Scalable L-Carnitine Production via Advanced Ruthenium-Catalyzed Asymmetric Hydrogenation
Scalable L-Carnitine Production via Advanced Ruthenium-Catalyzed Asymmetric Hydrogenation
The pharmaceutical and nutraceutical industries constantly seek robust synthetic routes for high-value active ingredients like L-carnitine (VBT), a critical nutrient for metabolic health. Patent CN102952028A introduces a transformative preparation method for L-carnitine and its key chiral intermediate, addressing long-standing inefficiencies in asymmetric synthesis. This technology leverages a specialized ruthenium-phosphine complex to achieve high enantioselectivity under remarkably mild conditions, followed by an optimized quaternary ammoniation step that drastically simplifies purification. For R&D directors and procurement strategists, this patent represents a significant leap forward in process chemistry, offering a pathway to high-purity products with reduced operational complexity and enhanced safety profiles compared to traditional high-pressure hydrogenation or fermentation methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of L-carnitine has been plagued by significant technical bottlenecks that hinder cost-effective manufacturing and consistent quality control. Prior art, such as that disclosed in patent EP295109, relied on phosphine-part ruthenium complexes that yielded products with unacceptably low enantiomeric excess (ee) values of only 67%, necessitating expensive and wasteful recycling or disposal of the unwanted enantiomer. Other methods, like those in EP0339764, achieved better stereocontrol but required extreme hydrogen pressures of 10MPa, creating severe safety hazards and demanding capital-intensive high-pressure reactor infrastructure that limits scalability. Furthermore, subsequent quaternary ammoniation steps in older processes often suffered from poor selectivity, generating substantial by-products that forced manufacturers to rely on tedious resin column chromatography, resulting in dismal overall yields of merely 30% to 40% in actual production environments.
The Novel Approach
The methodology outlined in CN102952028A fundamentally reengineers the synthetic landscape by decoupling the harsh conditions from high performance. The novel approach utilizes a specific [RuCl(p-cymene)((R)-C3-Tunephos)]Cl catalyst system that operates efficiently at moderate hydrogen pressures of just 4 to 5 atmospheres and temperatures between 90°C and 100°C. This shift not only mitigates safety risks associated with high-pressure hydrogenation but also significantly lowers energy consumption and equipment maintenance costs. In the second stage, the process replaces complex chromatographic separations with a streamlined simultaneous dropwise addition technique into an aqueous salt solution, facilitating easy product isolation via electrodialysis. This holistic optimization ensures that the final L-carnitine product achieves superior purity and yield without the burden of intricate downstream processing.
Mechanistic Insights into Ru-Catalyzed Asymmetric Hydrogenation and Quaternary Ammoniation
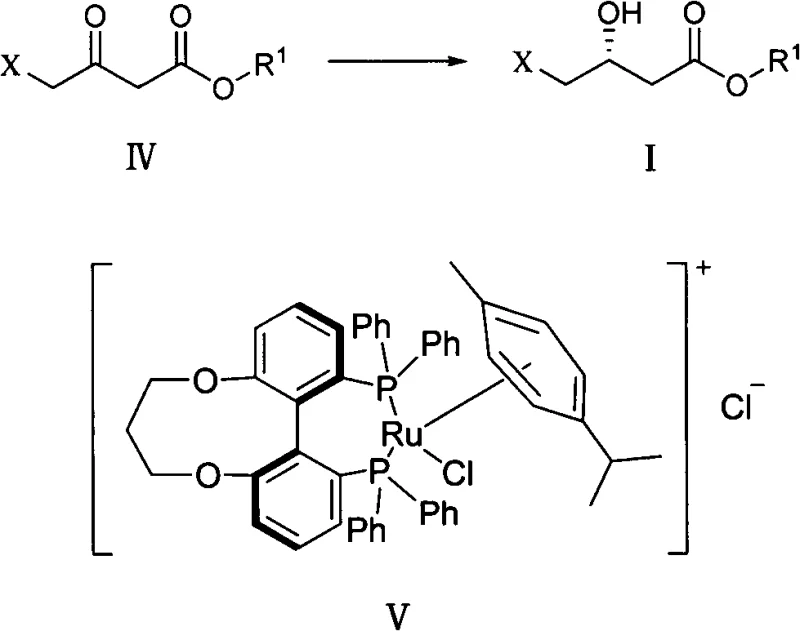
The core of this technological breakthrough lies in the precise stereochemical control exerted by the chiral ruthenium catalyst during the reduction of 4-chloroacetoacetic ester. The catalyst, featuring a Tunephos ligand framework, creates a highly defined chiral environment around the ruthenium center, effectively differentiating between the prochiral faces of the ketone substrate. This interaction directs the hydride transfer exclusively to form the desired (R)-configuration, minimizing the formation of the inactive (S)-enantiomer. The use of an ethanol-water mixed solvent system further enhances catalyst stability and substrate solubility, ensuring a homogeneous reaction phase that maximizes contact between the catalyst and the reactant. This mechanistic precision is crucial for pharmaceutical applications where regulatory bodies mandate strict limits on impurity profiles and enantiomeric purity.
