Advanced Synthesis of Flexible Ester-containing Diaminofluorene for Next-Gen Polymer Applications
Advanced Synthesis of Flexible Ester-containing Diaminofluorene for Next-Gen Polymer Applications
The development of high-performance polymer materials often hinges on the precise engineering of monomer structures to balance conflicting properties such as thermal stability and mechanical toughness. Patent CN102617382B introduces a significant breakthrough in this domain by disclosing a robust preparation method for ester group-containing diaminofluorene derivatives. These novel compounds serve as critical intermediates for curing agents in epoxy resins, polyimides, and benzoxazines, addressing the longstanding industry challenge where increasing cross-linking density for heat resistance typically results in brittle materials. By strategically incorporating ester linkages into the fluorene backbone, this technology enables the production of polymers that maintain excellent thermal performance while exhibiting vastly improved flexibility and impact resistance. For R&D directors and procurement specialists in the advanced materials sector, understanding the synthetic pathway and commercial viability of these monomers is essential for securing a competitive edge in aerospace, electronics, and coating applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional diamine monomers based on the fluorene skeleton, such as 9,9-bis(4-aminophenyl)fluorene, have long been valued for their rigidity and thermal stability. However, this inherent rigidity presents a significant drawback in high-performance applications where mechanical toughness is paramount. When used as curing agents for epoxy resins or precursors for polyimides, these conventional rigid molecules create highly cross-linked networks that are prone to brittleness and cracking under stress. Attempts to mitigate this by adding external toughening agents often compromise the thermal properties of the final matrix, creating a trade-off that limits material performance in extreme environments like aerospace or electronic packaging. Furthermore, the synthesis of modified fluorene diamines often involves complex multi-step sequences or harsh conditions that can lead to inconsistent impurity profiles, complicating the downstream polymerization process and affecting the dielectric properties of the final electronic materials.
The Novel Approach
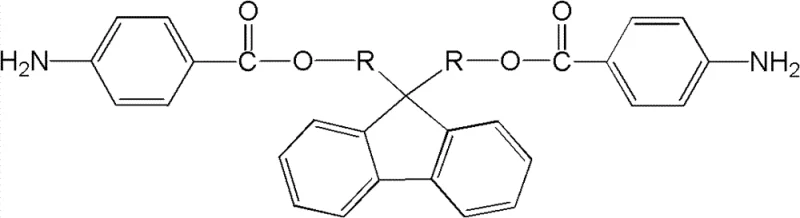
The methodology outlined in the patent offers a transformative solution by integrating flexible ester groups directly into the molecular architecture of the diamine monomer. This approach utilizes a straightforward two-step synthesis starting from readily available dihydroxyfluorene derivatives. The first step involves an acylation reaction with p-nitrobenzoyl chloride to form a dinitro intermediate, followed by a catalytic reduction to yield the target diamino compound. This structural modification introduces rotational freedom into the polymer backbone via the ester linkage, effectively acting as an internal plasticizer that enhances toughness without the need for external additives. Consequently, the resulting polymers exhibit a superior balance of properties, making them ideal candidates for next-generation composite materials and high-reliability coatings where both durability and thermal endurance are non-negotiable requirements.
Mechanistic Insights into Esterification and Hydrazine Reduction
The core of this synthesis lies in the efficient formation of the ester bond followed by the selective reduction of nitro groups. In the first stage, the reaction between the hydroxyl groups of the fluorene derivative and p-nitrobenzoyl chloride is facilitated by a base such as triethylamine. This acylation proceeds initially at low temperatures, specifically between -10°C and 10°C, to control the exothermic nature of the reaction and minimize side products. Following the initial addition, the mixture is allowed to react at room temperature and subsequently heated to reflux for an extended period of 10 to 24 hours. This prolonged heating ensures complete conversion of the hydroxyl groups to ester linkages, driving the equilibrium towards the dinitro intermediate. The use of solvents like anhydrous chloroform, tetrahydrofuran, or dioxane provides an optimal medium for dissolving both the bulky fluorene substrate and the acyl chloride, ensuring homogeneous reaction kinetics.
The second critical phase involves the reduction of the nitro groups to primary amines using hydrazine hydrate. This transformation is catalyzed by transition metal systems, with the patent highlighting the efficacy of Palladium on Carbon (Pd/C) as well as iron-based catalysts like FeCl3 or Fe(OH)3. The reaction is conducted in absolute ethanol under reflux conditions for 24 to 48 hours. The choice of catalyst offers strategic flexibility; while Pd/C provides high activity, iron salts present a cost-effective alternative that avoids the use of precious metals, which is a significant consideration for large-scale manufacturing. The mechanism likely involves the transfer of hydrogen from hydrazine to the nitro group on the catalyst surface, sequentially reducing the nitrogen oxidation state until the amine is formed. This method is particularly advantageous as it avoids the use of high-pressure hydrogenation equipment, thereby simplifying the safety profile and equipment requirements for commercial production facilities.
