Advanced Industrial Synthesis of Dobutamine Hydrochloride via Safe Carbamate Protection Strategy
Advanced Industrial Synthesis of Dobutamine Hydrochloride via Safe Carbamate Protection Strategy
The global demand for cardiotonic agents continues to rise alongside an aging population, placing immense pressure on the supply chains for critical active pharmaceutical ingredients like Dobutamine Hydrochloride. A groundbreaking patent, CN115322109A, disclosed in late 2022, introduces a transformative preparation method that fundamentally reimagines the synthetic architecture of this vital sympathomimetic drug. Unlike conventional pathways that rely on hazardous reagents or complex catalytic systems, this novel approach leverages a strategic carbamate protection protocol to achieve high-purity outcomes with significantly reduced operational risks. For R&D directors and procurement specialists alike, this technology represents a pivotal shift towards safer, more sustainable, and cost-effective manufacturing paradigms. By replacing aggressive demethylation or debenzylation steps with mild alkaline hydrolysis, the process not only enhances the impurity profile of the final API but also drastically simplifies the engineering requirements for commercial scale-up. This report provides a deep technical dissection of this innovation, highlighting its potential to redefine the standards for a reliable dobutamine hydrochloride supplier in the competitive global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Dobutamine Hydrochloride has been plagued by significant safety hazards and economic inefficiencies inherent to traditional protecting group strategies. One prevalent method, illustrated in prior art such as WO2008047382A2, employs 3,4-dibenzyloxy phenethylamine as a starting material, necessitating a final debenzylation step using palladium on carbon (Pd/C) under high-pressure hydrogenation conditions. This reliance on precious metal catalysts introduces severe supply chain vulnerabilities due to the fluctuating cost of palladium and creates critical quality control challenges regarding heavy metal residues in the final product. Furthermore, alternative routes utilizing methoxy-protected intermediates, as seen in patents like CN111807975A, depend heavily on hydrobromic acid (HBr) for the cleavage of ether bonds. The use of concentrated HBr presents acute safety risks, including severe corrosion of reactor vessels and potential harm to personnel, while often failing to consistently deliver the stringent purity levels required for injectable formulations. These legacy processes impose substantial burdens on facility maintenance and waste management, rendering them increasingly obsolete in the face of modern regulatory and economic pressures.
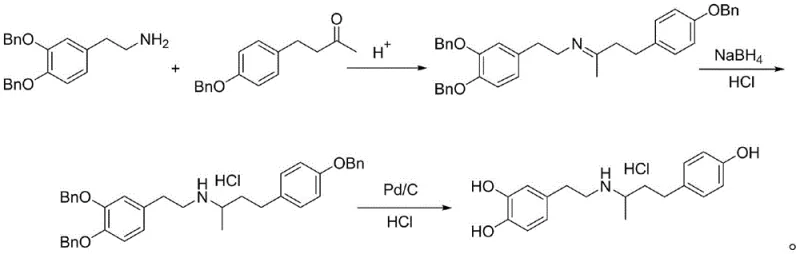
The Novel Approach
In stark contrast to these hazardous legacy protocols, the methodology described in CN115322109A pioneers a streamlined pathway that entirely circumvents the use of bromination agents and transition metal catalysts. The core innovation lies in the substitution of traditional ether or benzyl protecting groups with N,N-dimethylcarbamate moieties, which offer a unique balance of stability and lability. This strategic modification allows the synthetic sequence to proceed through mild condensation and reduction phases without compromising the integrity of the phenolic hydroxyls, only to be removed later under gentle alkaline conditions. By eliminating the need for high-pressure hydrogenation equipment and corrosive acid handling infrastructure, this approach dramatically lowers the barrier to entry for cost reduction in pharmaceutical intermediates manufacturing. The result is a robust, linear synthesis that minimizes side reactions and by-product formation, ensuring a cleaner crude profile before the final crystallization. This technological leap not only safeguards operator health and equipment longevity but also aligns perfectly with the industry's growing mandate for greener, more sustainable chemical processing solutions.
