Advanced Chelation Strategy for High-Purity Epsilon-N-Lauroyl Lysine Manufacturing
The pharmaceutical and cosmetic industries continuously demand higher purity intermediates to ensure safety and efficacy in final formulations. Patent CN102617390B introduces a sophisticated preparation method for epsilon-N-lauroyl lysine, a valuable amino acid derivative widely used as a mild surfactant and conditioning agent in personal care products. This technology addresses critical challenges in regioselective acylation by employing a divalent metal ion chelation strategy. Unlike conventional routes that struggle with isomeric mixtures, this novel approach utilizes the coordination chemistry of metals like calcium, zinc, or magnesium to temporarily mask the alpha-amino and carboxyl functionalities of lysine. By effectively blocking these reactive sites, the process forces the subsequent acylation with lauroyl chloride to occur exclusively at the epsilon-amino position. This results in a product with exceptional purity, reported to be greater than or equal to 98.5 percent, significantly reducing the burden of downstream purification and ensuring consistent quality for reliable cosmetic intermediate supplier networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl amino acids like epsilon-N-lauroyl lysine has been plagued by poor selectivity and harsh reaction conditions. Traditional "hot methods" involving the direct heating of lauric acid and lysine often yield complex mixtures containing significant amounts of the undesired alpha-N-lauroyl isomer, as well as N,N-disubstituted byproducts. Separating these structural analogs is notoriously difficult and costly, often requiring extensive chromatography or recrystallization steps that drastically reduce overall yield. Furthermore, alternative methods utilizing high-boiling organic solvents for dehydration introduce severe safety and environmental liabilities. The residual presence of these hazardous solvents in the final active ingredient is unacceptable for cosmetic applications, limiting the marketability of the product. Direct amidation using lauroyl chloride without protection also suffers from a lack of control, leading to random acylation at both amino groups and generating a crude product that requires tedious aftertreatment procedures to meet industry standards.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by introducing a "protection-reaction-deprotection" sequence mediated by metal coordination. Instead of relying on bulky organic protecting groups that require additional synthetic steps to install and remove, this process leverages the innate ability of lysine to form stable inner complexes with divalent metal ions. This chelation effectively locks the alpha-amine and carboxylate into a rigid cyclic structure, leaving the long-chain epsilon-amine free and nucleophilic. When lauroyl chloride is introduced to this system, the reaction becomes highly directional, targeting only the exposed epsilon-position. This intrinsic selectivity eliminates the formation of alpha-isomers and di-acylated impurities at the source. Consequently, the workup is simplified to a straightforward acid hydrolysis to release the metal and regenerate the free amino acid functionality, delivering a high-purity end product with minimal side reactions and a much cleaner impurity profile suitable for sensitive skin applications.
Mechanistic Insights into Regioselective Acylation via Metal Chelation
The core of this innovation lies in the precise manipulation of coordination chemistry to achieve chemoselectivity. In the first stage, lysine or its hydrochloride salt is reacted with a source of divalent metal ions, such as CaCl2, ZnSO4, or MgCl2, in an aqueous or semi-aqueous medium. Under controlled pH conditions, typically ranging from 6.5 to 11, the metal ion coordinates simultaneously with the alpha-amino nitrogen and the carboxylate oxygen atoms. This forms a stable five-membered chelate ring, effectively rendering these two functional groups inert to electrophilic attack. The stability of this intermediate, designated as Formula I in the patent documentation, is crucial for the success of the subsequent step. The choice of metal ion offers flexibility; while calcium and magnesium are cost-effective and benign, transition metals like copper or zinc may offer different kinetic profiles or solubility characteristics, allowing process engineers to tune the reaction parameters for specific manufacturing constraints.