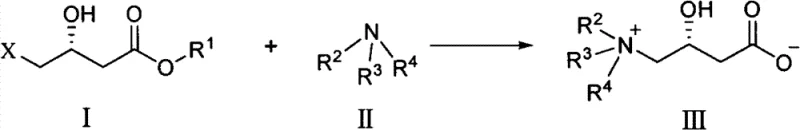
Following the hydrogenation, the transformation of the chiral intermediate into L-carnitine involves a nucleophilic substitution mechanism that is carefully managed to prevent side reactions. The process employs a simultaneous addition strategy where the halogenated intermediate and the trimethylamine base are introduced concurrently into a cooled aqueous medium containing inorganic salts. This specific protocol maintains a low local concentration of reactants, which kinetically favors the desired SN2 substitution over elimination or hydrolysis side reactions that typically degrade yield. The presence of inorganic salts like sodium chloride aids in the salting-out effect during workup, while the low temperature range of -10°C to 5°C suppresses thermal degradation. The result is a clean conversion to the quaternary ammonium salt, which can be efficiently purified through electrodialysis to remove inorganic ions, leaving behind high-purity L-carnitine suitable for sensitive nutritional applications.
How to Synthesize (R)-4-chloro-3-hydroxy ethyl butyrate Efficiently
Implementing this synthesis route requires careful attention to catalyst loading and reaction parameters to replicate the high ee values reported in the patent data. The process begins with the preparation of the hydrogenation mixture, ensuring strict exclusion of oxygen to protect the sensitive ruthenium catalyst from oxidation. Operators must maintain the hydrogen pressure within the narrow optimal window of 4 to 5 atm while heating the ethanol-water solvent system to roughly 90°C to initiate the reduction. Detailed standard operating procedures regarding the specific molar ratios of catalyst to substrate and the precise timing of reagent addition are critical for success. For a comprehensive breakdown of the exact experimental conditions and stoichiometry required to achieve these results, please refer to the standardized synthesis guide below.
- Perform asymmetric catalytic hydrogenation of 4-chloroacetoacetic ester using [RuCl(p-cymene)((R)-C3-Tunephos)]Cl catalyst in ethanol/water at 90-100°C.
- Simultaneously add the resulting chiral intermediate and trimethylamine/alkali solution into a cooled aqueous salt solution.
- Purify the crude L-carnitine product via electrodialysis desalination and precipitation recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere technical superiority. By eliminating the need for ultra-high-pressure reactors and complex chromatography columns, the capital expenditure (CAPEX) required for facility setup is significantly reduced, allowing for more flexible manufacturing footprints. The simplified workflow reduces the number of unit operations, which directly correlates to lower labor costs and reduced cycle times, enhancing the overall agility of the supply chain. Furthermore, the reliance on commercially available raw materials and standard solvents like ethanol minimizes exposure to volatile specialty chemical markets, ensuring a more stable and predictable cost structure for long-term production planning.
- Cost Reduction in Manufacturing: The transition from high-pressure hydrogenation (10MPa) to moderate pressure (4-5 atm) drastically reduces energy consumption and safety compliance costs associated with high-risk operations. Additionally, the replacement of resin column chromatography with electrodialysis and crystallization eliminates the recurring expense of specialized resins and solvents, leading to substantial operational expenditure (OPEX) savings. The higher yield per batch means less raw material waste, optimizing the cost of goods sold (COGS) and improving margin potential for the final active ingredient.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, particularly the tolerance for aqueous environments and moderate temperatures, reduces the risk of batch failures due to minor process deviations. This reliability ensures consistent output volumes, allowing supply chain planners to make more accurate forecasts and commitments to downstream customers. The use of common inorganic salts and alkalis further secures the supply line against disruptions often seen with exotic reagents, guaranteeing continuity of supply even during global raw material shortages.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing water-based systems in the second step which aligns with green chemistry principles by reducing organic solvent waste. The simplified purification via electrodialysis generates less hazardous waste compared to silica gel or resin disposal, easing the burden on wastewater treatment facilities and ensuring compliance with increasingly stringent environmental regulations. This environmental compatibility facilitates smoother regulatory approvals and permits for expansion, supporting seamless capacity increases from pilot to commercial scales.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this L-carnitine synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is essential for technical teams evaluating the integration of this route into existing manufacturing portfolios.
Q: What are the advantages of the Ru-catalyzed hydrogenation step in this patent?
A: The process utilizes a specific Ruthenium complex catalyst that operates under mild pressure (4-5 atm) compared to prior art requiring 10MPa, achieving high enantiomeric excess (ee) values up to 94.2% without severe reaction conditions.
Q: How does the new quaternary ammoniation method improve yield?
A: By simultaneously dropping the chiral intermediate and amine/alkali solution into a salt solution at low temperatures (-10 to 5°C), the method suppresses by-product formation, increasing yields to over 80% compared to 30-40% in older resin column methods.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly highlights broad application prospects due to mild conditions, convenient post-treatment via electrodialysis, and the elimination of complex chromatography purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Carnitine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into reliable commercial realities for our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this ruthenium-catalyzed process are fully realized in large-scale manufacturing. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of L-carnitine intermediate meets the highest international standards for enantiomeric excess and chemical purity, providing you with a secure foundation for your final product formulation.
We invite you to collaborate with us to leverage this advanced synthesis route for your specific market needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can optimize your supply chain and drive down costs while maintaining uncompromised quality.
