Advanced Preparation of Organic Sulfonic Acids via Silver Salt Precipitation for Commercial Scale-up
The synthesis of high-purity organic sulfonic acids represents a critical challenge in fine chemical manufacturing, particularly when dealing with substrates that exhibit poor solubility in aqueous media. Patent CN102746078A introduces a transformative preparation method that leverages the unique solubility properties of silver salts to overcome these longstanding limitations. This technology addresses the specific pain point where traditional acidification methods fail due to the low water solubility of certain organic sulfonates at ambient temperatures. By utilizing a double precipitation strategy involving soluble silver salts and hydrohalic acids, the process ensures the efficient conversion of diverse sulfonate salts—including alkyl, aryl, and heterocyclic derivatives—into their corresponding free acids. This approach is not merely a laboratory curiosity but a robust industrial solution applicable to the synthesis of surfactants, water-soluble dyes, ion exchange resins, and pharmaceutical intermediates. The method fundamentally shifts the paradigm from equilibrium-limited acidification to precipitation-driven purification, offering a pathway to higher purity products with simplified downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of organic sulfonic acids from their salt forms has relied heavily on strong mineral acid acidification or ion-exchange resin technologies, both of which suffer from significant operational and economic drawbacks. The traditional method of adding strong acids like sulfuric acid or hydrochloric acid gas to aqueous sulfonate solutions generates massive quantities of inorganic salt byproducts, such as sodium sulfate or sodium chloride, which complicate the isolation of the target organic acid. These inorganic contaminants often require energy-intensive removal steps, such as repeated crystallization or extensive washing, which drastically reduce overall yield and increase production costs. Furthermore, for organic sulfonates with long carbon chains, fluorinated groups, or bulky aromatic substituents, water solubility is often negligible at room temperature, rendering aqueous acidification ineffective or requiring excessive heating that risks thermal decomposition. Ion-exchange resin methods, while cleaner, are limited by resin capacity, slow flow rates, and the requirement for the feed solution to have sufficient solubility to pass through the column, making them unsuitable for many high-value, low-solubility intermediates.
The Novel Approach
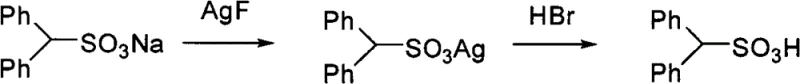
The innovative methodology described in the patent data circumvents these solubility and purity bottlenecks by employing a metathesis reaction driven by the low solubility product constants of silver compounds. Instead of forcing a soluble acid to displace a sulfonate in solution, this process first converts the organic sulfonate into an insoluble organic silver sulfonate precipitate using a soluble silver salt like Silver Nitrate or Silver Acetate. This initial precipitation step effectively separates the organic anion from the original cation (such as sodium, potassium, or calcium) and any soluble impurities. Subsequently, the isolated silver sulfonate is reacted with a hydrohalic acid, such as hydrochloric acid or hydrobromic acid. This second reaction exploits the even lower solubility of silver halides (AgCl, AgBr) to drive the equilibrium completely toward the formation of the free organic sulfonic acid. This dual-precipitation mechanism ensures that the final product is free from inorganic salt contamination, as both the intermediate and the byproduct are removed via simple filtration. 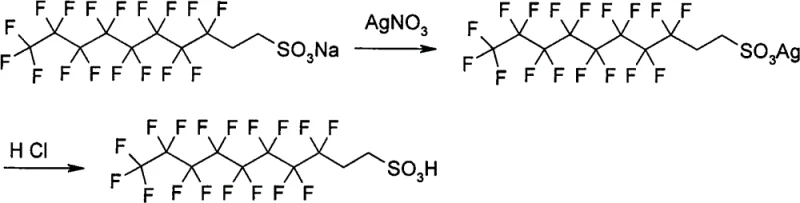
Mechanistic Insights into Silver-Mediated Sulfonate Metathesis
The core chemical mechanism relies on the hierarchical solubility differences between the reactants and products in the silver cycle. In the first stage, the organic sulfonate salt (R-SO3M) reacts with a soluble silver salt (AgX) in a polar solvent system, which may include water, alcohols, acetone, or acetonitrile depending on the lipophilicity of the R group. The formation of the organic silver sulfonate (R-SO3Ag) is favored because these species typically exhibit very low solubility in polar media, causing them to precipitate out of the solution immediately. This precipitation acts as a thermodynamic sink, pulling the reaction to completion even if the starting sulfonate is only sparingly soluble. The patent data highlights that this step can be optimized by adjusting the temperature between 20°C and 150°C and selecting solvents that balance the solubility of the reagents while maintaining the insolubility of the silver intermediate. For instance, fluorinated chains or long alkyl chains that resist dissolution in pure water can be processed effectively in mixed solvent systems like methanol-water or acetonitrile.
In the second mechanistic stage, the isolated organic silver sulfonate is treated with a hydrohalic acid (HY). The driving force here is the extremely low solubility product of silver halides (AgY), which is generally lower than that of the organic silver sulfonate. Upon addition of the acid, the silver ions are sequestered as an insoluble silver halide precipitate (e.g., AgCl or AgBr), releasing the proton to form the free sulfonic acid (R-SO3H). Since the silver halide is insoluble, it can be filtered off, leaving a filtrate that contains primarily the organic sulfonic acid and the solvent. This elegant exchange eliminates the need for complex ion-exchange columns or the generation of soluble inorganic waste salts. The process is versatile enough to handle a wide range of functional groups, including silyl groups, thioethers, and esters, as demonstrated by the successful synthesis of compounds like 3-TMS propanesulfonic acid and various aromatic sulfonic acids without degrading sensitive moieties.
