Advanced Manufacturing of Lenalidomide Intermediates Using Safe Transfer Hydrogenation Technology
The pharmaceutical industry constantly seeks robust and safe synthetic routes for critical oncology drugs, and patent CN103242215A presents a transformative approach to manufacturing the key intermediate for Lenalidomide. This patent specifically addresses the preparation of N-(4-amino-1-oxo-1,3-dihydro-2H-isoindol-2-yl)-L-glutamine methyl ester, a pivotal building block in the synthesis of the multiple myeloma treatment Revlimid. Traditional methods have long relied on hazardous high-pressure hydrogenation, which introduces significant safety risks and operational complexities. By shifting to a transfer hydrogenation protocol using ammonium formate or formic acid, this innovation allows the reaction to proceed rapidly at normal temperature and pressure. This breakthrough not only enhances operator safety by avoiding explosive hydrogen gas but also drastically simplifies the post-treatment process, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks seeking efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in US20060052609, typically employ molecular hydrogen as the reducing agent for converting the nitro precursor to the amino intermediate. This conventional approach necessitates the use of specialized high-pressure autoclaves operating at approximately 30 psi for extended periods, often ranging from three to four hours. The reaction involves a complex three-phase system where gaseous hydrogen, solid palladium-carbon catalyst, and liquid reactants must interact efficiently, which is inherently difficult to control. Furthermore, the storage and handling of large volumes of hydrogen gas pose severe safety hazards, including potential explosions, requiring expensive safety infrastructure and rigorous monitoring. These factors collectively increase the capital expenditure and operational costs, creating bottlenecks in the cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the novel method described in the patent utilizes ammonium formate or formic acid as a safe and effective hydrogen donor, completely eliminating the need for external hydrogen gas supply. This transfer hydrogenation strategy enables the reaction to occur under mild conditions, specifically at ambient temperatures between 20°C and 30°C and at atmospheric pressure. The operational simplicity is profound, as the reaction can be conducted in standard glass-lined or stainless steel reactors without the need for pressure-rated vessels. The workup procedure is equally streamlined, involving simple filtration to remove the catalyst followed by solvent evaporation and crystallization. This shift represents a paradigm change in commercial scale-up of complex pharmaceutical intermediates, offering a pathway that is both economically superior and environmentally friendlier.
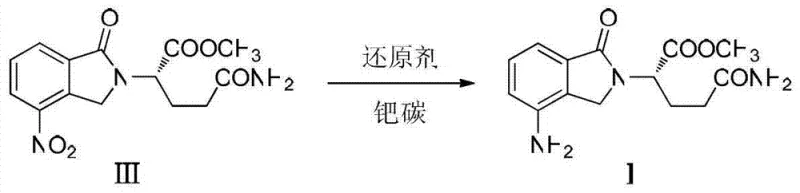
Mechanistic Insights into Transfer Hydrogenation Reduction
The core of this technological advancement lies in the mechanism of transfer hydrogenation facilitated by the palladium on carbon (Pd/C) catalyst. In this system, ammonium formate acts as the source of hydrogen, decomposing on the surface of the palladium catalyst to generate active hydrogen species in situ. These active species are immediately available to reduce the nitro group (-NO2) on the isoindoline ring to the corresponding amino group (-NH2) without generating free hydrogen gas bubbles that could lead to mass transfer limitations. The mild thermal conditions prevent the thermal degradation of sensitive functional groups, such as the ester and amide moieties present in the glutamine side chain. This precise control over the reduction environment ensures that the stereochemistry of the L-glutamine moiety is preserved, which is critical for the biological activity of the final drug product.
Furthermore, the selectivity of this reduction method plays a crucial role in impurity control and overall product quality. High-pressure hydrogenation can sometimes lead to over-reduction or hydrogenolysis of protecting groups, such as the benzyl carbamate if not carefully managed, although in this specific route the nitro reduction is the primary focus. By avoiding the harsh conditions associated with high-pressure hydrogen, the formation of by-products is significantly minimized, leading to a cleaner reaction profile. The subsequent crystallization step, performed by adding water to the concentrated residue at low temperatures (0°C to 5°C), further purifies the product by exploiting solubility differences. This results in a high-purity pharmaceutical intermediate with a defined melting point and consistent spectral characteristics, meeting the stringent requirements of global regulatory bodies.
