Revolutionizing Polyurethane Catalyst Production: A Safe, High-Purity Route to N,N'-Dimethyl Cyclohexanediamine
The global demand for high-performance rigid polyurethane foams, essential for insulation in refrigeration and construction, drives the critical need for efficient amine catalysts. Patent CN102993024A introduces a transformative methodology for producing N,N'-dimethyl cyclohexanediamine, a key catalyst in this sector, by fundamentally re-engineering the synthetic pathway to prioritize safety and purity. Unlike legacy processes that rely on hazardous reduction chemistry, this invention employs a sophisticated three-step sequence involving sulfonation protection, selective methylation, and subsequent hydrolysis. This strategic shift not only mitigates the severe safety risks associated with traditional reagents but also ensures a product purity exceeding 98%, addressing the stringent quality requirements of modern polymer manufacturers. For R&D directors and procurement specialists alike, this patent represents a pivotal opportunity to transition towards a more sustainable and economically viable supply chain for specialty chemical intermediates.
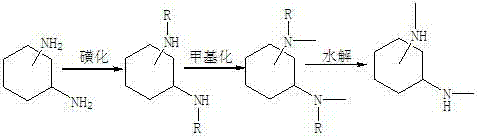
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N,N'-dimethyl cyclohexanediamine has been plagued by significant safety and economic hurdles inherent to reduction-based chemistries. The conventional route typically involves the reaction of cyclohexanediamine with chloroformates or formates, followed by a reduction step using lithium aluminum hydride (LiAlH4). This reagent is notoriously dangerous; it is pyrophoric, reacts violently with moisture, and generates substantial amounts of aluminum waste that complicates downstream processing and environmental compliance. Furthermore, the cost of lithium aluminum hydride is volatile and generally high, directly inflating the raw material costs for the final catalyst. From a supply chain perspective, the reliance on such a hazardous reagent necessitates specialized storage facilities and rigorous safety protocols, creating bottlenecks in production throughput and increasing the overall operational expenditure for manufacturers attempting to scale these processes.
The Novel Approach
The methodology disclosed in CN102993024A elegantly circumvents these challenges by replacing the dangerous reduction step with a robust protective group strategy. By first reacting cyclohexanediamine with aryl benzene sulfonyl chloride, the nitrogen atoms are effectively masked, preventing unwanted side reactions during the subsequent alkylation phase. This allows for the use of mild methylating agents such as methyl iodide or dimethyl sulfate in the presence of potassium carbonate, conditions that are far more manageable in a standard chemical plant. The final step involves a straightforward acid hydrolysis to remove the sulfonyl groups, regenerating the free amine functionality without the need for exotic reducing agents. This approach not only drastically simplifies the operational workflow but also utilizes commodity chemicals that are readily available in the global market, thereby stabilizing the supply chain and reducing the dependency on specialized, high-risk reagents.
Mechanistic Insights into Sulfonyl-Protection Mediated N-Alkylation
The core innovation of this process lies in the electronic modulation of the amine nitrogen through sulfonyl protection. In the initial sulfonation step, the electron-withdrawing nature of the sulfonyl group significantly reduces the nucleophilicity of the nitrogen atom. This deactivation is crucial because it prevents poly-alkylation, a common side reaction where excess methylating agents attack the nitrogen multiple times, leading to quaternary ammonium salts or other impurities. By carefully controlling the stoichiometry and reaction temperature (maintained below 60°C during the exothermic addition), the process ensures the formation of the N,N'-disulfonyl intermediate with high selectivity. This precise control over the electronic environment of the substrate is what enables the subsequent methylation to proceed cleanly, yielding the N,N'-dimethyl-N,N'-disulfonyl intermediate without the formation of difficult-to-separate byproducts that often plague direct alkylation attempts.
Furthermore, the hydrolysis mechanism employed in the final stage is optimized for maximum recovery and minimal degradation. The use of concentrated sulfuric acid at elevated temperatures (60-150°C) facilitates the cleavage of the strong sulfur-nitrogen bond through an acid-catalyzed pathway. This step is critical for impurity control; unlike reductive cleavage which might leave metal residues, acid hydrolysis results in water-soluble sulfonic acid byproducts that are easily separated from the organic phase during the extraction workup. The patent data indicates that this specific sequence results in a final product purity greater than 98%, demonstrating the efficacy of this mechanistic approach in suppressing impurity profiles. For quality control teams, this means a more consistent batch-to-batch performance, which is vital when the final product serves as a catalyst where trace impurities can affect the curing kinetics of polyurethane foams.
