Advanced Chelation Strategy for Regioselective Synthesis of Epsilon-N-Lauroyl Lysine
Advanced Chelation Strategy for Regioselective Synthesis of Epsilon-N-Lauroyl Lysine
The chemical industry's demand for high-purity amino acid derivatives, particularly for the personal care and cosmetic sectors, has driven significant innovation in regioselective synthesis. Patent CN102617390A introduces a groundbreaking preparation method for epsilon-N-lauroyl lysine, a valuable surfactant and emulsifier, by leveraging a sophisticated metal chelation protection strategy. Unlike traditional approaches that struggle with selectivity, this technology utilizes divalent metal ions to temporarily mask specific functional groups on the lysine backbone. This ensures that the acylation reaction occurs exclusively at the epsilon-amino position, eliminating the formation of unwanted alpha-isomers. The result is a streamlined process capable of delivering product purity greater than 98.5 percent, addressing a critical pain point for manufacturers of high-end cosmetic ingredients who require consistent quality and minimal impurity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl lysine derivatives has been plagued by significant regioselectivity challenges, primarily due to the presence of two distinct amino groups (alpha and epsilon) with comparable nucleophilicity. Conventional methods, such as the direct thermal reaction between lauric acid and lysine or the use of lauroyl chloride in high-boiling organic solvents, invariably produce complex mixtures containing epsilon-N-lauroyl lysine, alpha-N-lauroyl lysine, and even N,N-disubstituted byproducts. Separating these structural isomers is notoriously difficult and costly, often requiring extensive chromatography or recrystallization steps that drastically reduce overall yield. Furthermore, older solvent-based dehydration methods frequently rely on hazardous, high-boiling organic solvents, raising concerns about residual toxicity in the final product, which is unacceptable for cosmetic applications where safety standards are stringent.
The Novel Approach
The methodology disclosed in CN102617390A fundamentally alters the reaction landscape by introducing a protective chelation step prior to acylation. By reacting lysine or its salts with divalent metal ions such as calcium, magnesium, zinc, or copper, the process forms a stable inner complex that effectively locks the alpha-amino and carboxyl groups. This steric and electronic shielding leaves the epsilon-amino group as the sole reactive site for the subsequent amidation with lauroyl chloride. This directional control not only simplifies the reaction mixture but also eliminates the need for hazardous high-boiling solvents, allowing the reaction to proceed in safer aqueous or semi-aqueous media. The outcome is a robust, scalable pathway that minimizes side reactions and facilitates easier downstream purification, directly translating to higher commercial viability.
Mechanistic Insights into Metal Chelation Protection and Regioselective Amidation
The core of this technological advancement lies in the coordination chemistry between the amino acid substrate and the transition or alkaline earth metal ion. When lysine is exposed to a divalent metal cation (M2+) under controlled alkaline conditions (pH 6.5 to 11), the metal ion coordinates simultaneously with the alpha-amino nitrogen and the carboxylate oxygen. This coordination creates a rigid five-membered ring structure, significantly reducing the electron density and nucleophilicity of the alpha-amino group. Consequently, when the acylating agent, lauroyl chloride, is introduced, it cannot effectively attack the shielded alpha-position. Instead, the reaction is directed entirely toward the distal epsilon-amino group, which remains uncoordinated and highly nucleophilic. This precise molecular engineering ensures that the amide bond forms exclusively at the epsilon position, preventing the formation of regioisomeric impurities that plague non-chelated routes.
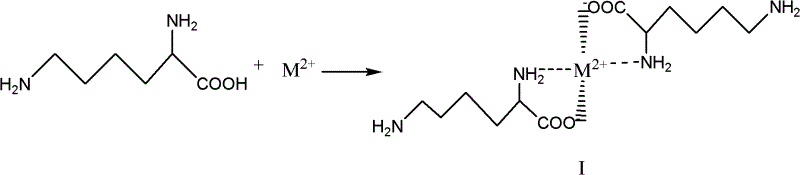
Following the formation of the chelate intermediate, the amidation step proceeds under mild conditions, typically between -20°C and 50°C, maintaining the integrity of the protective complex. The molar ratio of lauroyl chloride to the chelate is carefully managed, often between 1:1 and 2:1, to ensure complete conversion while minimizing excess reagent waste. Once the epsilon-acylation is complete, the final step involves acid hydrolysis. By lowering the pH to an acidic range (pH 1 to 6.5) using mineral acids like hydrochloric or sulfuric acid, the stability of the metal-ligand bond is compromised. The protons compete with the metal ion for the coordinating sites, effectively dismantling the chelate structure and releasing the free alpha-amino and carboxyl groups. This deprotection step regenerates the zwitterionic nature of the lysine headgroup while retaining the newly formed epsilon-amide bond, yielding the target epsilon-N-lauroyl lysine with exceptional purity.
