Advanced Catalytic Synthesis of Capecitabine Intermediates for Commercial Scale-Up
Advanced Catalytic Synthesis of Capecitabine Intermediates for Commercial Scale-Up
The global demand for effective antineoplastic agents continues to drive innovation in the synthesis of fluoropyrimidine carbamates, with Capecitabine remaining a cornerstone therapy for breast and colorectal cancers. A significant technological breakthrough in this domain is detailed in patent CN111100172A, which introduces a novel 5-deoxy-D-ribofuranose 1-[2-(1-styryl) benzoate] derivative. This specific intermediate serves as a highly efficient glycosyl donor, fundamentally altering the landscape of nucleoside assembly by enabling activation under exceptionally mild conditions. Unlike traditional methodologies that rely on harsh reagents, this new approach utilizes a catalytic system comprising trimethylsilyl trifluoromethanesulfonate (TMSOTf) and N-iodosuccinimide (NIS). The structural elegance of this donor molecule facilitates a highly selective coupling reaction, ensuring that the sensitive cytosine base remains intact while achieving superior stereocontrol. For pharmaceutical manufacturers, this represents a pivotal shift towards greener, more cost-effective production of high-purity API intermediates.
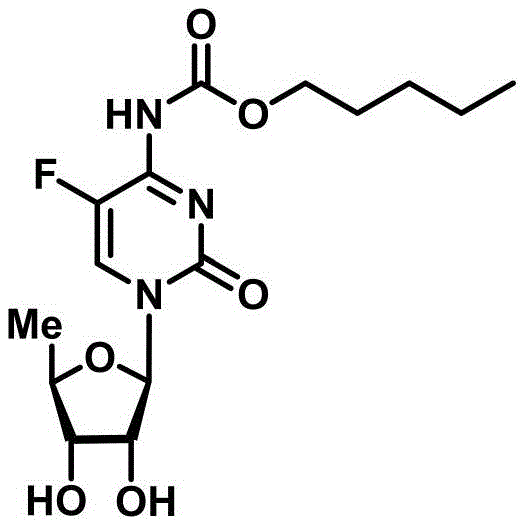
The strategic value of this patent lies not just in the molecule itself, but in the robustness of the synthetic pathway it enables. By replacing stoichiometric quantities of Lewis acids with a catalytic regime, the process drastically reduces the chemical load on the reaction system. This reduction is critical for maintaining the integrity of complex nucleoside structures, which are prone to decomposition under acidic stress. Furthermore, the versatility of the acyl protecting groups (such as benzoyl or acetyl) allows for fine-tuning of the solubility and reactivity profiles, making the process adaptable to various scale-up requirements. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances is essential for delivering consistent quality in the competitive oncology market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Capecitabine and related nucleoside analogues has been plagued by significant chemical inefficiencies and operational hazards. Conventional routes, such as those depicted in prior art schemes (e.g., Schemes 1-4 in the background of the patent), often necessitate the use of equivalent or excessive amounts of strong Lewis acids to activate the sugar donor. These harsh conditions frequently lead to the degradation of the pyrimidine base, resulting in complex impurity profiles that are difficult and expensive to purge during downstream purification. Moreover, the generation of stoichiometric metal waste poses substantial environmental compliance challenges and increases the overall cost of goods sold (COGS). The reliance on aggressive reagents also limits the scalability of these processes, as heat management and safety protocols become increasingly stringent at larger volumes. Consequently, manufacturers face prolonged lead times and reduced yields, creating bottlenecks in the supply chain for critical cancer therapies.
