Revolutionizing 3-Nitrophthalic Acid Production: A Deep Dive into Green Catalytic Oxidation Technology
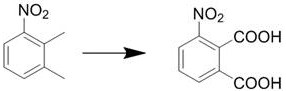
The chemical industry is currently witnessing a paradigm shift towards greener, more sustainable synthesis routes for critical organic intermediates, a trend vividly exemplified by the technological breakthroughs detailed in patent CN114685280A. This specific intellectual property outlines a novel preparation method for 3-nitrophthalic acid, a pivotal building block extensively utilized in the synthesis of photosensitive materials, pharmaceuticals, and high-performance dyes. Unlike traditional methodologies that rely heavily on hazardous reagents, this innovation leverages a sophisticated Co-Mn composite catalyst to drive the oxidation of 3-nitro-o-xylene with exceptional efficiency. For R&D directors and procurement strategists alike, understanding the nuances of this catalytic system is paramount, as it promises not only to elevate product purity but also to fundamentally alter the cost structure and environmental footprint of high-purity dye intermediate manufacturing. The patent explicitly highlights a conversion rate of the raw material reaching up to 96%, with mass yields approaching theoretical limits, signaling a robust pathway for commercial scale-up of complex organic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 3-nitrophthalic acid has been plagued by significant technical and environmental hurdles that compromise both economic viability and operational safety. The most prevalent conventional method involves the direct oxidation of 3-nitro-o-xylene using strong oxidants such as nitric acid or potassium permanganate, often under high temperature and pressure conditions. As detailed in the background art of the referenced patent, these processes impose severe corrosive stress on reactor equipment due to the aggressive nature of nitric acid, leading to frequent maintenance downtimes and capital expenditure on specialized alloy materials. Furthermore, the generation of substantial quantities of waste acid presents a formidable disposal challenge, creating toxic effluent streams that require complex and costly neutralization treatments before discharge. Another traditional route involving the nitration of phthalic anhydride suffers from poor atom economy and the formation of difficult-to-separate isomers, specifically 4-nitrophthalic acid, which drastically complicates downstream purification and reduces the overall yield of the desired 3-isomer.
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in patent CN114685280A introduces a liquid-phase catalytic oxidation system that utilizes molecular oxygen or air as the terminal oxidant, mediated by a highly active Co-Mn composite catalyst. This methodology effectively circumvents the need for stoichiometric amounts of hazardous strong acids, thereby eliminating the root cause of equipment corrosion and waste acid pollution. The core innovation lies in the specific formulation of the catalyst, which integrates manganese salts, cobalt salts, bromide salts, and crucially, zirconium salts, to create a synergistic effect that enhances both activity and selectivity. By operating under milder conditions ranging from 80°C to 180°C and moderate pressures of 0.1 MPa to 4 MPa, this process achieves a remarkable conversion rate of 3-nitro-o-xylene up to 96% while maintaining a mass yield close to 100%. This represents a transformative leap for cost reduction in dye intermediate manufacturing, as it simplifies the post-reaction workup to basic filtration and washing, removing the need for energy-intensive distillation or complex recrystallization steps often required to remove acid residues.
Mechanistic Insights into Co-Mn-Zr-Br Catalytic Oxidation
The efficacy of this synthesis route is deeply rooted in the intricate radical chain mechanism facilitated by the multi-component catalyst system. The Co-Mn composite acts as a redox mediator, cycling between different oxidation states to activate molecular oxygen and generate reactive radical species capable of abstracting hydrogen atoms from the methyl groups of the 3-nitro-o-xylene substrate. The inclusion of bromide salts serves as a crucial co-catalyst, generating bromine radicals that accelerate the initiation step of the oxidation cycle, ensuring rapid conversion even at lower temperatures. However, the true differentiator in this patent is the incorporation of zirconium salts, which are posited to stabilize the active catalytic species and prevent the formation of inactive precipitates that often deactivate standard Co-Mn systems over time. This stabilization effect extends the service life of the catalyst and maintains high shape selectivity, ensuring that the oxidation proceeds selectively at the methyl positions without degrading the sensitive nitro group or the aromatic ring structure.
Furthermore, the mechanistic pathway inherently suppresses the formation of over-oxidation byproducts and isomeric impurities, which is a critical factor for R&D teams focused on impurity profiling. The high selectivity observed, with HPLC purities consistently exceeding 98% in the provided examples, suggests that the catalyst surface or the solvated complex creates a steric environment that favors the formation of the 3-nitrophthalic acid geometry. This level of control is essential for downstream applications in photosensitive materials and pharmaceuticals, where trace impurities can compromise the performance of the final product. By understanding this mechanism, process chemists can fine-tune parameters such as the molar ratio of Mn:Co:Br:Zr (optimized in the patent at roughly 1:3~4:3~4:0.3~0.5) to maximize throughput while minimizing catalyst loading, which directly translates to improved process economics and reducing lead time for high-purity intermediates.
