Scaling High-Purity Atazanavir Intermediates via Novel Non-Polar Epoxidation Technology
The pharmaceutical industry continuously seeks robust synthetic routes for critical antiretroviral agents, particularly for HIV protease inhibitors like Atazanavir. Patent CN103044361A introduces a transformative preparation method for (2R,3S)-epoxidation amino-benzene butane, a pivotal chiral intermediate in this therapeutic class. Traditional synthesis pathways have long struggled with solvent inefficiencies and yield losses during the critical epoxidation step. This new technology addresses these bottlenecks by shifting from water-soluble polar solvents to a non-polar solvent system, fundamentally altering the crystallization and isolation dynamics. For R&D Directors and Procurement Managers, this represents a significant opportunity to enhance the supply chain reliability of high-purity pharmaceutical intermediates. By optimizing the reaction medium, the process not only improves molar yields to over 96% but also ensures product purity exceeds 99.5%, meeting the stringent specifications required for downstream API manufacturing. This technical breakthrough underscores the importance of selecting a reliable pharmaceutical intermediate supplier who understands the nuances of solvent engineering and process intensification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
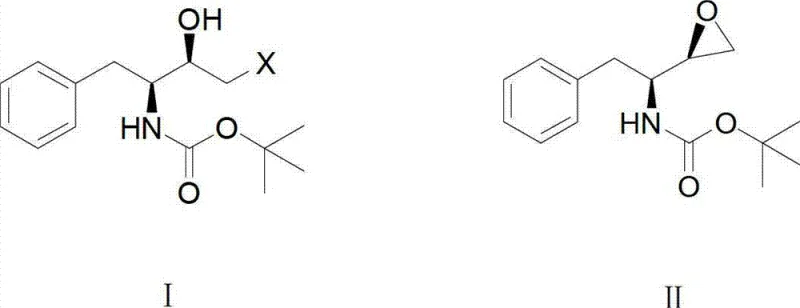
Historically, the synthesis of (2R,3S)-3-t-butoxycarbonyl amino-1,2-epoxy-4-phenyl butane has relied heavily on polar solvents that are miscible with water, as disclosed in prior art such as US6344572B1 and EP1215209A1. In these conventional protocols, the reaction mixture requires the addition of substantial volumes of water to induce crystallization, a step that inherently compromises the overall mass balance. Because the product possesses a degree of solubility in aqueous environments, significant amounts of the valuable intermediate are lost to the mother liquor during the precipitation phase. Furthermore, the resulting filter cake retains high moisture content due to the hydrophilic nature of the solvent system, necessitating energy-intensive drying processes that can potentially degrade heat-sensitive chiral centers. These inefficiencies accumulate to increase the cost reduction in API manufacturing challenges, as more raw material is required to compensate for yield losses, and utility costs for drying and wastewater treatment escalate disproportionately.
The Novel Approach
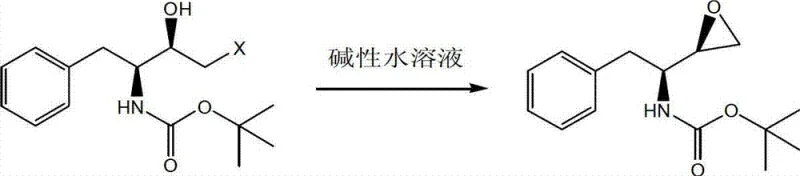
The innovative method described in CN103044361A circumvents these issues by utilizing a non-polar solvent system, such as petroleum ether, hexane, or cyclohexane, for the cyclization reaction. In this regime, the reaction proceeds smoothly under the influence of an alkaline aqueous solution, but crucially, the organic and aqueous phases remain immiscible. Upon completion of the reaction, the aqueous phase containing the base and byproducts is simply separated via standing and layering, leaving the product dissolved in the organic phase. Crystallization is then achieved by cooling the organic layer, eliminating the need for water dilution. This approach ensures that the product remains almost entirely within the organic phase, drastically minimizing solubility losses. Additionally, the use of volatile non-polar solvents facilitates rapid and efficient drying at lower temperatures, preserving the stereochemical integrity of the molecule while significantly lowering energy consumption and operational complexity.
Mechanistic Insights into Non-Polar Solvent Cyclization
The core of this synthetic advancement lies in the intramolecular nucleophilic substitution that forms the epoxide ring. The starting material, (2R,3S)-3-t-butoxycarbonyl amino-1-halogen-2-hydroxy-4-phenyl butane, undergoes deprotonation of the hydroxyl group by the alkaline aqueous phase at the interface. The resulting alkoxide ion attacks the adjacent carbon bearing the halogen leaving group, displacing it to form the strained three-membered epoxide ring. In a polar solvent system, the transition state is highly solvated, which can sometimes lead to side reactions or slower kinetics due to hydrogen bonding networks. However, in the non-polar medium described in this patent, the reaction interface is sharply defined. The addition of a minor amount of polar co-solvent, such as acetone or ethanol, in a specific volume ratio (e.g., 1:4 to 1:8 relative to the non-polar solvent) acts as a phase transfer mediator. This fine-tunes the polarity at the interface, accelerating the ring-closure reaction rate without compromising the phase separation benefits. This precise control over the reaction environment is critical for maintaining the high diastereomeric purity required for HIV drug synthesis.