How to Synthesize Organic Sulfonic Acid Efficiently
The practical implementation of this silver-mediated pathway offers a streamlined route for R&D teams aiming to produce high-purity sulfonic acids for electronic or pharmaceutical applications. The process begins with the selection of an appropriate soluble silver salt and solvent system tailored to the specific solubility profile of the organic sulfonate substrate. Following the precipitation and filtration of the silver intermediate, the subsequent acidification step is straightforward and scalable, requiring only standard filtration and distillation equipment. This operational simplicity reduces the technical barrier for scale-up compared to continuous ion-exchange processes. For detailed procedural parameters, stoichiometry, and specific workup conditions tailored to your specific molecule, please refer to the standardized synthesis guide below.
- Dissolve the organic sulfonate salt and a soluble silver salt (e.g., Silver Nitrate) in a polar solvent to precipitate the intermediate organic silver sulfonate.
- Filter the organic silver sulfonate precipitate and react it with a hydrohalic acid (e.g., HCl or HBr) to form the final sulfonic acid and insoluble silver halide.
- Filter off the insoluble silver halide byproduct and distill the filtrate to remove solvents, yielding the pure organic sulfonic acid compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers substantial advantages in terms of cost reduction in fine chemical manufacturing and supply chain reliability. The primary economic benefit stems from the drastic simplification of the purification process. By avoiding the generation of large quantities of soluble inorganic salts, manufacturers can significantly reduce the energy consumption associated with wastewater treatment and product drying. The absence of inorganic contaminants also means that the final product often requires fewer recrystallization steps to meet stringent purity specifications, directly translating to higher throughput and reduced processing time. Furthermore, the ability to process water-insoluble substrates expands the range of accessible raw materials, allowing procurement teams to source cheaper or more readily available sulfonate salts that were previously difficult to acidify efficiently.
- Cost Reduction in Manufacturing: The elimination of soluble inorganic byproducts removes the need for extensive washing and salt-removal procedures, which are often the most costly steps in traditional sulfonic acid production. Additionally, the silver byproduct (silver halide) generated in the second step is a valuable commodity that can be recovered and recycled back into silver nitrate or other silver salts, mitigating the high initial cost of silver reagents. This closed-loop potential for silver recovery transforms what could be a cost liability into a manageable operational expense, ensuring long-term economic viability for large-scale production.
- Enhanced Supply Chain Reliability: The robustness of this method against substrate solubility issues ensures a more consistent supply of critical intermediates. Traditional methods often face batch-to-batch variability when dealing with borderline soluble materials, leading to production delays. This precipitation-based approach is less sensitive to such variations, providing a more stable and predictable manufacturing timeline. The use of common reagents like Silver Nitrate and Hydrochloric Acid further secures the supply chain, as these are globally available commodity chemicals, reducing the risk of raw material shortages.
- Scalability and Environmental Compliance: The process is inherently scalable, relying on standard unit operations such as mixing, filtration, and distillation that are easily adapted from pilot plant to commercial tonnage scales. From an environmental standpoint, the reduction in inorganic salt waste aligns with increasingly strict global regulations on industrial effluent discharge. The ability to recycle silver components minimizes heavy metal waste, supporting sustainability goals and reducing the environmental footprint of the manufacturing facility, which is a key consideration for modern supply chain heads.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this silver salt precipitation technology. These insights are derived directly from the experimental data and claims within the patent literature, providing a factual basis for evaluating the method's suitability for your specific production needs. Understanding these nuances is essential for R&D directors and process engineers planning to integrate this chemistry into their existing workflows.
Q: Why is the silver salt method superior for water-insoluble sulfonates?
A: Traditional acidification requires the sulfonate to be soluble in water for ion exchange or strong acid treatment. This silver precipitation method works effectively even for organic sulfonates with poor water solubility at room temperature, as the reaction can be driven in polar organic solvents or heated suspensions.
Q: What types of silver salts are suitable for this synthesis?
A: The process utilizes soluble silver salts such as Silver Nitrate, Silver Fluoride, Silver Perchlorate, or Silver Monoacetate. The choice depends on cost and the specific solubility requirements of the organic substrate.
Q: How does this method improve product purity compared to strong acid acidification?
A: By converting the sulfonate to an insoluble silver intermediate and then reacting it to form an insoluble silver halide byproduct, the method avoids the generation of large amounts of soluble inorganic salts (like NaCl or Na2SO4) that typically contaminate the product in traditional strong acid methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organic Sulfonic Acid Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic methodologies to deliver superior chemical solutions to the global market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We understand that the production of complex organic sulfonic acids requires stringent purity specifications and rigorous QC labs to verify the absence of trace metals and inorganic residues. Our state-of-the-art facilities are equipped to handle the specific filtration and solvent recovery requirements of the silver salt method, guaranteeing a consistent supply of high-quality intermediates for your downstream applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our implementation of this patent-protected method can optimize your supply chain and enhance your product quality.