The Novel Approach
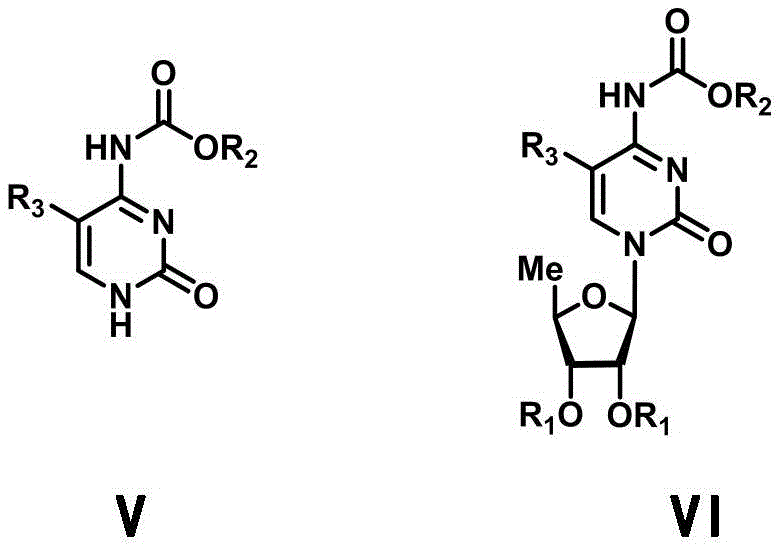
In stark contrast, the methodology disclosed in CN111100172A offers a refined solution through the deployment of the 5-deoxy-D-ribofuranose 1-[2-(1-styryl) benzoate] derivative. This innovative donor is uniquely designed to be activated by merely catalytic amounts of Lewis acid in conjunction with NIS, creating a much gentler reaction environment. The presence of the styryl benzoate moiety enhances the leaving group ability, allowing the glycosylation to proceed efficiently at temperatures ranging from 0°C to room temperature. This mildness preserves the structural fidelity of the N4-oxycarbonyl-5-cytosine acceptor, minimizing side reactions such as hydrolysis or rearrangement. The result is a streamlined process that delivers high-purity intermediates with exceptional stereochemical control, specifically favoring the desired beta-anomer. This approach not only simplifies the purification workflow but also aligns with modern principles of green chemistry by reducing reagent consumption and waste generation.
Mechanistic Insights into Catalytic Glycosylation
The core of this technological advancement lies in the unique activation mechanism of the styryl benzoate donor. In the presence of the catalytic promoter system (TMSOTf/NIS), the double bond of the styryl group undergoes electrophilic activation, generating a highly reactive oxocarbenium ion intermediate. This transient species is perfectly poised for nucleophilic attack by the silylated cytosine base. The use of a catalytic amount of TMSOTf is particularly ingenious; it regenerates during the reaction cycle, meaning that only a fraction of the reagent is required compared to traditional stoichiometric methods. This catalytic turnover significantly lowers the acidity of the bulk medium, thereby protecting the acid-labile carbamate protection on the cytosine ring. Furthermore, the neighboring group participation from the 2-position ester (if configured correctly) or the steric bulk of the protecting groups aids in directing the incoming nucleophile to the beta-face, ensuring high anomeric selectivity. This level of control is paramount for R&D teams focused on minimizing chiral impurities that could compromise the biological activity of the final drug.
Impurity control is further enhanced by the stability of the intermediate species formed during the reaction. Traditional methods often suffer from the formation of alpha-anomers or hydrolyzed sugar byproducts due to the prolonged exposure to strong acids. However, the rapid kinetics of this catalytic system allow the reaction to reach completion quickly, typically within 2 hours at room temperature, as demonstrated in the patent examples. The subsequent quenching with triethylamine effectively neutralizes any residual acidity, preventing post-reaction degradation. This clean reaction profile translates directly into a simplified workup procedure, where standard silica gel chromatography is sufficient to isolate the product in high purity. For process chemists, this means fewer unit operations, reduced solvent usage, and a more robust process that is less sensitive to minor variations in reaction parameters, ultimately leading to a more reliable supply of high-purity pharmaceutical intermediates.
How to Synthesize Capecitabine Efficiently
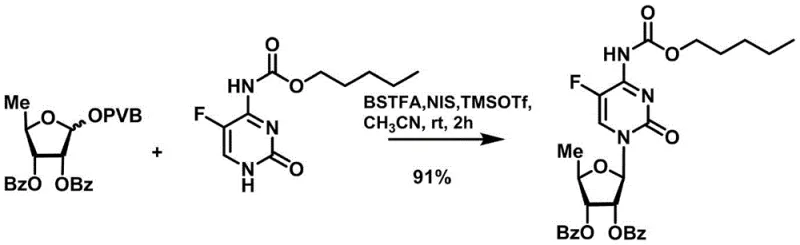
The synthesis of Capecitabine via this novel route is a testament to the power of rational molecular design in process chemistry. The protocol begins with the preparation of the specialized sugar donor, followed by the key glycosylation step, and concludes with a straightforward deprotection sequence. This modular approach allows for the isolation and characterization of intermediates, providing multiple quality control checkpoints throughout the manufacturing campaign. The use of common organic solvents such as dichloromethane and acetonitrile ensures that the process is compatible with existing infrastructure in most multipurpose chemical plants. Detailed standard operating procedures for each stage, including precise temperature controls and reagent addition rates, are critical to replicating the high yields reported in the patent literature. For a comprehensive breakdown of the experimental conditions and stoichiometry required to achieve these results, please refer to the standardized synthesis guide below.
