Advanced Manufacturing Strategy for High-Purity Cabozantinib Intermediates
Advanced Manufacturing Strategy for High-Purity Cabozantinib Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for complex kinase inhibitors like cabozantinib, a broad-spectrum anticancer agent targeting MET and VEGFR. Patent CN110903240A introduces a transformative preparation method that fundamentally alters the synthetic landscape by replacing harsh chlorination steps with a mild ester-hydrolysis strategy. This technical breakthrough addresses critical pain points in API manufacturing, specifically focusing on impurity control and operational safety. By utilizing a mono-ester intermediate rather than direct dicarboxylic acid activation, the process achieves superior regioselectivity and minimizes side reactions. For R&D directors and procurement specialists, this patent represents a viable pathway to secure a reliable pharmaceutical intermediates supplier capable of delivering high-quality materials with reduced environmental footprints. The following analysis dissects the mechanistic advantages and commercial implications of this novel route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for cabozantinib, such as those disclosed in WO 2005030140 and CN 201080012656.5, rely heavily on the direct activation of 1,1-cyclopropanedicarboxylic acid. These legacy methods typically involve converting both carboxyl groups into acyl chlorides using aggressive reagents like thionyl chloride or oxalyl chloride. As illustrated in the reaction scheme below, this approach necessitates the use of corrosive chemicals that demand specialized, corrosion-resistant reactor equipment and generate significant amounts of acidic waste gas. Furthermore, the lack of differentiation between the two carboxyl groups often leads to statistical mixtures and bis-amide by-products, complicating purification. Some prior art routes even require extreme thermal conditions, with reaction temperatures reaching 165°C, which not only increases energy consumption but also promotes thermal degradation and the formation of difficult-to-remove impurities.
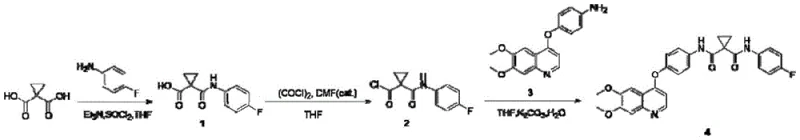
The Novel Approach
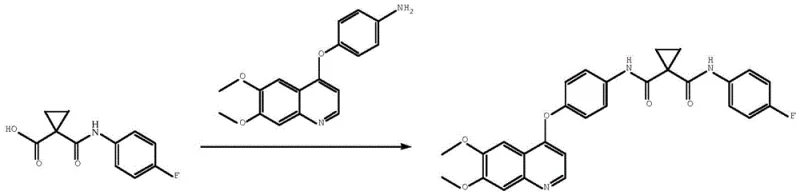
In stark contrast, the methodology presented in CN110903240A employs a strategic "protect-and-deprotect" logic using a mono-alkyl ester of 1,1-cyclopropanedicarboxylic acid as the starting material. This subtle structural modification allows for the selective formation of the first amide bond with 4-fluoroaniline under mild coupling conditions, effectively bypassing the need for acyl chlorides in the initial stage. The subsequent hydrolysis of the ester group to reveal the free carboxylic acid is performed under controlled basic conditions, ensuring the integrity of the cyclopropane ring and the existing amide bond. This sequence culminates in a final condensation with the quinoline moiety at temperatures between -10°C and 50°C. This dramatic reduction in thermal stress and the elimination of chlorinating agents result in a cleaner reaction profile, simpler work-up procedures, and a final product with exceptional purity, making it highly suitable for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into EDC-Mediated Amide Coupling
The core of this innovative synthesis lies in the precise application of carbodiimide-mediated coupling chemistry, specifically using agents like EDC (1-ethyl-3-(3-dimethylaminopropyl)carbodiimide). In the initial step, the mono-methyl ester of 1,1-cyclopropanedicarboxylic acid reacts with 4-fluoroaniline. The carboxylic acid of the ester is activated by EDC to form an O-acylisourea intermediate, which is then attacked by the nucleophilic amine. The presence of an acid-binding agent, such as triethylamine or DIPEA, is crucial here to neutralize the proton released during amide formation, driving the equilibrium forward. Unlike direct dicarboxylic acid activation where both ends compete for the reagent, the ester group acts as a temporary mask, ensuring that coupling occurs exclusively at the free acid site. This regio-control is vital for preventing the formation of symmetrical bis-amides, which are notoriously difficult to separate from the desired mono-amide intermediate.
Following the formation of the mono-amide ester, the mechanism shifts to base-catalyzed hydrolysis. The ester moiety is cleaved using inorganic bases like sodium hydroxide or potassium carbonate, regenerating the carboxylic acid functionality required for the final assembly. The final coupling with 4-(4-aminophenoxy)-6,7-dimethoxyquinoline mirrors the first step but requires careful optimization of stoichiometry to ensure complete conversion without excessive reagent waste. The patent data highlights that maintaining a molar ratio of the acid intermediate to the quinoline amine between 1:1 and 1:1.5 optimizes yield. The reaction mechanism avoids the high-energy transition states associated with acid chloride formation, thereby preserving the stereochemical integrity of the cyclopropane ring and minimizing racemization or ring-opening side reactions that could compromise the biological activity of the final API.
