Scalable Synthesis of Azoxystrobin Intermediates via Efficient Trimethylamine Catalysis Technology
The global demand for high-efficiency fungicides continues to drive innovation in the synthesis of key agricultural intermediates. Specifically, the preparation of azoxystrobin intermediates, namely Compound B and Compound C, represents a critical bottleneck in the supply chain for this top-tier agrochemical. A significant technological breakthrough detailed in patent CN109721545B introduces a novel catalytic system that fundamentally alters the economic and environmental landscape of this synthesis. By replacing traditional, difficult-to-recover catalysts with a trimethylamine-based system, this method addresses long-standing issues regarding yield consistency and wastewater management. For R&D directors and procurement specialists alike, understanding this shift is vital for securing a competitive edge in the market. The patent outlines a robust pathway that not only accelerates reaction kinetics but also facilitates the recycling of the catalyst, thereby creating a closed-loop system that minimizes waste. This report analyzes the technical nuances of this invention, providing a comprehensive overview for stakeholders evaluating reliable agrochemical intermediate supplier options.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of azoxystrobin intermediates has relied heavily on the ring-opening and etherification of benzofuranone (Compound A) with 4,6-dichloropyrimidine. As illustrated in the traditional reaction scheme below, early methods such as those disclosed in CN1062139A utilized sodium methoxide in methanol without any additional amine catalyst. While chemically straightforward, this approach suffers from severe inefficiencies, typically capping yields at merely 60-70%. The lack of catalytic activation leads to incomplete conversion and the generation of substantial by-products, which complicates downstream purification and increases the overall cost of goods sold. Furthermore, alternative attempts to improve kinetics using high-boiling amine catalysts like DABCO introduced new logistical nightmares. Although DABCO accelerates the reaction, its high boiling point makes recovery from the reaction matrix nearly impossible, resulting in catalyst loss and elevated ammonia nitrogen levels in the effluent. This creates a dual burden of increased raw material costs and expensive wastewater treatment protocols, rendering such processes unsustainable for large-scale commercial operations seeking cost reduction in agrochemical intermediate manufacturing.
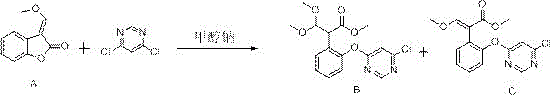
The Novel Approach
In stark contrast to these legacy methods, the invention disclosed in CN109721545B leverages the unique physicochemical properties of trimethylamine to drive the reaction forward with exceptional efficiency. As depicted in the optimized reaction pathway below, the introduction of trimethylamine—whether as a gas, a solution, or a salt—acts as a potent nucleophilic catalyst that significantly lowers the activation energy for the etherification step. This catalytic intervention allows the reaction to proceed rapidly even at mild temperatures ranging from -20°C to 30°C, preventing thermal degradation of sensitive intermediates. The true brilliance of this approach lies in the volatility of trimethylamine; unlike DABCO, it can be easily stripped from the reaction mixture and recovered from the wastewater stream. This capability transforms the catalyst from a consumable expense into a reusable asset. By integrating this catalytic system, manufacturers can achieve a mixture of Compound B and Compound C with yields consistently surpassing 85%, effectively eliminating the yield ceiling imposed by uncatalyzed routes. This represents a paradigm shift for any entity aiming to become a high-purity agrochemical intermediate supplier, as it aligns production efficiency with environmental stewardship.
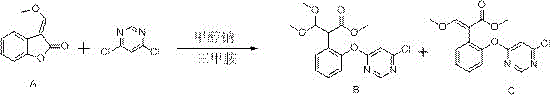
Mechanistic Insights into Trimethylamine-Catalyzed Etherification
The efficacy of the trimethylamine catalyst stems from its ability to function as a superior nucleophile compared to the alkoxide base alone. In the reaction mechanism, the lone pair of electrons on the nitrogen atom of trimethylamine attacks the electron-deficient carbon at the 4-position of the 4,6-dichloropyrimidine ring. This forms a highly reactive quaternary ammonium intermediate, which significantly enhances the electrophilicity of the pyrimidine ring towards the nucleophilic attack by the enolate derived from benzofuranone. This "activation" step is crucial because it bypasses the slower, direct displacement of the chloride ion that characterizes the uncatalyzed background reaction. The steric profile of trimethylamine is also optimal; it is small enough to access the reaction center without causing significant steric hindrance, yet bulky enough to stabilize the transition state effectively. Consequently, the reaction proceeds with high regioselectivity, favoring the formation of the desired ether linkage while suppressing side reactions such as hydrolysis or polymerization. This mechanistic precision ensures that the impurity profile of the crude product is much cleaner, reducing the load on crystallization or chromatography steps during final purification.
Furthermore, the mechanism facilitates a cleaner workup process that directly impacts the environmental footprint of the synthesis. In traditional amine-catalyzed reactions, the catalyst often ends up protonated in the acidic quench step, remaining dissolved in the aqueous phase as a non-volatile salt. However, due to the low boiling point of trimethylamine (approximately 3°C), it can be liberated from its salt form by basification and subsequently removed via nitrogen bubbling or mild heating. The patent data indicates that this recovery process can reduce the ammonia nitrogen content in the wastewater from roughly 300ppm to below 20ppm. This drastic reduction is not merely a regulatory compliance metric; it signifies a fundamental change in the mass balance of the process. By recovering over 90% of the trimethylamine and reusing it in subsequent batches, the process minimizes the introduction of foreign nitrogenous species into the ecosystem. For R&D teams focused on green chemistry metrics, this mechanism offers a textbook example of how catalyst selection can dictate the sustainability profile of an entire manufacturing campaign.
