Advanced Synthetic Route for N4-Carbamoyl-5-Fluorocytosine Derivatives and Capecitabine Production
Advanced Synthetic Route for N4-Carbamoyl-5-Fluorocytosine Derivatives and Capecitabine Production
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of antineoplastic agents, particularly fluoropyrimidine derivatives like capecitabine. Patent CN100383128C introduces a pivotal advancement in this domain by disclosing a novel preparation method for N4-oxygen carbonyl cytosine derivatives. This technology addresses critical bottlenecks in the traditional synthesis of capecitabine, specifically focusing on the strategic timing of the carbamate introduction. By utilizing N4-pentyloxycarbonyl-5-fluorocytosine as a foundational building block, the process streamlines the overall synthetic pathway, offering a compelling alternative for manufacturers aiming to optimize their production lines for high-purity pharmaceutical intermediates.
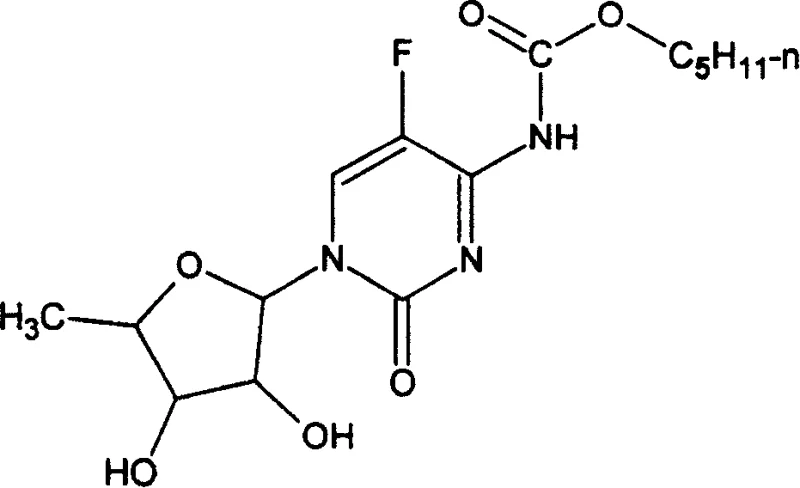
This patent represents a significant shift from conventional multi-step protection strategies to a more direct condensation approach. The core innovation lies in the stability and reactivity of the N4-carbamoyl intermediate, which serves as a versatile precursor not only for capecitabine but potentially for a broader range of cytosine aminoformate-type antitumor medicines. For R&D directors and process chemists, understanding the nuances of this route provides a clear pathway to enhancing process efficiency while maintaining stringent quality standards required for oncology therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of capecitabine and related nucleoside analogs has been plagued by complex protection and deprotection sequences that inflate both cost and environmental footprint. Traditional routes, such as those referenced in prior art like US005476932, often involve synthesizing the nucleoside first and then attempting to selectively protect the exocyclic amine at the N4 position. This late-stage functionalization is notoriously difficult due to the presence of multiple reactive hydroxyl groups on the sugar moiety, leading to regioselectivity issues and the formation of difficult-to-remove impurities.
Furthermore, conventional methods frequently rely on harsh reaction conditions or expensive reagents to achieve the necessary selectivity, which complicates scale-up and poses safety risks in a commercial manufacturing setting. The accumulation of byproducts from these inefficient steps necessitates extensive purification protocols, such as repeated chromatography or recrystallization, which drastically reduce overall yield and throughput. For procurement managers, these inefficiencies translate into higher raw material costs and longer lead times, creating vulnerabilities in the supply chain for critical cancer medications.