Mechanistic Insights into Carbamate-Mediated Protection and Deprotection
The mechanistic elegance of this new synthesis rests on the precise orchestration of nucleophilic substitution and controlled hydrolysis, facilitated by the unique electronic properties of the dimethylcarbamate group. In the initial phase, dopamine hydrochloride and 4-hydroxyphenyl-2-butanone undergo protection via reaction with N,N-dimethylcarbamoyl chloride in the presence of an acid-binding agent like potassium carbonate. This transformation converts the reactive phenolic hydroxyls into stable carbamates, effectively masking their nucleophilicity to prevent unwanted side reactions during the subsequent carbon-carbon bond-forming steps. Unlike bulky silyl ethers or stubborn benzyl groups, these carbamate functionalities are sufficiently robust to withstand the Lewis acid-catalyzed dehydration condensation required to form the imine intermediate, yet they remain susceptible to cleavage by hydroxide ions. This differential reactivity is the cornerstone of the process, enabling a highly selective deprotection in the final stage that preserves the sensitive amine backbone of the dobutamine molecule while efficiently regenerating the free phenols.
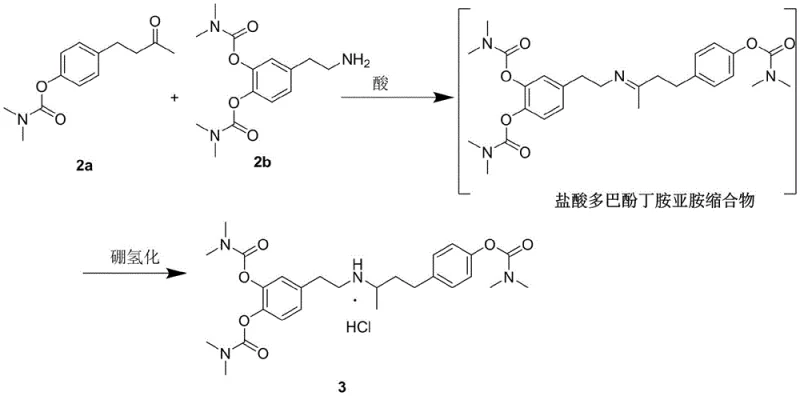
Following the protection phase, the synthesis proceeds through a critical condensation and reduction sequence that constructs the central pharmacophore of the drug. The protected ketone and amine intermediates are subjected to acid-catalyzed dehydration to generate an imine species, which is subsequently reduced using sodium borohydride to establish the secondary amine linkage. This hydroboration step is conducted under mild thermal conditions, typically between 0°C and 30°C, which suppresses the formation of racemic impurities and over-reduction by-products that often plague more aggressive reduction protocols. The final transformation involves the hydrolysis of the carbamate groups using a strong base such as sodium hydroxide at elevated temperatures, followed by neutralization with hydrochloric acid to precipitate the target salt. This final deprotection mechanism is exceptionally clean, yielding carbon dioxide and dimethylamine as volatile by-products that are easily removed, thereby leaving behind a high-purity API free from the persistent organic contaminants associated with traditional ether cleavage methods.
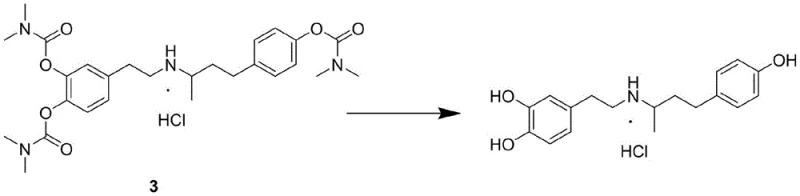
How to Synthesize Dobutamine Hydrochloride Efficiently
The practical implementation of this novel synthetic route offers a clear roadmap for laboratories and production facilities aiming to adopt this superior methodology. The process is divided into three distinct operational stages: the initial protection of raw materials, the core coupling and reduction to form the protected intermediate, and the final hydrolysis and salt formation. Each step utilizes common, commercially available reagents and solvents, avoiding the need for exotic catalysts or specialized high-pressure reactors. The detailed standardized synthesis steps, including specific molar ratios, temperature controls, and workup procedures derived directly from the patent examples, are outlined below to guide technical teams in replicating this high-efficiency protocol.
- Protect the hydroxyl groups of 4-hydroxyphenyl-2-butanone and dopamine hydrochloride using N,N-dimethylcarbamoyl chloride and an acid-binding agent to form stable carbamate intermediates.
- Perform catalytic dehydration condensation followed by hydroboration reduction using sodium borohydride to construct the carbon-nitrogen backbone of the dobutamine skeleton.