How to Synthesize Azoxystrobin Intermediates Efficiently
Implementing this catalytic technology requires precise control over reaction parameters to maximize the benefits of yield and purity. The general procedure involves charging benzofuranone and 4,6-dichloropyrimidine into a reactor containing a suitable organic solvent such as toluene. The trimethylamine catalyst is then introduced, with the molar ratio of Compound A to catalyst carefully maintained between 1:0.002 and 1:0.05. The reaction mixture is cooled to a controlled temperature, preferably between 0°C and 30°C, before the slow addition of sodium methoxide methanol solution. This dropwise addition, typically conducted over a period of 5 hours, is critical to manage the exotherm and prevent local overheating which could degrade the catalyst or the product. Following the addition, the mixture is stirred for an additional hour to ensure complete conversion. The workup involves acidification to pH 1, followed by washing and solvent removal to isolate the crude mixture of Compound B and Compound C. The detailed standardized operating procedures for scaling this reaction are provided in the technical guide below.
- Charge Compound A (benzofuranone) and 4,6-dichloropyrimidine into a reactor with toluene solvent under stirring.
- Introduce the trimethylamine catalyst (gas, solution, or salt) and cool the mixture to a temperature range of -20°C to 30°C.
- Dropwise add sodium methoxide methanol solution over 5 hours, maintain temperature, then acidify and wash to isolate the product mixture.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the trimethylamine-catalyzed process offers tangible strategic advantages that extend beyond simple yield metrics. The primary value driver is the substantial reduction in operational expenditure associated with catalyst consumption and waste disposal. Unlike high-boiling amines that are lost in every batch, the recoverability of trimethylamine means that the effective cost per kilogram of catalyst approaches zero after the initial investment. This creates a defensible cost structure that protects margins against fluctuations in raw material pricing. Additionally, the simplified wastewater profile reduces the burden on effluent treatment plants, lowering utility costs and minimizing the risk of environmental non-compliance penalties. These factors combine to create a more resilient supply chain capable of sustaining high-volume production without the bottlenecks associated with complex waste management.
- Cost Reduction in Manufacturing: The elimination of expensive, non-recoverable catalysts like DABCO results in direct material cost savings. Since the trimethylamine catalyst can be recycled with a recovery rate exceeding 90%, the recurring cost of this reagent is negligible. Furthermore, the increase in reaction yield from the typical 60-70% range to over 85% means that less starting material is required to produce the same amount of finished intermediate. This improvement in atom economy translates to significant savings on the purchase of benzofuranone and dichloropyrimidine, which are often the most expensive components of the bill of materials. The cumulative effect of higher throughput and lower input costs provides a robust foundation for competitive pricing strategies in the global agrochemical market.
- Enhanced Supply Chain Reliability: The simplicity of the catalyst recovery process ensures consistent production cycles. In methods using difficult-to-remove catalysts, equipment fouling or extended cleaning times between batches can lead to unpredictable downtime. The volatile nature of trimethylamine allows for rapid turnover of reactors, as the catalyst can be stripped and the vessel prepared for the next charge with minimal delay. This operational agility enables manufacturers to respond more quickly to surges in demand for azoxystrobin, ensuring that downstream formulation plants receive their supplies on schedule. For supply chain planners, this reliability reduces the need for excessive safety stock, freeing up working capital and warehouse space.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies waste issues, but this technology mitigates that risk through inherent design. The drastic reduction in ammonia nitrogen content simplifies the permitting process for new production lines, as the effluent is much easier to treat biologically or chemically. This environmental compatibility is increasingly critical as global regulations tighten around nitrogen discharge. Facilities adopting this method future-proof their operations against stricter environmental mandates, avoiding costly retrofits down the line. Moreover, the use of common solvents like toluene and standard reagents like sodium methoxide ensures that the supply chain for inputs remains stable and diversified, reducing the risk of single-source dependency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trimethylamine-catalyzed synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, offering clarity on why this method is superior to prior art. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer or for procurement officers negotiating supply contracts based on specific process capabilities.
Q: Why is trimethylamine preferred over DABCO for this synthesis?
A: Trimethylamine has a significantly lower boiling point than DABCO, allowing for easy recovery and reuse via distillation or stripping, whereas DABCO remains in the wastewater, increasing treatment costs and ammonia nitrogen levels.
Q: What is the expected yield improvement with this catalytic method?
A: While traditional uncatalyzed methods often struggle to maintain yields around 60-70%, the trimethylamine-catalyzed process described in CN109721545B consistently achieves yields exceeding 85%, with some embodiments reaching over 90%.
Q: How does this process impact wastewater treatment?
A: The process drastically reduces the ammonia nitrogen content in synthetic wastewater from approximately 300ppm down to below 20ppm, simplifying effluent management and ensuring compliance with stricter environmental regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azoxystrobin Intermediate Supplier
The transition to more efficient catalytic systems like the one described in CN109721545B requires a manufacturing partner with deep technical expertise and robust infrastructure. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the precise temperature controls and catalyst recovery systems necessary to exploit the full potential of this trimethylamine-mediated process. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of azoxystrobin intermediate meets the exacting standards required by global agrochemical formulators. Our commitment to process excellence ensures that we can deliver high-purity intermediates consistently, supporting your long-term production goals.
We invite you to collaborate with us to optimize your supply chain for azoxystrobin production. By leveraging our technical capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can implement this advanced synthesis technology to achieve superior economic and environmental outcomes, securing your position as a leader in the agrochemical industry.
