Advanced Synthesis of Flexible Ester-Containing Diaminofluorene for High-Performance Polymers
The rapid evolution of high-performance materials in aerospace, electronics, and advanced composites demands monomers that offer a precise balance between thermal stability and mechanical toughness. Patent CN102617382B introduces a groundbreaking class of ester group-containing diaminofluorenes that effectively resolve the longstanding trade-off between rigidity and flexibility in polymer networks. By strategically incorporating ester linkages into the 9,9-fluorene scaffold, this technology enables the synthesis of resins with superior dielectric properties and enhanced processability compared to traditional rigid analogues. For R&D directors and procurement specialists seeking reliable polymer additive supplier solutions, this synthetic route represents a significant leap forward in material science capability. The methodology outlined in the patent provides a robust framework for producing these specialized intermediates with high reproducibility and purity, ensuring consistent performance in downstream polymerization applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional fluorene-based diamines, such as 9,9-bis(4-aminophenyl)fluorene, have long been valued for their exceptional thermal resistance and mechanical strength in polyimide and epoxy formulations. However, the inherent rigidity of the direct carbon-carbon or ether linkages in these conventional monomers often results in polymer networks that are excessively brittle and prone to cracking under thermal stress or mechanical impact. This lack of molecular flexibility limits their utility in applications requiring high toughness, such as flexible circuit boards or impact-resistant composite coatings. Furthermore, modifying these rigid structures often requires complex multi-step syntheses involving expensive reagents or harsh conditions that are difficult to scale safely. The inability to fine-tune the chain flexibility without sacrificing thermal performance has been a persistent bottleneck in the development of next-generation high-performance resins for demanding industrial environments.
The Novel Approach
The innovative strategy presented in this patent overcomes these limitations by introducing ester functional groups as flexible spacers within the fluorene architecture. This structural modification allows for greater rotational freedom within the polymer chain, significantly improving toughness and impact resistance while maintaining the high glass transition temperatures characteristic of the fluorene moiety. The versatility of this approach is demonstrated by the ability to vary the linker group R, which can be adjusted from simple alkyl chains to more complex phenoxy structures to fine-tune material properties.  . This adaptability makes the process highly valuable for cost reduction in polymer additive manufacturing, as it allows producers to select starting materials based on availability and cost without compromising the core functionality of the final monomer. The result is a new generation of curing agents that offer a superior balance of properties for advanced composite and electronic material applications.
. This adaptability makes the process highly valuable for cost reduction in polymer additive manufacturing, as it allows producers to select starting materials based on availability and cost without compromising the core functionality of the final monomer. The result is a new generation of curing agents that offer a superior balance of properties for advanced composite and electronic material applications.
Mechanistic Insights into Esterification and Hydrazine Reduction
The synthesis mechanism relies on a carefully controlled two-step sequence beginning with the acylation of dihydroxyfluorene derivatives. In the first stage, the hydroxyl groups of the fluorene precursor react with p-nitrobenzoyl chloride in the presence of a base like triethylamine to form the ester linkage. This reaction is initiated at low temperatures ranging from -10 to 10°C to control exothermicity and prevent side reactions, followed by a prolonged reflux period of 10 to 24 hours to drive the conversion to completion. The use of anhydrous solvents such as chloroform or tetrahydrofuran is critical to prevent hydrolysis of the acid chloride, ensuring high yields of the dinitro intermediate which typically exceed 85% in optimized examples. This precise control over reaction conditions is essential for minimizing impurities that could affect the color and electrical properties of the final polyimide or epoxy resin.
The second critical phase involves the reduction of the nitro groups to primary amines using hydrazine hydrate as the reducing agent. This transformation is facilitated by a catalyst, which can be selected from noble metals like palladium on carbon or more cost-effective iron salts such as FeCl3 or Fe(OH)3. The reaction proceeds under reflux conditions in ethanol for 24 to 48 hours, allowing for the complete conversion of the nitro functionality to the amine without affecting the sensitive ester bonds. 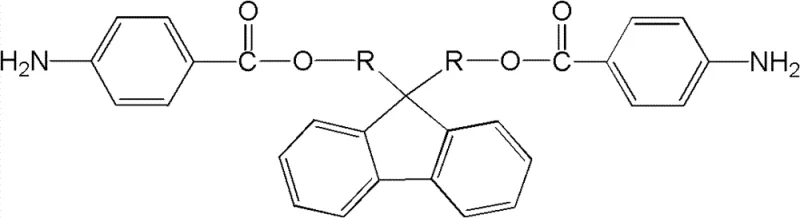 . The choice of catalyst offers a strategic advantage for supply chain managers, as iron-based catalysts provide a lower-cost alternative to precious metals while still delivering high purity white solid products. The final isolation involves filtration to remove the catalyst followed by solvent removal and vacuum drying, yielding the target diaminofluorene with the structural integrity required for high-performance polymer synthesis.
