Advanced Synthesis of Tenofovir Alafenamide Intermediates: A Scalable Commercial Route
Advanced Synthesis of Tenofovir Alafenamide Intermediates: A Scalable Commercial Route
The global demand for potent antiviral agents continues to drive innovation in the synthesis of nucleotide reverse transcriptase inhibitors, specifically focusing on the efficient production of Tenofovir Alafenamide (TAF). A pivotal development in this sector is detailed in patent CN106946935B, which discloses a novel preparation method for diastereoisomeric nucleoside derivatives. This technology addresses a critical bottleneck in the supply chain of TAF, a prodrug widely used for treating HIV and Hepatitis B. The patent outlines a robust chemical pathway that transforms a racemic precursor into a highly selective diastereomer, thereby streamlining the manufacturing process for this essential active pharmaceutical ingredient (API). By leveraging thermal configuration conversion, this method offers a distinct advantage over traditional resolution techniques, promising enhanced purity and operational simplicity for industrial-scale production.
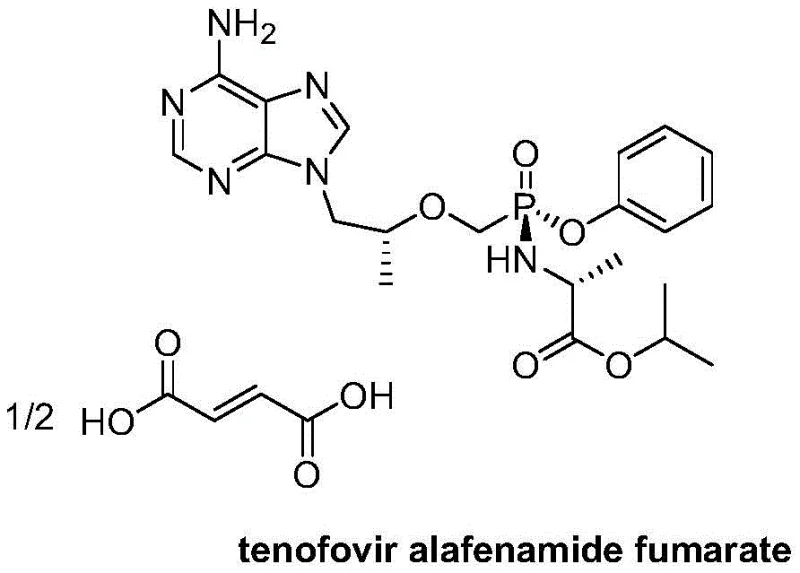
Tenofovir alafenamide fumarate represents a significant advancement in antiviral therapy due to its improved plasma stability and intracellular delivery compared to earlier generations. However, the synthesis of its key intermediates poses substantial challenges, particularly regarding stereochemical control at the phosphorus atom. The molecule contains a chiral center at the phosphorus, leading to the formation of diastereoisomers. Historically, separating these isomers has been a resource-intensive endeavor, often requiring complex chromatographic separations that hinder scalability. The technology described in CN106946935B provides a strategic solution by enabling the direct conversion of the less desirable racemic form into the therapeutically active diastereomer through a controlled thermal process. This breakthrough not only simplifies the synthetic route but also aligns with the industry's push towards greener and more cost-effective manufacturing practices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing the key intermediate Compound I have historically suffered from significant inefficiencies that impact both cost and throughput. Traditional approaches often rely on the synthesis of a racemic mixture at the phosphorus center, followed by arduous separation processes to isolate the desired diastereomer. Literature references such as WO2015/40640A2 and WO2015/107451A2 describe routes where diastereoisomer selectivity is almost non-existent during the initial synthesis, necessitating downstream purification steps that are technically demanding. Furthermore, existing conversion methods disclosed in documents like US 2013/0090473 A1 often require specific solvent systems like tetrahydrofuran or dichloromethane, which can present safety and environmental hazards on a large scale. These conventional techniques frequently result in lower overall yields, sometimes falling below 50%, and generate substantial chemical waste, creating a burden on waste management systems and increasing the carbon footprint of the manufacturing process.
