, mixed, and passed over a metal salt catalyst at temperatures between 300°C and 500°C. This single step replaces both the acyl chlorination and Friedel-Crafts reactions, effectively removing the need for phosphorus trichloride, aluminum trichloride, and benzene solvent. The second section utilizes a one-pot alkali chloride hydrolysis method where the intermediate ketone reacts with carbon tetrachloride and sodium hydroxide in the presence of a phase transfer catalyst. This consolidation not only removes the need for gaseous chlorine but also streamlines the workflow, resulting in a safer, cleaner, and more economically viable production line for high-purity photoinitiator intermediates.

Mechanistic Insights into High-Temperature Catalytic Ketonization
The core of this technological advancement lies in the precise engineering of the catalytic system used for the direct ketonization of carboxylic acids. The patent specifies the preparation of a robust catalyst by impregnating zinc oxide (ZnO) and magnesium oxide (MgO) onto a carrier such as aluminum oxide, silicon dioxide, or titanium dioxide. Specifically, a weight ratio of ZnO to MgO of 9:1 is preferred, with the total metal salt loading on the carrier ranging from 5% to 20%. This catalyst is activated through a rigorous calcination process at 500°C under nitrogen, creating a stable active surface that facilitates the dehydration and decarboxylation of the acid mixture. When the preheated acid mixture contacts this catalyst bed at optimized temperatures of 375-450°C, the reaction proceeds with remarkable selectivity, achieving crude product yields of up to 96% and selectivity levels exceeding 92%, thereby minimizing the formation of difficult-to-remove byproducts.
Following the ketone synthesis, the process employs a sophisticated one-pot chlorination and hydrolysis mechanism driven by phase transfer catalysis. In this stage, tetrabutylammonium bromide acts as the crucial phase transfer agent, facilitating the interaction between the organic phase containing the ketone and carbon tetrachloride, and the aqueous phase containing sodium hydroxide. This setup allows for the in-situ generation of the reactive species necessary for alpha-chlorination and subsequent hydrolysis without the need for isolating unstable chloroketone intermediates. The reaction is conducted at moderate temperatures of 80-90°C, ensuring controlled kinetics that favor the formation of the target 1-hydroxycyclohexyl phenyl ketone while suppressing side reactions. This mechanistic efficiency translates directly to higher overall process yields, with final isolation yields consistently reaching approximately 90% after vacuum rectification.
How to Synthesize Photoinitiator 184 Efficiently
Implementing this synthesis route requires careful attention to catalyst preparation and thermal management to maximize the benefits of the continuous flow ketonization step. The process begins with the meticulous formulation of the ZnO/MgO catalyst slurry, followed by drying and calcination to ensure structural integrity under high-temperature conditions. Once the reactor is charged, the liquid feed of mixed acids is introduced at a controlled rate using nitrogen as a carrier gas, maintaining a steady state within the fixed bed. The subsequent one-pot conversion requires precise stoichiometric control of the base and halogen source to ensure complete conversion without excessive degradation. For a detailed breakdown of the specific operational parameters, equipment setups, and workup procedures required to replicate this high-yield synthesis, please refer to the standardized protocol outlined below.
- Prepare a fixed-bed catalyst by impregnating zinc oxide and magnesium oxide onto a carrier like aluminum oxide, then calcine at 500°C.
- Mix benzoic acid and cyclohexanecarboxylic acid, preheat to 85°C, and pass over the catalyst bed at 300-500°C to form cyclohexyl phenyl ketone.
- React the ketone intermediate with carbon tetrachloride and sodium hydroxide using tetrabutylammonium bromide as a phase transfer catalyst in a one-pot system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers transformative advantages that extend far beyond simple chemical efficiency. By fundamentally altering the input materials and reaction conditions, the new route dismantles the complex cost structures associated with hazardous waste disposal and specialized safety infrastructure. The elimination of corrosive and toxic reagents like phosphorus trichloride and chlorine gas significantly lowers the barrier to entry for production facilities, allowing for operation in regions with stricter environmental regulations without incurring prohibitive compliance costs. This shift not only stabilizes the supply chain against regulatory shocks but also opens up opportunities for substantial cost reduction in photoinitiator manufacturing through simplified logistics and reduced insurance premiums.
- Cost Reduction in Manufacturing: The economic impact of removing hazardous reagents cannot be overstated, as it eliminates the need for expensive corrosion-resistant equipment and complex scrubbing systems required for handling chlorine and hydrogen chloride gases. Furthermore, the consolidation of four reaction steps into two significantly reduces energy consumption, labor hours, and solvent usage, leading to a drastically simplified cost structure. By avoiding the generation of phosphorus and aluminum-containing waste streams, manufacturers can bypass costly wastewater treatment processes and solid waste disposal fees, resulting in significant operational expenditure savings that improve overall margin potential.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as benzoic acid and cyclohexanecarboxylic acid ensures a robust and stable supply chain, free from the volatility often associated with specialized chlorinating agents. Since these feedstocks are widely produced and available from multiple global sources, the risk of supply disruption due to raw material shortages is minimized. Additionally, the continuous nature of the fixed-bed ketonization step allows for scalable production rates that can be easily adjusted to meet fluctuating market demand, ensuring consistent delivery schedules and reducing lead time for high-purity photoinitiator intermediates.
- Scalability and Environmental Compliance: From an environmental standpoint, the process aligns perfectly with modern green chemistry principles by reducing the E-factor (mass of waste per mass of product) through the elimination of stoichiometric inorganic byproducts. The absence of benzene as a solvent removes a major carcinogenic hazard from the workplace, simplifying occupational health and safety compliance and reducing the administrative burden on EHS teams. This cleaner profile makes the technology highly scalable, as expanding capacity does not require proportional increases in waste treatment infrastructure, facilitating smoother commercial scale-up of complex polymer additives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis pathway. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to legacy technologies. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement specialists assessing supplier capabilities.
Q: What are the primary safety advantages of the new Photoinitiator 184 process?
A: The new process eliminates the use of hazardous chlorine gas, phosphorus trichloride, and carcinogenic benzene solvents, significantly reducing operational risks and environmental compliance burdens.
Q: How does the catalytic ketonization step improve yield compared to traditional methods?
A: By utilizing a specific ZnO/MgO catalyst system at optimized temperatures (375-450°C), the process achieves high selectivity (over 92%) and yields (up to 96%) while avoiding the complex purification steps associated with Friedel-Crafts byproducts.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes continuous fixed-bed reactor technology for the ketonization step and simplified batch processing for the final conversion, making it highly scalable and easier to automate than the multi-step traditional route.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Photoinitiator 184 Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthesis routes is critical for the future of the fine chemical industry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the catalytic ketonization process are translated into reliable, high-volume supply. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced reactor systems capable of handling high-temperature continuous flow chemistry, allowing us to meet stringent purity specifications required by top-tier UV curing applications. We are committed to delivering Photoinitiator 184 that not only meets performance benchmarks but also adheres to the highest standards of safety and environmental stewardship.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your supply chain needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can drive value for your organization. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can support your long-term strategic goals.