The Novel Approach
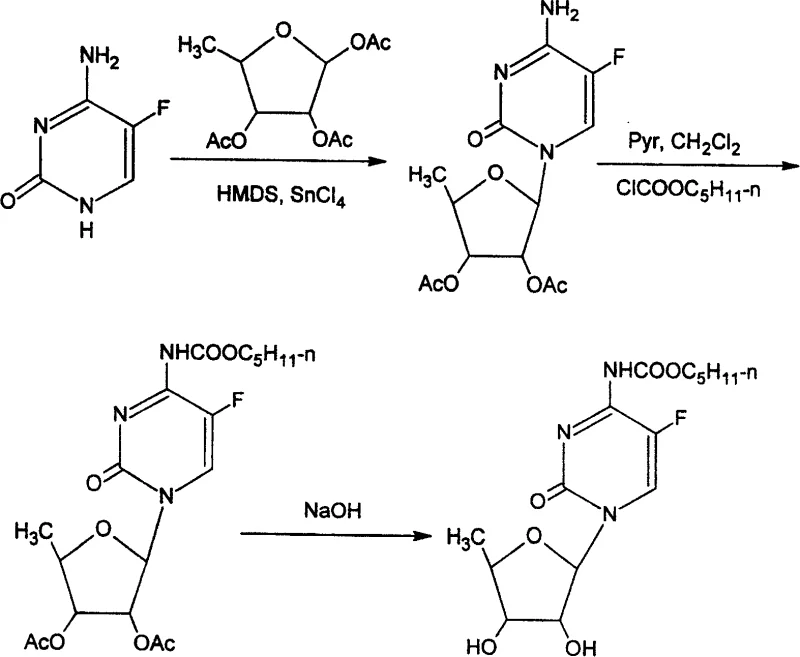
In stark contrast, the methodology outlined in CN100383128C reverses the logical order of synthesis by pre-installing the carbamate functionality on the cytosine base before glycosylation. This strategy effectively 'masks' the reactive amine, preventing it from interfering with the subsequent coupling reaction. The novel approach utilizes a condensation reaction between the N4-carbamoyl-5-fluorocytosine derivative and a protected ribofuranose compound, catalyzed by Lewis acids such as tin tetrachloride. This ensures that the glycosidic bond forms exclusively at the N1 position with high fidelity.
By shifting the carbamate introduction to the beginning of the sequence, the process eliminates the need for post-glycosylation amination and protection steps. This results in a significantly shorter synthetic route with fewer unit operations. The use of mild reaction temperatures, typically ranging from -20°C to 20°C, further enhances the operational safety and energy efficiency of the process. For supply chain heads, this simplification means a more reliable production schedule with reduced dependency on specialized reagents that might face market volatility.
Mechanistic Insights into SnCl4-Catalyzed Glycosylation
The heart of this synthetic innovation is the Lewis acid-catalyzed glycosylation mechanism, which leverages the unique electronic properties of the silylated intermediate. In the preferred embodiment, the N4-pentyloxycarbonyl-5-fluorocytosine is first treated with hexamethyldisilazane (HMDS) to generate a trimethylsilyl-protected species in situ. This silylation increases the lipophilicity of the base and activates the N1 nitrogen for nucleophilic attack. When combined with 5-deoxy-1,2,3-tri-O-acetyl-D-ribofuranose in the presence of SnCl4, the Lewis acid coordinates with the anomeric acetate, generating a highly reactive oxocarbenium ion intermediate.
The nucleophilic attack by the silylated base on this oxocarbenium ion proceeds with high stereoselectivity, favoring the formation of the beta-anomer required for biological activity. The presence of the carbamate group at the N4 position exerts a subtle electronic influence that stabilizes the transition state, minimizing the formation of N3-glycosylated byproducts which are common impurities in uncatalyzed or poorly catalyzed reactions. This mechanistic precision is crucial for R&D teams focused on impurity profiling, as it ensures that the crude product already possesses a high degree of purity before final workup.
Furthermore, the reaction conditions described allow for precise control over the stoichiometry, with a molar ratio of base to sugar ranging from 1:1.05 to 1:1.20. This slight excess of the sugar donor drives the equilibrium towards product formation without leading to significant waste. The subsequent workup involves a careful quenching with sodium bicarbonate solution, which neutralizes the acidic catalyst and removes silyl byproducts. This gentle aqueous workup is compatible with large-scale reactors and avoids the use of hazardous quenching agents, aligning with modern green chemistry principles.