- Preparation of the 5-deoxy-D-ribofuranose 1-[2-(1-styryl) benzoate] donor via thioglycoside formation and subsequent esterification.
- Coupling the sugar donor with N4-oxycarbonyl-5-cytosine using catalytic TMSOTf and NIS under mild conditions.
- Final deprotection of the benzoyl groups using sodium hydroxide in methanol to yield the active pharmaceutical ingredient.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic glycosylation technology offers tangible strategic benefits that extend beyond simple yield improvements. The shift from stoichiometric to catalytic reagent usage fundamentally alters the cost structure of the synthesis. By eliminating the need for large quantities of expensive Lewis acids and the associated waste disposal costs, the overall manufacturing expense is significantly reduced. This cost efficiency is compounded by the higher purity of the crude product, which reduces the burden on purification resources and shortens the overall production cycle time. Furthermore, the mild reaction conditions enhance operational safety, lowering the risk profile associated with handling hazardous chemicals on a multi-ton scale. These factors collectively contribute to a more resilient and cost-competitive supply chain for oncology APIs.
- Cost Reduction in Manufacturing: The transition to a catalytic system eliminates the procurement and disposal costs associated with stoichiometric Lewis acids, which are often expensive and generate significant hazardous waste. This reduction in reagent intensity directly lowers the variable cost per kilogram of the intermediate. Additionally, the high selectivity of the reaction minimizes the formation of hard-to-remove impurities, thereby reducing the consumption of chromatography media and solvents during purification. The cumulative effect is a substantial decrease in the cost of goods sold, allowing for more competitive pricing in the generic pharmaceutical market without compromising margin.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance and avoiding production delays. The use of stable, commercially available starting materials mitigates the risk of raw material shortages that can plague more exotic synthetic pathways. Moreover, the mild conditions reduce the likelihood of runaway reactions or equipment corrosion, leading to higher asset utilization rates and fewer unplanned maintenance shutdowns. This reliability translates into shorter lead times for customers and a more predictable delivery schedule for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard unit operations and common solvents, facilitating a smooth transition from pilot plant to commercial production. The reduction in chemical waste aligns with increasingly stringent environmental regulations, simplifying the permitting process and reducing the carbon footprint of the manufacturing site. By adopting this greener chemistry, companies can enhance their corporate social responsibility profiles while simultaneously improving operational efficiency. This dual benefit makes the technology an attractive option for long-term strategic partnerships in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios. The answers provided reflect the current state of the art as described in the intellectual property, offering a clear perspective on the capabilities and limitations of the technology.
Q: What is the primary advantage of the new glycosyl donor in patent CN111100172A?
A: The 5-deoxy-D-ribofuranose 1-[2-(1-styryl) benzoate] derivative allows for activation using only catalytic amounts of Lewis acid (TMSOTf) and NIS, avoiding the harsh conditions and waste associated with stoichiometric promoters.
Q: How does this method improve impurity control compared to traditional routes?
A: By operating under mild conditions (0°C to room temperature) and avoiding excess Lewis acids, the reaction minimizes side reactions and degradation of the sensitive cytosine base, resulting in a cleaner crude product profile.
Q: Is this synthetic route suitable for large-scale manufacturing?
A: Yes, the process utilizes common solvents like dichloromethane and acetonitrile, and the high yields reported (e.g., 91% for glycosylation) indicate excellent atom economy and scalability for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Capecitabine Intermediate Supplier
The technological potential of the 5-deoxy-D-ribofuranose 1-[2-(1-styryl) benzoate] derivative represents a significant opportunity for optimizing the production of Capecitabine. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust commercial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of catalytic activation and stereocontrol is maintained at every scale. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for oncology drug manufacturing. Our commitment to quality assurance ensures that our clients receive materials that facilitate seamless downstream processing and regulatory approval.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume and cost requirements. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this catalytic method. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about your supply chain strategy. Together, we can accelerate the availability of life-saving medications while driving efficiency and sustainability in pharmaceutical manufacturing.