How to Synthesize N,N'-Dimethyl Cyclohexanediamine Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible roadmap for laboratory and pilot-scale production. The process begins with the dissolution of cyclohexanediamine in a solvent such as pyridine, followed by the controlled addition of the sulfonyl chloride. After isolation of the protected intermediate, the methylation is conducted in acetonitrile with potassium carbonate acting as the base scavenger. The final deprotection is achieved via thermal hydrolysis. While the general workflow is established, precise adherence to temperature gradients and stoichiometric ratios is essential for maximizing yield. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are provided in the technical guide below.
- Sulfonation: React cyclohexanediamine with aryl benzene sulfonyl chloride in pyridine at controlled temperatures (<60°C) to form the protected intermediate.
- Methylation: Treat the protected intermediate with a methylating agent (e.g., methyl iodide) and potassium carbonate in acetonitrile under ice-bath conditions.
- Hydrolysis: Cleave the sulfonyl protecting groups using sulfuric acid at elevated temperatures (60-150°C), followed by neutralization and extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this sulfonyl-protection route offers profound strategic advantages beyond mere technical feasibility. The most immediate impact is the elimination of lithium aluminum hydride from the bill of materials. This single change removes a major cost driver and a significant logistical liability, as LiAlH4 requires hazmat shipping and specialized handling infrastructure. By substituting this with common alkyl halides and sulfonic acid derivatives, the raw material basket becomes composed of widely traded commodity chemicals, insulating the production cost from the volatility associated with specialty reducing agents. This structural change in the input mix leads to a more predictable and stable cost structure, allowing for better long-term budgeting and pricing strategies for the final polyurethane catalyst.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the simplification of the reaction sequence and the substitution of expensive reagents. By avoiding the use of lithium aluminum hydride, manufacturers eliminate the high unit cost of the reductant itself, as well as the downstream costs associated with quenching and disposing of aluminum waste. Additionally, the mild conditions of the methylation step reduce energy consumption compared to high-pressure hydrogenation or extreme reduction environments. The overall process efficiency is enhanced by the high selectivity of the protection group strategy, which minimizes the loss of valuable starting materials to side products, thereby improving the effective yield per batch and lowering the cost of goods sold significantly.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by relying on a feedstock profile that utilizes globally available chemicals. Aryl benzene sulfonyl chlorides and methylating agents like methyl iodide are produced at massive scales for various industries, ensuring a steady supply even during market fluctuations. In contrast, the supply of high-purity reducing agents can be constrained by regulatory pressures and production capacity limits. By decoupling the production of N,N'-dimethyl cyclohexanediamine from these bottlenecked reagents, manufacturers can secure a more continuous production schedule, reducing lead times and ensuring reliable delivery to downstream polyurethane foam producers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is inherently superior. The absence of heavy metal residues (such as aluminum or lithium) in the final product simplifies the purification process and reduces the burden on wastewater treatment facilities. The byproducts of the hydrolysis step are sulfonic acids, which are generally easier to neutralize and manage than the complex sludge generated by hydride reductions. This alignment with green chemistry principles facilitates easier permitting for capacity expansion and reduces the risk of regulatory shutdowns. Consequently, the commercial scale-up of complex polymer additives via this route is not only technically feasible but also environmentally sustainable, meeting the increasingly strict ESG criteria of multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims presented in patent CN102993024A, providing a factual basis for decision-making. Understanding these nuances is critical for evaluating the feasibility of adopting this technology within existing manufacturing frameworks.
Q: Why is the sulfonyl protection method safer than the traditional lithium aluminum hydride reduction?
A: The traditional method relies on lithium aluminum hydride (LiAlH4), a pyrophoric and highly toxic reducing agent that poses severe safety risks during storage and handling. The patented sulfonyl protection route eliminates the need for strong reducing agents entirely, utilizing standard alkylation and hydrolysis conditions that are significantly safer for industrial operators and easier to manage in large-scale reactors.
Q: What purity levels can be achieved with this new synthesis route?
A: According to the patent data (CN102993024A), this method consistently yields N,N'-dimethyl cyclohexanediamine with a purity greater than 98%. The selective nature of the sulfonyl protection prevents over-alkylation and side reactions, resulting in a cleaner crude product that requires less intensive purification compared to reduction-based methods.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is explicitly designed for industrial feasibility. It uses common solvents like pyridine and acetonitrile and avoids hazardous reagents. The reaction conditions (temperatures below 60°C for sulfonation and moderate heating for hydrolysis) are easily controllable in standard stainless steel reactors, making the commercial scale-up of complex polymer additives straightforward and cost-effective.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-Dimethyl Cyclohexanediamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity catalysts play in the performance of advanced polymer systems. Our technical team has extensively analyzed the protective group strategies outlined in recent patents and possesses the expertise to adapt these routes for GMP-compliant and industrial-scale production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to reactor vessel is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of N,N'-dimethyl cyclohexanediamine meets the exacting standards required for rigid polyurethane foam applications.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains. By leveraging our process development capabilities, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply security.