- Execute mild alkaline hydrolysis to remove the carbamate protecting groups, followed by salt formation with hydrochloric acid to yield the final pharmaceutical grade product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this carbamate-based synthesis route translates into tangible strategic advantages that extend far beyond simple unit cost metrics. By fundamentally altering the chemical architecture of the production process, this method mitigates several long-standing bottlenecks that have historically constrained the availability and pricing of high-purity dobutamine hydrochloride. The elimination of palladium catalysts removes exposure to the volatile precious metals market, while the absence of hydrobromic acid reduces the regulatory burden associated with handling and disposing of highly corrosive hazardous wastes. These factors combine to create a more resilient supply chain capable of sustaining continuous production runs without the frequent interruptions caused by equipment corrosion or catalyst poisoning. Furthermore, the simplified purification requirements reduce solvent consumption and energy usage, contributing to a lower overall carbon footprint and enhanced sustainability credentials for the final product.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and hazardous reagents with cost-effective alternatives. By removing the requirement for palladium on carbon, manufacturers eliminate both the high upfront cost of the catalyst and the downstream expenses associated with metal scavenging and recovery systems. Additionally, the avoidance of hydrobromic acid significantly reduces maintenance costs for reactor vessels and piping, as the mild alkaline conditions used in the final step are far less corrosive than traditional acidic cleavage methods. These operational efficiencies accumulate to deliver substantial cost savings over the lifecycle of the production facility, allowing for more competitive pricing structures in the global marketplace without compromising on quality margins.
- Enhanced Supply Chain Reliability: From a logistics perspective, this synthesis route enhances reliability by relying on commodity chemicals that are widely available from multiple global sources. Starting materials such as dopamine hydrochloride and 4-hydroxyphenyl-2-butanone are established bulk chemicals, reducing the risk of supply disruptions that often accompany specialized or proprietary intermediates. The robustness of the reaction conditions also means that production is less sensitive to minor variations in raw material quality or environmental parameters, ensuring consistent batch-to-batch performance. This stability is crucial for maintaining the continuity of supply required by large-scale pharmaceutical customers who demand strict adherence to delivery schedules and quality specifications.
- Scalability and Environmental Compliance: The design of this process inherently supports seamless commercial scale-up of complex pharmaceutical intermediates due to its reliance on standard unit operations. The reactions proceed at atmospheric pressure and moderate temperatures, removing the need for specialized high-pressure autoclaves or cryogenic cooling systems that limit batch sizes in traditional methods. Moreover, the generation of benign by-products during the hydrolysis step simplifies wastewater treatment protocols, facilitating easier compliance with increasingly stringent environmental regulations. This alignment with green chemistry principles not only future-proofs the manufacturing asset against regulatory changes but also enhances the brand value of the supplier as a responsible partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this innovative preparation method, we have compiled a set of frequently asked questions based on the specific details provided in the patent documentation. These inquiries address common concerns regarding safety, purity, and scalability, offering transparent insights into how this technology outperforms legacy synthesis routes. Understanding these nuances is essential for technical decision-makers evaluating the feasibility of integrating this new process into their existing manufacturing portfolios.
Q: Why is the carbamate protection strategy superior to traditional benzyl or methoxy protection for Dobutamine synthesis?
A: Traditional methods often rely on benzyl groups requiring hazardous hydrogenation with palladium catalysts or methoxy groups requiring corrosive hydrobromic acid for removal. The carbamate strategy utilizes mild alkaline hydrolysis for deprotection, eliminating heavy metal contamination risks and severe corrosion issues, thereby enhancing product purity and equipment longevity.
Q: How does this novel route impact the environmental footprint of Dobutamine manufacturing?
A: By avoiding the use of high-risk reagents like hydrobromic acid and eliminating the need for pressurized hydrogenation steps, the process significantly reduces hazardous waste generation. The mild reaction conditions and aqueous workup procedures align with green chemistry principles, simplifying wastewater treatment and lowering overall environmental compliance costs.
Q: Is this synthesis method scalable for commercial production volumes?
A: Yes, the process is specifically designed for industrial scalability. It utilizes readily available starting materials like dopamine hydrochloride and operates under moderate temperatures and atmospheric pressure for most steps. The elimination of specialized high-pressure equipment and the use of common solvents facilitate seamless scale-up from pilot batches to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dobutamine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven execution capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the carbamate protection strategy are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions outlined in this patent, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international pharmacopeial standards. We are committed to leveraging this cutting-edge chemistry to deliver a superior product that enhances the therapeutic efficacy and safety profile of your final formulations.
We invite you to engage with our technical procurement team to explore how this optimized synthesis route can drive value for your organization. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic impact of switching to this safer, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of this high-quality intermediate into your supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