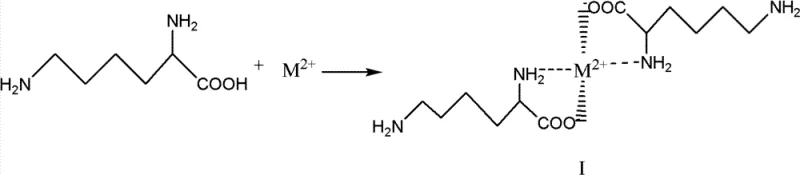
Once the protective chelate is established, the system undergoes the key transformation: acylation. Lauroyl chloride, a long-chain fatty acid chloride, is added to the reaction mixture containing the metal-lysine complex. Because the alpha-amino group is tied up in the coordination sphere of the metal, the nucleophilic attack is directed exclusively toward the epsilon-amino group located at the end of the butyl side chain. This reaction proceeds under mild conditions, often at temperatures between -20°C and 50°C, depending on the specific metal salt and pH optimization. The result is the formation of Formula II, an intermediate where the lauroyl chain is attached solely to the epsilon-nitrogen, while the alpha-end remains protected by the metal ion. This step demonstrates remarkable regiocontrol, avoiding the statistical distribution of products seen in unprotected reactions.
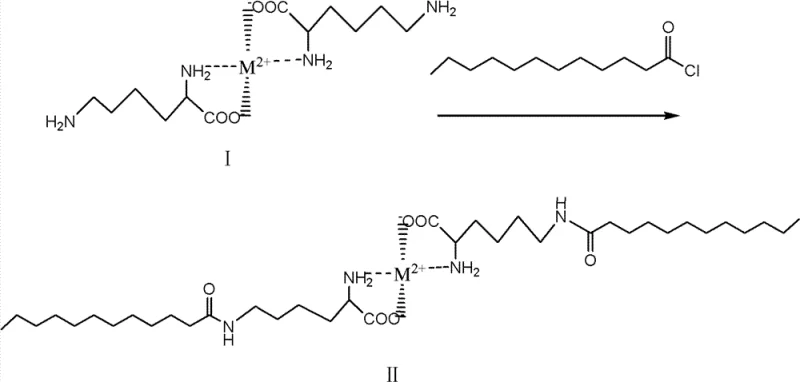
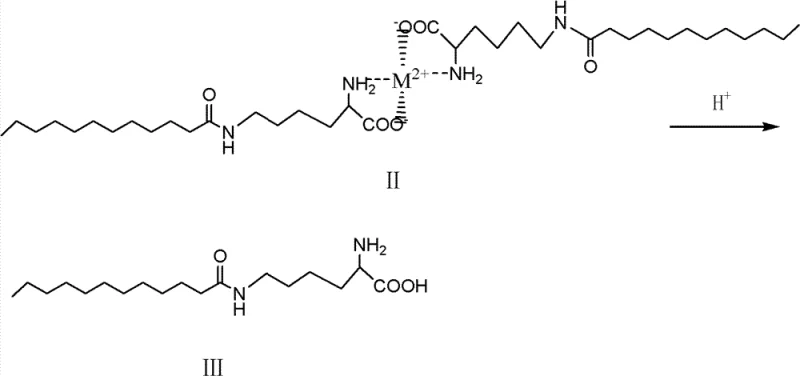
The final stage involves the liberation of the target molecule from the metal template. The Formula II intermediate is subjected to acid hydrolysis using mineral acids like hydrochloric acid or sulfuric acid, or potentially organic acids. Lowering the pH to a range of 1 to 6.5 protonates the amino and carboxyl groups, destabilizing the metal-ligand bonds and causing the chelate structure to collapse. This releases the divalent metal ions back into the solution as salts and regenerates the free alpha-amino and carboxyl groups of the lysine backbone. The final product, epsilon-N-lauroyl lysine (Formula III), precipitates or can be isolated through standard filtration and washing techniques. This deprotection step is clean and efficient, ensuring that no residual organic protecting groups remain, which is a common contamination issue in peptide synthesis. The entire mechanistic pathway ensures that the final product is chemically defined and free from the isomeric impurities that plague older manufacturing technologies.
How to Synthesize Epsilon-N-Lauroyl Lysine Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing this high-value surfactant intermediate. The process is designed to be operationally simple, avoiding the need for exotic reagents or extreme conditions that complicate scale-up. By utilizing water as a primary solvent for the chelation step, the method aligns with green chemistry principles, reducing the reliance on volatile organic compounds. The reaction parameters, including pH, temperature, and molar ratios, have been optimized to maximize yield and purity. For research and development teams looking to replicate or adapt this process, the following guide summarizes the critical operational phases derived from the patent examples. Detailed standardized synthesis steps are provided in the section below to facilitate immediate laboratory implementation.