. The choice of catalyst offers a strategic advantage for supply chain managers, as iron-based catalysts provide a lower-cost alternative to precious metals while still delivering high purity white solid products. The final isolation involves filtration to remove the catalyst followed by solvent removal and vacuum drying, yielding the target diaminofluorene with the structural integrity required for high-performance polymer synthesis.
How to Synthesize 9,9-Bis(4-aminobenzoate)fluorene Efficiently
Executing this synthesis requires strict adherence to temperature profiles and stoichiometric ratios to ensure safety and maximize yield. The process begins with the preparation of the dinitro precursor, where maintaining the initial low temperature is vital for controlling the reaction rate of the acid chloride. Following the acylation, the reduction step demands careful monitoring of the hydrazine addition to manage gas evolution and maintain reaction stability. Detailed standard operating procedures for scaling this pathway from laboratory to production are essential for maintaining quality consistency.
- Perform acylation of dihydroxyfluorene with p-nitrobenzoyl chloride at -10 to 10°C, followed by reflux to obtain the dinitro intermediate.
- Conduct catalytic reduction of the dinitro intermediate using hydrazine hydrate and a metal catalyst (Pd/C or Iron salts) under reflux conditions.
- Purify the final white solid product through filtration, solvent removal, and vacuum drying to ensure high purity for polymer applications.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits for organizations focused on optimizing their supply chain for high-purity polymer additives. The reliance on readily available commodity chemicals such as p-nitrobenzoyl chloride and hydrazine hydrate reduces dependency on exotic or single-source reagents, thereby enhancing supply security. Additionally, the flexibility in catalyst selection allows procurement teams to pivot between precious metal and base metal catalysts based on market pricing volatility, providing a buffer against raw material cost fluctuations. The robust nature of the reaction conditions also implies a lower risk of batch failure, contributing to more predictable production schedules and reliable delivery timelines for downstream customers.
- Cost Reduction in Manufacturing: The ability to utilize iron-based catalysts like FeCl3 instead of expensive palladium systems significantly lowers the operational expenditure associated with catalyst procurement and recovery. Eliminating the need for complex purification steps to remove trace heavy metals further simplifies the downstream processing workflow, reducing both time and utility costs. The high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted as byproducts, which directly translates to improved gross margins for large-scale production runs. This economic efficiency makes the technology particularly attractive for cost reduction in electronic chemical manufacturing where margin pressure is high.
- Enhanced Supply Chain Reliability: The starting materials, including various dihydroxyfluorenes and p-nitrobenzoyl chloride, are established industrial chemicals with stable global supply chains. This reduces the risk of production stoppages due to raw material shortages, a critical factor for maintaining continuous operation in high-volume facilities. Furthermore, the reaction tolerates a range of solvent choices including chloroform, THF, and dioxane, allowing manufacturers to adapt to regional solvent availability or regulatory restrictions without revalidating the entire process. This adaptability ensures reducing lead time for high-purity polymer additives by minimizing logistical bottlenecks.
- Scalability and Environmental Compliance: The process operates at atmospheric pressure and utilizes standard reflux equipment, making it highly amenable to commercial scale-up of complex polymer additives without requiring specialized high-pressure reactors. The use of hydrazine hydrate, while requiring careful handling, avoids the generation of heavy metal waste streams associated with other reduction methods, simplifying wastewater treatment protocols. The solid nature of the final product facilitates easy storage and transport, reducing the hazards and costs associated with shipping liquid intermediates. These factors collectively support a more sustainable and scalable manufacturing model that aligns with modern environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized fluorene derivatives. Understanding these details is crucial for integrating this material into your existing formulation workflows effectively.
Q: How does the ester group improve the performance of fluorene-based polymers?
A: The introduction of ester groups into the fluorene backbone significantly enhances molecular flexibility without substantially compromising thermal stability, addressing the brittleness issues common in rigid diamine monomers.
Q: What catalysts are suitable for the reduction step in this synthesis?
A: The process supports versatile catalytic systems including Palladium on Carbon (Pd/C), Ferric Chloride (FeCl3), or Ferric Hydroxide (Fe(OH)3), allowing manufacturers to optimize for cost or specific reaction kinetics.
Q: What are the primary industrial applications of these diaminofluorene derivatives?
A: These compounds serve as critical curing agents for high-performance epoxy resins, amine sources for polyimide synthesis, and precursors for benzoxazine resins used in aerospace and electronic packaging.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9,9-Bis(4-aminobenzoate)fluorene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced monomers play in the development of next-generation high-performance materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ester-containing diaminofluorene meets the exacting standards required for aerospace and electronic applications. Our commitment to quality ensures that the thermal and mechanical properties of your final resin systems remain consistent and reliable.
We invite you to collaborate with us to leverage this innovative technology for your specific product needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to help you accelerate your development timeline and secure a competitive advantage in the global polymer market.