The Novel Approach
In stark contrast, the novel approach presented in the patent introduces a streamlined thermal epimerization strategy that dramatically improves process efficiency. By heating Compound III, which acts as a racemate, in specific organic solvents, the configuration at the phosphorus center is selectively converted to yield Compound I with high diastereoisomeric purity. This method eliminates the need for expensive chiral resolving agents or complex column chromatography, which are often the most costly and time-consuming steps in fine chemical synthesis. The process utilizes mild reaction conditions, typically ranging from 60°C to 75°C, and employs solvents such as methyl cyclopentyl ether and n-heptane, which are safer and more environmentally benign than chlorinated alternatives. This shift allows for a direct transformation that achieves yields greater than 80%, representing a substantial improvement over the less than 50% yields typical of older technologies. The simplicity of the work-up procedure, often involving direct evaporation and recrystallization, further underscores the operational advantages of this new methodology.
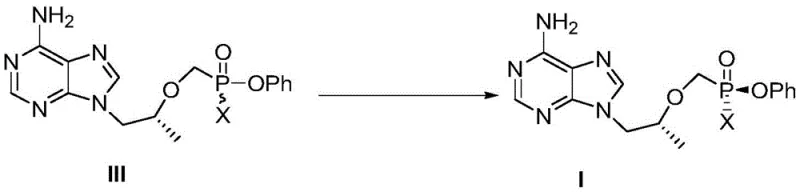
Mechanistic Insights into Thermal Diastereomer Conversion
The core of this technological advancement lies in the thermal instability of the phosphorus-chlorine (or bromine) bond in Compound III under specific solvent conditions, which facilitates a reversible dissociation-recombination mechanism. When Compound III is heated in solvents like methyl cyclopentyl ether, the energy input allows for the temporary breaking of the bond between the phosphorus atom and the leaving group X (where X is a halogen such as chlorine or bromine). This generates a transient planar phosphorus intermediate that is achiral. Upon recombination with the halogen or interaction with the solvent environment, the phosphorus center reforms, but statistically favors the thermodynamically more stable diastereomer, which is Compound I. This dynamic equilibrium is driven by the subtle steric and electronic differences between the two diastereomers, where Compound I possesses a lower energy state in the given solvent matrix. The presence of the halogen atom is crucial, as it acts as a leaving group that enables this configurational lability without degrading the sensitive nucleoside backbone.
Controlling impurities in this reaction is paramount, and the patent details a rigorous monitoring protocol using Nuclear Magnetic Resonance (NMR) spectroscopy to ensure high fidelity. The reaction progress is tracked by sampling the mixture at regular intervals, typically every two hours, to monitor the ratio of Compound I to Compound III. The process is terminated once the content of Compound I exceeds 95%, ensuring that the equilibrium has shifted sufficiently towards the desired product. This precise endpoint determination prevents over-reaction or degradation of the product, which could occur if the thermal treatment were prolonged unnecessarily. Additionally, the choice of solvent plays a critical role in impurity control; solvents like n-heptane and methyl cyclopentyl ether not only facilitate the conversion but also aid in the subsequent purification by allowing for effective recrystallization. This dual function of the solvent system minimizes the introduction of foreign contaminants and simplifies the isolation of the final high-purity solid.
How to Synthesize Compound I Efficiently
The synthesis of Compound I is executed through a logical two-stage sequence that begins with the halogenation of the hydroxyl precursor (Compound II) followed by the thermal conversion step. This workflow is designed for maximum operational ease, utilizing standard reactor equipment and readily available reagents. The initial halogenation activates the phosphorus center, setting the stage for the subsequent stereochemical adjustment. Detailed standardized synthesis steps for this high-efficiency route are provided in the guide below.
- Dissolve compound II in an organic solvent such as methyl cyclopentyl ether and react with a halogenating reagent like thionyl chloride at 50-75°C to produce compound III.
- Suspend the resulting compound III in a solvent system like methyl cyclopentyl ether or n-heptane and heat the mixture to 60-75°C with stirring.