- Form a chelate complex by reacting lysine or its salt with a divalent metal ion (e.g., Ca2+, Zn2+) at pH 6.5-11 to protect the alpha-amino and carboxyl groups.
- Perform regioselective amidation by reacting the metal chelate intermediate with lauroyl chloride at controlled pH and temperature to acylate the epsilon-amino group.
- Execute acid hydrolysis using mineral or organic acids to destroy the chelate structure, deprotecting the alpha-groups and isolating the final epsilon-N-lauroyl lysine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this chelation-based technology offers tangible benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification train. Since the reaction inherently suppresses the formation of alpha-isomers and di-substituted byproducts, the need for expensive and time-consuming separation techniques is significantly reduced. This directly translates to lower processing costs and higher throughput. Furthermore, the use of abundant and inexpensive metal salts like calcium chloride or zinc sulfate as temporary protecting agents replaces costly organic reagents, driving down the raw material cost per kilogram. The ability to achieve purity levels exceeding 98.5 percent without complex chromatography ensures a consistent supply of quality material, reducing the risk of batch rejections and production delays in downstream cosmetic formulation.
- Cost Reduction in Manufacturing: The elimination of high-boiling hazardous solvents and the reduction in side reactions lead to substantial cost savings. Traditional methods often require energy-intensive distillation to remove solvents or extensive recrystallization to purify isomeric mixtures. In contrast, this aqueous-based chelation method allows for simpler isolation via filtration and washing. The removal of expensive organic protecting groups and the associated reagents for their installation and removal further lowers the bill of materials. Additionally, the high selectivity means less raw material is wasted on forming unwanted byproducts, improving the overall atom economy of the process and enhancing the cost-effectiveness of large-scale production runs.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as lysine, lauroyl chloride, and common metal salts ensures a stable and resilient supply chain. Unlike processes dependent on specialized catalysts or rare reagents that may face geopolitical or logistical bottlenecks, the inputs for this method are widely available from multiple global suppliers. The robustness of the reaction conditions, which tolerate a range of temperatures and pH levels, also reduces the risk of batch failures due to minor process deviations. This reliability is critical for maintaining continuous production schedules and meeting the just-in-time delivery requirements of major personal care brands, ensuring that inventory levels remain optimal without the need for excessive safety stock.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process is highly favorable for commercial scale-up. The avoidance of toxic organic solvents minimizes the generation of hazardous waste streams, simplifying effluent treatment and reducing disposal costs. The use of water as a reaction medium aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. Moreover, the metal salts used can often be recovered or treated easily, further mitigating environmental impact. This "green" profile not only facilitates easier permitting for new manufacturing facilities but also appeals to eco-conscious consumers and brands seeking sustainable sourcing options, thereby adding intangible value to the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of epsilon-N-lauroyl lysine using this patented chelation technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the process capabilities and product specifications. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing formulations or manufacturing lines.
Q: How does the metal chelation method improve purity compared to traditional hot methods?
A: Traditional hot methods often produce mixtures containing alpha-isomers and di-substituted byproducts. The chelation method specifically protects the alpha-amino and carboxyl groups using divalent metal ions, ensuring the acylation occurs exclusively at the epsilon-position, resulting in purity levels exceeding 98.5%.
Q: What divalent metal ions are suitable for this synthesis process?
A: The patent specifies a range of effective divalent metal ions including Calcium (Ca2+), Magnesium (Mg2+), Copper (Cu2+), Iron (Fe2+), and Zinc (Zn2+). Salts such as chlorides and sulfates of these metals can be utilized to form the necessary protective inner complex.
Q: Is this process scalable for industrial production of cosmetic intermediates?
A: Yes, the process utilizes aqueous systems for chelation and standard acid hydrolysis, avoiding high-boiling hazardous organic solvents. This simplifies waste treatment and equipment requirements, making it highly suitable for commercial scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epsilon-N-Lauroyl Lysine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation cosmetic and pharmaceutical products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chelation chemistry described in patent CN102617390B can be seamlessly transferred from the laboratory to the plant. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the absence of alpha-isomers and other impurities in every batch. Our commitment to quality assurance guarantees that the epsilon-N-lauroyl lysine supplied meets the exacting standards required for safe and effective personal care applications.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By leveraging our manufacturing expertise, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your product portfolio.