How to Synthesize N4-Carbamoyl-5-Fluorocytosine Efficiently
The practical implementation of this technology relies on a straightforward three-stage protocol that can be adapted for both pilot and commercial scales. The process begins with the formation of the key carbamate intermediate, followed by the coupling reaction, and concludes with a deprotection step to yield the final active pharmaceutical ingredient. Each stage has been optimized in the patent examples to maximize yield and minimize processing time, providing a clear roadmap for process engineers looking to adopt this technology.
- Condense 5-fluorocytosine with n-amyl chloroformate in the presence of pyridine at 0°C to form N4-pentyloxycarbonyl-5-fluorocytosine.
- Protect the base with hexamethyldisilazane (HMDS) and couple with 5-deoxy-1,2,3-tri-O-acetyl-D-ribofuranose using SnCl4 catalyst.
- Perform ammonolysis in methanol to remove acetyl protecting groups and yield the final capecitabine product.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting the synthetic route described in CN100383128C offers tangible benefits that extend beyond the laboratory, directly impacting the bottom line and operational resilience of pharmaceutical manufacturers. By fundamentally restructuring the synthesis to eliminate redundant steps, companies can achieve substantial cost savings in raw material consumption and waste disposal. The reduction in process complexity also lowers the barrier to entry for scaling up production, allowing suppliers to respond more agilely to market demand fluctuations for capecitabine and its intermediates.
- Cost Reduction in Manufacturing: The elimination of late-stage protection and deprotection cycles removes the need for expensive reagents and solvents associated with those specific transformations. By consolidating the synthesis into fewer steps, the overall consumption of utilities such as heating, cooling, and agitation is drastically reduced. Furthermore, the high yields reported in the patent embodiments suggest that less starting material is required to produce the same amount of final product, directly lowering the cost of goods sold (COGS) without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials like 5-fluorocytosine and n-amyl chloroformate mitigates the risk of supply disruptions often associated with exotic or highly specialized reagents. The robustness of the reaction conditions, which tolerate a broad temperature range and utilize common solvents like dichloromethane and toluene, ensures that production can continue uninterrupted even under varying facility conditions. This stability is critical for maintaining continuous supply contracts with downstream API manufacturers.
- Scalability and Environmental Compliance: The process generates significantly less chemical waste due to the atom-economic nature of the condensation reaction and the avoidance of multiple purification stages. The use of tin tetrachloride, while requiring careful handling, is a well-understood industrial catalyst that can be effectively managed and recycled in modern facilities. This alignment with environmental regulations reduces the compliance burden and potential fines, making the operation more sustainable and socially responsible in the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making.
Q: What is the primary advantage of using N4-carbamoyl-5-fluorocytosine as a starting material?
A: Using the N4-carbamoyl derivative as the initial raw material eliminates the need for late-stage protection of the exocyclic amine, significantly simplifying the synthetic sequence and reducing the generation of hazardous waste associated with additional protection-deprotection cycles.
Q: Which catalyst system is recommended for the glycosylation step in this patent?
A: The patent specifies the use of tin tetrachloride (SnCl4) in conjunction with hexamethyldisilazane (HMDS) for silylation, which facilitates high-yield coupling under mild conditions compared to traditional heavy metal catalysts.
Q: How does this method impact the purity profile of the final API intermediate?
A: By introducing the carbamate group early in the synthesis, the method minimizes side reactions at the N4 position during glycosylation, resulting in a cleaner impurity profile and facilitating easier purification via crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Capecitabine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic routes like the one detailed in CN100383128C for securing a competitive edge in the oncology market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless and efficient. We are committed to delivering high-purity N4-carbamoyl-5-fluorocytosine derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation.
We invite global partners to collaborate with us to leverage this technology for their own supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply chain security.
