Advanced Synthesis of N,N'-Dimethyl Cyclohexanediamine for Commercial Scale-Up and High-Purity Applications
The chemical landscape for producing rigid urethane foam catalysts has evolved significantly with the disclosure of patent CN102993024B, which details a robust method for synthesizing N,N'-dimethyl cyclohexanediamine. This specific intermediate, also known as dimethylcyclohexylamine, serves as a critical component in the formulation of refrigerator hard bubbles, sheet materials, and spraying applications. Historically, the synthesis of such dialkyl amines relied heavily on hazardous reduction pathways, but this new intellectual property introduces a strategic shift towards a sulfonation-protection methodology. By replacing dangerous reducing agents with a sequence of sulfonation, methylation, and hydrolysis, the process achieves a product purity greater than 98% while drastically mitigating safety risks associated with traditional routes. For global procurement teams and R&D directors, this patent represents a pivotal opportunity to secure a reliable pharma intermediate supplier capable of delivering high-specification materials through a greener, more sustainable manufacturing protocol that aligns with modern environmental compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for generating N,N'-dimethyl cyclohexanediamine often depended on the reaction of cyclohexanediamine with chloroformic esters or formates, followed by a reduction step utilizing lithium aluminum hydride. This conventional approach presents severe drawbacks that hinder efficient cost reduction in fine chemical manufacturing. Lithium aluminum hydride is a potent reducing agent known for its extreme reactivity with water and air, posing significant explosion and fire hazards during large-scale operations. Furthermore, the handling of such hazardous substances requires specialized equipment, rigorous safety protocols, and expensive waste treatment procedures, all of which inflate the overall production cost. The toxicity profile of these reagents also complicates the regulatory approval process for downstream applications, particularly in sensitive sectors like pharmaceuticals or food-contact polyurethanes. Consequently, the industry has long sought an alternative pathway that eliminates these high-risk variables without compromising the structural integrity or yield of the final diamine product.
The Novel Approach
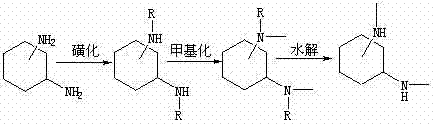
The innovative strategy outlined in the patent data circumvents these dangers by employing a protective group chemistry approach centered on aryl benzene sulfonyl chloride. Instead of forcing a difficult reduction, the process first protects the nitrogenous base of the cyclohexanediamine through a controlled sulfonation reaction. This creates a stable intermediate that can withstand subsequent alkylation conditions without degradation. The methylation step is then performed under mild conditions using common alkylating agents like methyl iodide or dimethyl sulfate in the presence of potassium carbonate. Finally, the protecting group is cleanly removed via acid hydrolysis using sulfuric acid, regenerating the free amine functionality with high selectivity. This three-step sequence not only avoids the use of pyrophoric reagents but also simplifies the purification workflow, resulting in a final product with exceptional purity levels that exceed 98%, thereby ensuring consistent performance in catalytic applications.
Mechanistic Insights into Sulfonation-Protection and Methylation Strategy
The core mechanistic advantage of this synthesis lies in the temporary masking of the nucleophilic amine centers. In the initial sulfonation stage, the cyclohexanediamine reacts with aryl benzene sulfonyl chloride in a solvent system such as pyridine or triethylamine. The temperature is strictly controlled below 60°C during the addition to prevent exothermic runaway, ensuring the formation of the N,N'-dialkyl benzene alkylsulfonyl cyclohexanediamine intermediate. This sulfonyl group acts as an electron-withdrawing protector, modulating the reactivity of the nitrogen atoms and preventing unwanted poly-alkylation or side reactions that often plague direct methylation attempts. By stabilizing the molecular framework, the process allows for precise stoichiometric control during the subsequent methylation phase, where the remaining active sites are selectively targeted.
Following the protection phase, the methylation reaction proceeds via an SN2 mechanism where the methylating reagent attacks the nitrogen center. The use of potassium carbonate as a base facilitates the deprotonation necessary for nucleophilic attack while maintaining a pH environment that preserves the sulfonamide linkage. Once the methyl groups are installed, the final hydrolysis step employs concentrated sulfuric acid at elevated temperatures ranging from 60°C to 150°C. This harsh acidic environment cleaves the robust sulfonamide bond, releasing the aryl sulfonic acid byproduct and freeing the N,N'-dimethyl cyclohexanediamine. The robustness of this mechanism ensures that impurities are minimized, as the intermediate solids can be filtered and washed prior to the final deprotection, acting as a built-in purification checkpoint that guarantees the high purity required for advanced industrial applications.