How to Synthesize N-(4-amino-1-oxo-1,3-dihydro-2H-isoindol-2-yl)-L-glutamine methyl ester Efficiently
The synthesis begins with L-glutamine, which undergoes protection and esterification to form the necessary precursor, followed by cyclization with a nitro-substituted benzoate to yield the nitro-intermediate (Compound III). The critical step involves dissolving Compound III in methanol and adding the Pd/C catalyst along with ammonium formate, stirring at room temperature until the reaction is complete as monitored by TLC. After filtration of the catalyst and removal of the solvent, the crude product is crystallized from water to obtain the pure amino intermediate (Compound I), which can then be cyclized to form Lenalidomide. For detailed operational parameters and stoichiometry, refer to the standardized guide below.
- Protect L-Glutamine with Cbz-Cl and esterify to form the methyl ester precursor.
- Perform cyclization with 2-bromoethyl-3-nitrobenzoate to generate the nitro-intermediate (Compound III).
- Reduce the nitro group to an amino group using ammonium formate and Pd/C at ambient temperature to yield the final intermediate (Compound I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this transfer hydrogenation technology offers compelling strategic advantages that go beyond mere technical feasibility. The elimination of high-pressure hydrogen gas removes a major logistical and safety burden, allowing manufacturing to take place in facilities that may not be equipped with specialized high-pressure infrastructure. This flexibility significantly broadens the base of potential contract manufacturing organizations (CMOs) capable of producing this intermediate, thereby enhancing supply chain resilience and reducing the risk of single-source dependency. Additionally, the use of common, inexpensive reagents like ammonium formate instead of compressed hydrogen cylinders contributes to a more stable and predictable cost structure for long-term production contracts.
- Cost Reduction in Manufacturing: The transition to ambient pressure processing eliminates the need for expensive autoclaves and the associated maintenance and certification costs required for high-pressure equipment. By removing the requirement for specialized high-pressure reactors, manufacturers can utilize standard agitation tanks, which represent a substantially lower capital investment and reduced depreciation costs over time. Furthermore, the simplified workup procedure reduces labor hours and energy consumption associated with heating and pressurizing reactors, leading to significant operational expenditure savings. These cumulative efficiencies translate directly into a more competitive pricing model for the final active pharmaceutical ingredient without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on ammonium formate as a hydrogen donor mitigates the supply risks associated with industrial hydrogen gas, which can be subject to regional availability fluctuations and strict transportation regulations. The reagents used in this process are commodity chemicals with robust global supply chains, ensuring consistent availability even during market disruptions. This stability allows for better production planning and inventory management, reducing the likelihood of delays caused by raw material shortages. Consequently, pharmaceutical companies can maintain a steady flow of intermediates, ensuring uninterrupted production of the final life-saving medication for patients.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the engineering challenges of scaling up high-pressure gas-liquid reactions, which often suffer from poor mass transfer at larger volumes. Operating at atmospheric pressure simplifies the scale-up trajectory from pilot plant to commercial production, reducing the time-to-market for new generic versions or process improvements. From an environmental perspective, the method generates less hazardous waste and avoids the carbon footprint associated with the production and transport of high-pressure hydrogen, aligning with modern green chemistry principles and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. Understanding these details is essential for R&D teams evaluating process transfers and procurement officers negotiating supply agreements. The answers are derived directly from the experimental data and technical specifications provided in the patent literature to ensure accuracy and reliability.
Q: What are the safety advantages of using ammonium formate over hydrogen gas?
A: Using ammonium formate eliminates the need for high-pressure hydrogen gas and autoclaves, significantly reducing explosion risks and simplifying reactor requirements.
Q: Does this method affect the purity of the Lenalidomide intermediate?
A: Yes, the mild reaction conditions (ambient temperature and pressure) help minimize side reactions and degradation, resulting in high-purity products with yields exceeding 90%.
Q: Is this process scalable for industrial production?
A: Absolutely. The process uses standard atmospheric reactors and common solvents like methanol, making it highly scalable and easier to implement in existing manufacturing facilities compared to high-pressure hydrogenation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lenalidomide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to ensure the consistent supply of high-quality oncology intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this transfer hydrogenation technology are fully realized at an industrial level. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of N-(4-amino-1-oxo-1,3-dihydro-2H-isoindol-2-yl)-L-glutamine methyl ester meets the highest international standards for safety and efficacy.
We invite global partners to collaborate with us to leverage this cost-effective and safe manufacturing route for your supply chain needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can drive value and security for your pharmaceutical projects.