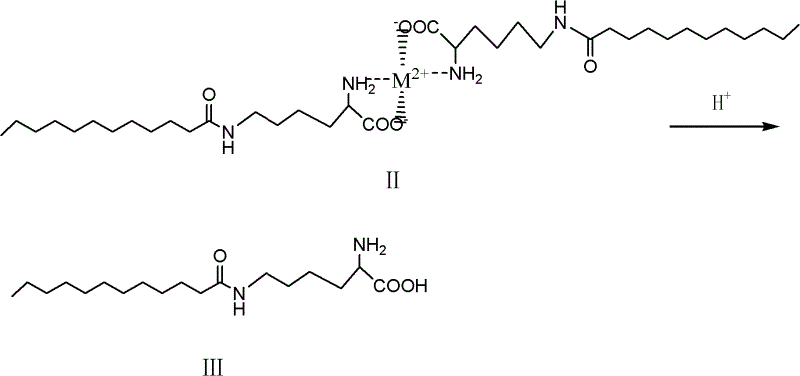
How to Synthesize Epsilon-N-Lauroyl Lysine Efficiently
The synthesis protocol outlined in the patent provides a clear, three-stage operational framework designed for reproducibility and scale-up. The process begins with the preparation of the metal chelate in an aqueous environment, followed by the controlled addition of the acyl chloride, and concludes with an acid workup to isolate the final product. This sequence avoids the complexities of anhydrous conditions required by traditional peptide coupling methods, making it highly accessible for industrial manufacturing. For detailed operational parameters, including specific stirring rates, addition times, and filtration techniques, operators should refer to the standardized synthesis steps provided below which encapsulate the critical process controls identified in the patent examples.
- Form a chelate complex by reacting lysine or its salt with a divalent metal ion (e.g., Ca2+, Zn2+) at pH 6.5-11 to protect the alpha-amino and carboxyl groups.
- Perform regioselective amidation by reacting the metal chelate with lauroyl chloride at controlled pH (6.5-12.5) and low temperature (-20°C to 50°C).
- Execute acid hydrolysis at pH 1-6.5 to destroy the chelate structure, deprotecting the alpha-groups and isolating the final epsilon-N-lauroyl lysine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this chelation-based synthesis route offers substantial strategic benefits over legacy manufacturing methods. The primary advantage lies in the drastic reduction of purification complexity; because the reaction is inherently regioselective, the crude product contains significantly fewer isomeric impurities. This high intrinsic purity reduces the burden on downstream processing units, such as crystallization or chromatography columns, leading to faster batch cycle times and lower utility consumption. Additionally, the ability to operate in aqueous or semi-aqueous systems eliminates the need for expensive and regulated volatile organic compounds (VOCs), thereby reducing solvent procurement costs and waste disposal fees associated with hazardous chemical management.
- Cost Reduction in Manufacturing: The elimination of complex separation processes for alpha- and epsilon-isomers represents a major driver for cost efficiency. In conventional synthesis, a significant portion of the budget is allocated to separating unwanted byproducts, which often results in substantial material loss. By ensuring that the reaction yields almost exclusively the desired epsilon-isomer, the effective yield of usable product per kilogram of raw material is maximized. Furthermore, the use of inexpensive and abundant divalent metal salts like calcium chloride or zinc sulfate as protecting agents replaces costly specialized protecting groups, driving down the raw material cost basis significantly while maintaining high product quality standards.
- Enhanced Supply Chain Reliability: The robustness of this chemical process contributes directly to supply chain stability. The reaction conditions are relatively mild and tolerant, reducing the risk of batch failures due to minor fluctuations in temperature or pH. Moreover, the raw materials required—lysine, lauroyl chloride, and common metal salts—are commodity chemicals with well-established global supply chains, minimizing the risk of raw material shortages. This reliability ensures consistent production schedules and allows manufacturers to maintain steady inventory levels, crucial for meeting the continuous demand of the fast-moving consumer goods (FMCG) and cosmetic industries.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the simplicity of the unit operations involved, which are primarily mixing, filtration, and drying. The avoidance of high-boiling hazardous solvents simplifies the environmental compliance profile, as there is less risk of solvent residues in the final product and reduced emissions during manufacturing. This aligns with increasingly stringent global environmental regulations and the growing consumer demand for 'clean label' cosmetic ingredients, providing a competitive market advantage for suppliers who can certify their products as manufactured via greener, safer chemical pathways.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this chelation-based synthesis route. Understanding these nuances is essential for process engineers and quality assurance teams looking to integrate this technology into their existing manufacturing lines. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industrial application.
Q: How does the metal chelation method improve purity compared to direct amidation?
A: Direct amidation of lysine often results in a mixture of alpha and epsilon acylated products due to similar reactivity of the amine groups. The chelation method selectively blocks the alpha-amino and carboxyl groups using divalent metal ions, forcing the acylation to occur exclusively at the epsilon-position, thereby achieving purity levels exceeding 98.5%.
Q: Which divalent metal ions are suitable for this chelation protection strategy?
A: The patent specifies a range of divalent metal ions including Calcium (Ca2+), Magnesium (Mg2+), Copper (Cu2+), Iron (Fe2+), and Zinc (Zn2+). These ions form stable inner complexes with lysine under alkaline conditions (pH 6.5-11), effectively masking the alpha-functionalities during the subsequent acylation step.
Q: What are the critical conditions for the acid hydrolysis step?
A: The chelate structure is destroyed via acid hydrolysis using mineral acids like hydrochloric or sulfuric acid. The process is optimally conducted at a pH between 2 and 3 and temperatures ranging from 40°C to 50°C for approximately 4 to 5 hours to ensure complete deprotection without degrading the amide bond.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epsilon-N-Lauroyl Lysine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the formulation of next-generation cosmetic and personal care products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate chelation chemistry described in CN102617390A can be translated into efficient, large-scale manufacturing operations. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the regioselectivity and purity of every batch, guaranteeing that our epsilon-N-lauroyl lysine meets the exacting standards required by global brands.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for amino acid-based surfactants. By leveraging our expertise in metal-mediated synthesis, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your transition to this superior manufacturing technology is seamless, compliant, and economically advantageous.