How to Synthesize N,N'-dimethyl cyclohexanediamine Efficiently
Implementing this synthesis route requires careful attention to thermal management and stoichiometry, particularly during the exothermic sulfonation and hydrolysis stages. The patent provides a clear roadmap for transitioning from laboratory bench scales to pilot plant operations, emphasizing the use of readily available solvents like acetonitrile and pyridine which facilitate easy recovery and recycling. Operators must ensure that the pH adjustment during the workup of the sulfonation step is precise, targeting a pH of 6-7 to maximize the precipitation of the intermediate solid. Detailed standard operating procedures regarding the addition rates of methyl iodide and the temperature ramping during hydrolysis are critical for maintaining safety and yield consistency across different batch sizes.
- Perform sulfonation reaction by reacting cyclohexanediamine with aryl benzene sulfonyl chloride in pyridine at controlled temperatures below 60°C to protect the nitrogenous base.
- Execute methylation by dissolving the sulfonated intermediate in acetonitrile with potassium carbonate and adding a methylating reagent like methyl iodide under ice bath conditions.
- Conduct hydrolysis by heating the methylated intermediate with sulfuric acid at 60-150°C for 4 hours, followed by neutralization and extraction to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this sulfonation-based methodology offers profound strategic benefits beyond mere technical feasibility. The elimination of lithium aluminum hydride removes a major bottleneck in raw material sourcing, as this reagent is often subject to strict transportation regulations and volatile pricing due to its hazardous classification. By switching to stable sulfonyl chlorides and common alkylating agents, manufacturers can stabilize their supply chains and reduce the administrative burden associated with handling dangerous goods. This transition directly translates to enhanced supply chain reliability, as the risk of production stoppages due to safety incidents or regulatory audits is significantly diminished. Furthermore, the simplified workup procedures, which rely on standard filtration and extraction techniques rather than complex distillation or chromatography required for removing metal residues, streamline the manufacturing timeline.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the substitution of expensive and hazardous reducing agents with cost-effective sulfonyl chlorides and bases. The removal of lithium aluminum hydride not only lowers direct material costs but also eliminates the need for specialized quenching protocols and heavy metal waste disposal services, which are notoriously expensive in the fine chemical sector. Additionally, the high purity of the crude product (>98%) reduces the load on downstream purification units, lowering energy consumption and solvent usage per kilogram of finished goods. These cumulative efficiencies result in substantial cost savings that improve the overall margin profile for producers of polyurethane catalysts and pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: From a logistics perspective, the raw materials required for this synthesis, such as tosyl chloride, pyridine, and methyl iodide, are commodity chemicals with robust global supply networks. This abundance ensures that production schedules are less vulnerable to the supply shocks that often affect specialty reagents. The non-hazardous nature of the intermediates compared to organometallic species also simplifies warehousing and transportation requirements, allowing for more flexible inventory management. Consequently, partners can expect reduced lead time for high-purity intermediates, as the manufacturing process is less prone to delays caused by safety compliance checks or the unavailability of restricted reagents.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex polymer additives, utilizing unit operations that are standard in any multipurpose chemical plant. The absence of heavy metals means the wastewater stream is easier to treat, facilitating compliance with increasingly stringent environmental regulations regarding effluent discharge. The ability to recycle solvents like acetonitrile and pyridine further enhances the sustainability profile of the operation, aligning with the ESG goals of major multinational corporations. This scalability ensures that suppliers can meet surging demand for rigid foam catalysts without requiring prohibitive capital investment in new specialized infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is crucial for R&D teams evaluating process transfer and procurement officers assessing vendor capabilities. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a factual basis for decision-making.
Q: Why is the sulfonation-methylation route preferred over lithium aluminum hydride reduction?
A: The traditional method using lithium aluminum hydride involves hazardous substances and severe toxicity risks. The patented sulfonation route eliminates the need for strong reducing agents, significantly improving operational safety and reducing raw material costs while maintaining product purity above 98%.
Q: What represents the critical quality attribute in this synthesis process?
A: The critical quality attribute is the final product purity, which consistently exceeds 98% as demonstrated in the patent embodiments. This high purity is achieved through the selective protection of the amine groups via sulfonation, preventing over-alkylation side reactions common in direct methylation attempts.
Q: Can this process be scaled for industrial polyurethane catalyst production?
A: Yes, the process is designed for industrial feasibility. It utilizes common solvents like pyridine and acetonitrile and avoids cryogenic conditions or exotic catalysts. The simple workup procedures involving filtration and extraction make it highly suitable for commercial scale-up of complex polymer additives and catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-dimethyl cyclohexanediamine Supplier
As the global demand for high-performance polyurethane catalysts continues to rise, securing a partner with deep technical expertise in amine synthesis is paramount. NINGBO INNO PHARMCHEM stands ready to leverage this advanced sulfonation-methylation technology to deliver superior quality intermediates tailored to your specific application needs. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your requirements whether you are in the clinical trial phase or full-scale industrial manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch meets the >98% purity benchmark established by the patent.
We invite you to engage with our technical procurement team to discuss how this optimized route can benefit your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic advantages of switching to this safer, more efficient manufacturing protocol. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate the technical merits of this process against your current internal standards and drive value across your organization.