How to Synthesize 3-Nitrophthalic Acid Efficiently
Implementing this advanced oxidation protocol requires precise control over reaction parameters to fully realize the benefits outlined in the patent literature. The process begins with the preparation of the catalyst solution, where specific salts such as manganese nitrate, cobalt bromide, and zirconium bromide are dissolved in acetic acid, which serves as both the solvent and a reaction medium. The substrate, 3-nitro-o-xylene, is then introduced into a high-pressure reactor equipped with efficient agitation and temperature control systems. Following the charging of materials, the system is pressurized with oxygen or air, and the temperature is ramped to the optimal range of 100°C to 130°C. The reaction progress is monitored to ensure complete conversion, typically achieved within 3 to 15 hours depending on the specific catalyst loading and pressure settings. Upon completion, the mixture is cooled, and the solid product is isolated via filtration, washed to neutral pH, and dried to obtain the final high-purity acid. For a detailed, standardized operating procedure including exact dosing sequences and safety protocols, please refer to the technical guide below.
- Charge the reactor with 3-nitro-o-xylene and the Co-Mn composite catalyst solution in acetic acid solvent.
- Introduce oxygen or compressed air at pressures between 0.1 MPa and 4 MPa to initiate the oxidation reaction.
- Maintain reaction temperature between 80°C and 180°C for 1 to 36 hours, followed by cooling, filtration, and drying to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, the adoption of this Co-Mn catalytic oxidation technology offers profound advantages that extend far beyond simple yield improvements. The elimination of nitric acid and other strong oxidants fundamentally alters the risk profile of the manufacturing process, removing the logistical burdens associated with the storage, handling, and disposal of hazardous corrosive materials. This shift not only enhances workplace safety but also significantly reduces the regulatory compliance costs related to environmental protection and waste management. For procurement managers, this translates into a more stable and predictable cost structure, as the process is less susceptible to fluctuations in the price of specialized oxidizing agents or the costs of waste treatment services. The robustness of the catalyst system also implies longer campaign runs with fewer interruptions for reactor maintenance or cleaning, thereby ensuring a consistent and reliable supply of material to meet downstream production schedules.
- Cost Reduction in Manufacturing: The transition to an air or oxygen-based oxidation system eliminates the recurring expense of purchasing stoichiometric quantities of expensive and hazardous chemical oxidants like nitric acid or potassium permanganate. Additionally, the high conversion rates and selectivity minimize the loss of raw materials to byproducts, maximizing the atom economy of the process. The simplified downstream processing, which avoids complex acid recovery or neutralization steps, further drives down utility consumption and labor costs associated with purification. Consequently, the overall cost of goods sold (COGS) for 3-nitrophthalic acid is substantially reduced, providing a competitive edge in pricing strategies for reliable dye intermediate suppliers.
- Enhanced Supply Chain Reliability: By utilizing air or oxygen as the primary oxidant, the process decouples production from the supply chain vulnerabilities associated with specialized chemical reagents. Air and oxygen are universally available commodities, ensuring that production can continue uninterrupted even during periods of chemical supply shortages. Furthermore, the stability and durability of the Co-Mn-Zr composite catalyst mean that catalyst replenishment frequencies are drastically lowered, reducing the dependency on external catalyst vendors. This self-sufficiency enhances the resilience of the supply chain, allowing manufacturers to guarantee delivery timelines and maintain inventory levels with greater confidence, which is critical for just-in-time manufacturing environments.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of heavy metal strong oxidants make this process inherently safer and easier to scale from pilot plant to full commercial production. The reduction in hazardous waste generation aligns perfectly with increasingly stringent global environmental regulations, mitigating the risk of fines or shutdowns due to non-compliance. The "green" nature of the process also enhances the brand value of the final product, appealing to end-users in the pharmaceutical and electronics sectors who are under pressure to reduce the carbon footprint of their supply chains. This environmental stewardship facilitates smoother permitting processes for capacity expansion, ensuring long-term supply continuity.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this synthesis technology, we have compiled a set of answers based on the specific data points and claims found within the patent documentation. These responses are designed to clarify the operational feasibility and the distinct advantages of the Co-Mn composite catalyst system over legacy methods. Understanding these details is crucial for technical teams evaluating the integration of this process into their existing manufacturing frameworks. The following section provides concise answers to the most pertinent questions regarding catalyst composition, reaction conditions, and product quality assurance.
Q: What are the primary advantages of the Co-Mn composite catalyst over traditional nitric acid oxidation?
A: The Co-Mn composite catalyst eliminates the need for corrosive strong acids like nitric acid, thereby avoiding severe equipment corrosion and the generation of difficult-to-treat waste acid, while achieving conversion rates up to 96%.
Q: How does the addition of Zirconium salts impact the catalytic performance?
A: Zirconium salts act as a stabilizer and promoter within the composite system, enhancing the durability and shape selectivity of the catalyst, which leads to higher yields and easier catalyst recovery compared to standard Co-Mn systems.
Q: Is this process suitable for large-scale industrial production of dye intermediates?
A: Yes, the process operates under relatively mild conditions (80-180°C) using air or oxygen as the oxidant, making it highly scalable, environmentally friendly, and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Nitrophthalic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic processes like the one described in CN114685280A requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO and supplier in the fine chemicals sector, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. Our facilities are equipped with state-of-the-art high-pressure reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-nitrophthalic acid meets the exacting standards required for high-end dye and pharmaceutical applications. We are committed to leveraging such green chemistry advancements to deliver superior value to our global clientele.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic impacts of switching to this catalytic method. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a partnership built on transparency, quality, and mutual growth.