How to Synthesize 1-(4-Fluorophenylcarbamoyl)cyclopropanecarboxylic Acid Efficiently
The synthesis of the key intermediate, 1-(4-fluorophenylcarbamoyl)cyclopropanecarboxylic acid, serves as the linchpin for the entire cabozantinib production line. This compound acts as the central scaffold upon which the quinoline pharmacophore is attached. The patent outlines a streamlined protocol where the mono-ester is first coupled with 4-fluoroaniline, followed by a straightforward hydrolysis step. This two-step sequence from the ester precursor offers a significant advantage over direct dicarboxylic acid methods by simplifying the purification of the intermediate. Detailed standard operating procedures regarding solvent selection, temperature gradients, and quenching protocols are essential for replicating the high yields reported in the examples. For process chemists looking to implement this route, understanding the solubility profiles of the intermediate in solvents like dichloromethane and methanol is critical for maximizing recovery rates during the crystallization phases.
- Condense 1,1-cyclopropyldicarboxylic acid monoalkyl ester with 4-fluoroaniline using EDC and a base to form the mono-amide ester intermediate.
- Hydrolyze the ester group of the intermediate using an inorganic base like sodium hydroxide to obtain 1-(4-fluorophenylcarbamoyl)cyclopropanecarboxylic acid.
- Condense the resulting carboxylic acid with 4-(4-aminophenoxy)-6,7-dimethoxyquinoline using EDC and triethylamine to yield cabozantinib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthesis route described in CN110903240A offers tangible strategic benefits beyond mere chemical elegance. The primary value proposition lies in the substantial simplification of the manufacturing infrastructure required. By eliminating the need for handling large quantities of corrosive chlorinating agents like oxalyl chloride and thionyl chloride, facilities can reduce their dependency on specialized Hastelloy reactors and extensive scrubbing systems for acidic off-gases. This reduction in equipment specialization translates directly into lower capital expenditure (CAPEX) for new production lines and reduced maintenance costs for existing ones. Furthermore, the milder reaction conditions decrease the risk of runaway exotherms, enhancing overall plant safety and potentially lowering insurance premiums associated with hazardous chemical processing.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive and hazardous reagents. Traditional routes require stoichiometric amounts of chlorinating agents and generate equivalent amounts of salt waste that require costly disposal. By switching to EDC-mediated coupling and base hydrolysis, the waste stream becomes significantly less hazardous and easier to treat. Additionally, the higher selectivity of the mono-ester route reduces the loss of valuable starting materials to bis-amide by-products. This improved atom economy means that less raw material is needed to produce the same amount of API, directly lowering the cost of goods sold (COGS). The ability to operate at near-ambient temperatures also results in significant energy savings compared to processes requiring prolonged heating at 165°C.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized reagents and the regulatory scrutiny surrounding hazardous chemicals. The reagents used in this novel pathway, such as EDC, triethylamine, and common inorganic bases, are commodity chemicals with robust global supply chains. This reduces the risk of production stoppages due to raw material shortages. Moreover, the stability of the intermediates generated in this process is reported to be good, allowing for potential storage or transport between different manufacturing sites if a multi-vendor strategy is employed. The simplified purification steps, often involving simple recrystallization from ethanol and water, ensure that production bottlenecks related to complex chromatography or distillation are minimized, leading to faster batch turnover times.
- Scalability and Environmental Compliance: As regulatory bodies worldwide tighten restrictions on pharmaceutical manufacturing emissions, the environmental profile of a synthesis route becomes a critical compliance factor. This method generates significantly less halogenated waste and acidic effluent, aligning with green chemistry principles and facilitating easier permitting for commercial scale-up. The process has been demonstrated to be effective on scales up to 2000L reactors in the patent examples, proving its viability for commercial scale-up of complex pharmaceutical intermediates. The high purity of the crude product (often exceeding 98%) reduces the burden on downstream purification units, allowing for higher throughput and consistent quality that meets stringent international pharmacopeia standards without extensive reprocessing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cabozantinib synthesis technology. These insights are derived directly from the experimental data and claims within patent CN110903240A, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the specific advantages of the ester-hydrolysis strategy over conventional acyl chloride methodologies.
Q: What are the primary advantages of the ester-hydrolysis route over traditional acyl chloride methods?
A: The ester-hydrolysis route described in CN110903240A eliminates the need for corrosive chlorinating agents like thionyl chloride or oxalyl chloride, significantly reducing equipment corrosion risks and hazardous waste generation while maintaining high product purity.
Q: What yields and purity levels can be expected from this synthesis method?
A: Experimental data from the patent indicates that optimizing the alkyl group (e.g., methyl) and reaction conditions can achieve yields up to 91% for the final step and overall product purity exceeding 99%, ensuring suitability for pharmaceutical applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction temperatures ranging from -10°C to 50°C and avoids high-energy steps, making it energetically efficient and safer for scale-up compared to prior art methods requiring temperatures as high as 165°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cabozantinib Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team has extensively analyzed the pathway described in CN110903240A and possesses the capability to execute this mild, high-yield synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped to handle the specific solvent systems and coupling reagents required for this process, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of cabozantinib intermediate meets the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthetic route for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us directly to request specific COA data from our pilot runs and comprehensive route feasibility assessments. Let us help you optimize your cabozantinib supply chain with a process that balances efficiency, safety, and cost-effectiveness.