- Monitor the reaction progress via NMR until the content of compound I exceeds 95%, then purify the crude product through recrystallization to obtain the final white solid powder.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic benefits that extend beyond mere technical feasibility. The shift from low-yielding, chromatography-dependent methods to a high-yield thermal conversion process fundamentally alters the cost structure of producing Tenofovir Alafenamide intermediates. By removing the dependency on specialized chiral separation columns and reducing the number of unit operations, manufacturers can achieve significant cost reduction in API manufacturing. The elimination of these bottlenecks allows for faster batch turnover times and reduces the capital expenditure required for specialized purification equipment. Furthermore, the use of common, recoverable solvents like n-heptane and methyl cyclopentyl ether simplifies solvent management and recovery protocols, contributing to a leaner and more agile supply chain operation.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic increase in yield and the simplification of the purification train. Traditional methods often suffer from yields below 50% due to losses during chiral resolution, effectively doubling the raw material cost per kilogram of final product. In contrast, this new method consistently delivers yields exceeding 80%, meaning that nearly double the amount of product can be generated from the same quantity of starting materials. Additionally, the avoidance of expensive chiral stationary phases and the reduction in solvent consumption for chromatography lead to substantial operational savings. The ability to use cheaper halogenating reagents like thionyl chloride, combined with the high atom economy of the thermal conversion, ensures that the cost of goods sold (COGS) is minimized, providing a competitive edge in the global market for antiviral intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is a critical concern for pharmaceutical manufacturers, and this process enhances reliability by relying on robust, commodity-grade raw materials. The reagents required, such as thionyl chloride and common organic solvents, are widely available from multiple global suppliers, reducing the risk of supply disruptions associated with niche or proprietary chemicals. The simplicity of the reaction conditions—requiring only heating and stirring without the need for cryogenic temperatures or inert atmosphere complexities beyond standard nitrogen protection—makes the process highly transferable across different manufacturing sites. This flexibility allows for multi-sourcing strategies and geographic diversification of production, ensuring that the supply of this critical intermediate remains uninterrupted even in the face of regional logistical challenges or raw material shortages.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden complexities, but this thermal conversion method is inherently scalable due to its reliance on simple physical parameters like temperature and time. The absence of exothermic hazards associated with rapid reagent addition and the use of high-boiling, stable solvents make the process safe to operate in large-scale reactors. From an environmental perspective, the reduction in waste generation is significant; by avoiding the silica gel and solvent mixtures used in column chromatography, the volume of hazardous solid waste is drastically reduced. The process aligns with green chemistry principles by maximizing atom efficiency and minimizing the use of auxiliary substances, thereby facilitating easier regulatory approval and reducing the environmental compliance burden on the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation to provide clarity on the process capabilities.
Q: What is the primary advantage of this new preparation method over prior art?
A: The primary advantage is the ability to convert the racemic mixture (Compound III) into the desired diastereomer (Compound I) with high selectivity and yield (>80%) using simple thermal treatment, eliminating the need for difficult chiral column chromatography.
Q: Which solvents are preferred for the thermal conversion step?
A: The patent specifies that organic solvents such as toluene, n-heptane, and methyl cyclopentyl ether, either individually or in combination, are highly effective. Methyl cyclopentyl ether is particularly preferred for achieving optimal yields.
Q: How does this process impact environmental compliance?
A: This process utilizes milder reaction conditions and avoids highly toxic reagents often found in alternative synthetic routes. The use of recoverable solvents and the generation of less waste contribute to a more environmentally compliant manufacturing profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tenofovir Alafenamide Intermediate Supplier
As the pharmaceutical industry continues to evolve, the need for efficient and scalable synthesis routes for complex molecules like Tenofovir Alafenamide becomes increasingly critical. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced technologies such as the thermal diastereomer conversion described in CN106946935B to deliver superior value to our partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global supply chains. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of intermediate meets the highest international standards for safety and efficacy.
We invite you to explore how our optimized manufacturing capabilities can enhance your supply chain resilience and cost efficiency. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to bring life-saving medications to patients faster and more affordably through innovative chemical engineering.