How to Synthesize Ester-containing Diaminofluorene Efficiently
The synthesis protocol described in the patent provides a clear roadmap for producing these high-value monomers with consistent quality. The process begins with the precise stoichiometric mixing of dihydroxyfluorene and p-nitrobenzoyl chloride in a molar ratio ranging from 1:2 to 1:2.2, ensuring a slight excess of the acylating agent to drive the reaction to completion. Temperature control is vital during the initial addition to prevent thermal runaway, after which the system is heated to reflux to maximize yield. The subsequent reduction step requires careful monitoring of the hydrazine addition rate and maintaining the reflux temperature to ensure full conversion of the nitro intermediates. Detailed standardized operating procedures for scaling this chemistry from laboratory benchtop to pilot plant reactors are essential for maintaining product purity and batch-to-batch consistency.
- React dihydroxyfluorene derivatives with p-nitrobenzoyl chloride in organic solvent with triethylamine at low temperature, followed by reflux to form the dinitro intermediate.
- Reduce the dinitro intermediate using hydrazine hydrate and a catalyst (Pd/C or Iron salts) in ethanol under reflux conditions to obtain the final diamino product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this synthesis route offers distinct logistical and economic benefits over traditional methods for producing toughened diamine monomers. The raw materials required, specifically various dihydroxyfluorenes and p-nitrobenzoyl chloride, are commercially available commodity chemicals with stable supply chains, reducing the risk of raw material shortages. Furthermore, the reaction conditions utilize standard organic solvents and atmospheric pressure reflux, eliminating the need for specialized high-pressure hydrogenation reactors which are capital intensive and require rigorous safety maintenance. This simplification of the process equipment directly translates to lower barrier-to-entry for manufacturing and reduced operational expenditures. The ability to choose between precious metal catalysts and abundant iron salts allows procurement managers to optimize cost structures based on current market prices for palladium, providing a hedge against volatile metal markets.
- Cost Reduction in Manufacturing: The elimination of high-pressure hydrogenation equipment significantly lowers capital expenditure requirements for production facilities. Additionally, the option to use iron-based catalysts instead of palladium reduces the recurring cost of catalyst consumption and simplifies the downstream removal of heavy metal residues, which often requires expensive purification steps. The high yields reported in the patent examples, often exceeding 80 percent for the intermediate and substantial yields for the final amine, indicate an efficient atom economy that minimizes waste disposal costs and maximizes output per batch.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as chloroform, ethanol, and triethylamine ensures that production is not bottlenecked by exotic reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and times without catastrophic failure, contributes to high process reliability and consistent on-time delivery. This stability is crucial for downstream polymer manufacturers who operate on tight just-in-time schedules and cannot afford interruptions in their monomer supply due to complex synthesis failures.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-ton industrial production without fundamental changes to the chemistry. The use of hydrazine hydrate, while requiring careful handling, avoids the generation of large volumes of acidic or basic aqueous waste streams associated with some other reduction methods like metal-acid reductions. The ability to recover and recycle solvents like chloroform or dioxane further aligns the process with modern environmental regulations and sustainability goals, reducing the overall carbon footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these ester-containing diaminofluorene derivatives. These answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for feasibility assessments. Understanding these specifics helps stakeholders evaluate the fit of this technology within their existing manufacturing frameworks and product portfolios.
Q: How does the ester group improve polymer performance?
A: The introduction of ester linkages increases molecular chain flexibility, significantly enhancing the toughness of the resulting cured epoxy or polyimide without drastically compromising thermal stability.
Q: What catalysts are suitable for the reduction step?
A: The patent specifies Palladium on Carbon (Pd/C), Ferric Chloride (FeCl3), or Ferric Hydroxide (Fe(OH)3) as effective catalysts for the hydrazine reduction of the nitro groups.
Q: Can this method be scaled for industrial production?
A: Yes, the process utilizes common solvents like chloroform or THF and standard reflux conditions, making it highly adaptable for commercial scale-up from kilogram to tonnage levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ester-containing Diaminofluorene Supplier
The technological potential of ester-containing diaminofluorene represents a significant opportunity for manufacturers of high-performance polymers seeking to differentiate their products through superior mechanical properties. NINGBO INNO PHARMCHEM stands ready to support this innovation with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with the necessary infrastructure to handle the specific solvent systems and reflux conditions required for this synthesis, ensuring stringent purity specifications are met for every batch. With our rigorous QC labs and commitment to process optimization, we can deliver high-purity diaminofluorene derivatives that meet the exacting standards of the aerospace and electronic materials industries.
We invite potential partners to engage with our technical team to discuss how this novel monomer can enhance your polymer formulations. By requesting a Customized Cost-Saving Analysis, you can quantify the economic benefits of switching to this more efficient synthesis route. We encourage you to contact our technical procurement team to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of these advanced materials into your supply chain.