Impurity control is another critical aspect where this mechanism excels. In traditional water-based crystallization, impurities with similar polarities often co-precipitate with the product, requiring extensive recrystallization steps to achieve the necessary purity levels. In contrast, the non-polar solvent system exploits the differential solubility of the product versus polar impurities. Since the product is highly soluble in the non-polar organic phase but insoluble in the aqueous waste phase, polar byproducts and inorganic salts are effectively sequestered in the aqueous layer during the separation step. The subsequent cooling crystallization from the organic phase further purifies the material, as the solubility curve of the product in non-polar solvents is steep, allowing for high recovery of pure crystals while leaving trace impurities in the mother liquor. This inherent purification capability ensures that the final product consistently achieves purity levels greater than 99.5%, reducing the burden on downstream quality control labs.
How to Synthesize (2R,3S)-Epoxidation Amino-Benzene Butane Efficiently
The operational protocol for this synthesis is designed for industrial robustness, beginning with the dissolution of the halogenated precursor in a selected non-polar solvent like petroleum ether or cyclohexane. A controlled amount of polar co-solvent may be added to optimize reaction kinetics, followed by the slow addition of an alkaline aqueous solution, such as sodium hydroxide or potassium carbonate, while maintaining the temperature between 0°C and 40°C. The detailed standardized synthesis steps see the guide below for precise parameters regarding stoichiometry and agitation rates.
- Dissolve (2R,3S)-3-t-butoxycarbonyl amino-1-halogen-2-hydroxy-4-phenyl butane in a non-polar solvent such as petroleum ether or hexane.
- Add an alkaline aqueous solution (e.g., NaOH or KOH) at 0°C to 40°C to initiate the intramolecular cyclization reaction.
- Separate the organic phase, cool to crystallize the product, and dry under vacuum to obtain >99.5% purity material.
Commercial Advantages for Procurement and Supply Chain Teams
For Supply Chain Heads and Procurement Managers, the transition to this non-polar solvent methodology offers tangible strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of the isolation process. By eliminating the need for large volumes of water for crystallization, the volume of wastewater generated is significantly reduced, lowering environmental compliance costs and waste treatment burdens. Furthermore, the solvents used, such as hexane or petroleum ether, are immiscible with water and can be easily recovered and recycled from the mother liquor through distillation. This closed-loop solvent management system translates to substantial cost savings in raw material procurement over the long term. The reduced drying time and lower temperature requirements also decrease utility consumption, contributing to a lower overall cost of goods sold (COGS) for this critical intermediate.
- Cost Reduction in Manufacturing: The elimination of water-soluble solvents removes the need for energy-intensive evaporation steps to remove water from the product cake. Since the product crystallizes directly from the organic phase, the residual solvent is volatile and easily removed under mild vacuum conditions. This efficiency means that manufacturing facilities can process larger batches in the same amount of time without expanding drying capacity. Additionally, the high molar yield of over 96% means less starting material is wasted, directly improving the material cost efficiency. The ability to recycle the non-polar solvent further amplifies these savings, making the process economically superior to traditional polar solvent routes.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including the halogenated precursor and common non-polar solvents, are readily available in the global chemical market. Unlike specialized polar solvents that may face supply constraints, petroleum ether and hexane are commodity chemicals with stable pricing and availability. This ensures reducing lead time for high-purity pharmaceutical intermediates, as production schedules are less likely to be disrupted by raw material shortages. The robustness of the reaction conditions, which tolerate a range of temperatures (0°C to 40°C), also means that the process is less sensitive to minor fluctuations in cooling or heating utility performance, ensuring consistent batch-to-batch output.
- Scalability and Environmental Compliance: Scaling up this reaction is straightforward because it avoids the handling of large volumes of aqueous waste streams that require complex treatment before discharge. The phase separation step is easily scalable in standard stirred-tank reactors, and the crystallization by cooling is a unit operation well-understood in chemical engineering. The reduced environmental footprint, characterized by lower wastewater generation and solvent recycling, aligns with modern green chemistry principles and regulatory expectations. This makes the commercial scale-up of complex pharmaceutical intermediates smoother, with fewer regulatory hurdles related to environmental impact assessments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel epoxidation technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a factual basis for decision-making.
Q: How does the non-polar solvent method improve yield compared to traditional polar solvent methods?
A: Traditional methods use water-soluble polar solvents requiring large amounts of water for crystallization, which dissolves some product and reduces yield. The non-polar method allows phase separation where the product remains in the organic layer, minimizing loss and achieving yields over 96%.
Q: What are the drying advantages of this new epoxidation process?
A: Conventional processes leave products wet with water, requiring high-energy drying. This novel method crystallizes from organic solvents, resulting in a filter cake that is easier to dry at lower temperatures, significantly reducing energy consumption and thermal degradation risks.
Q: Is this process suitable for large-scale commercial production of HIV drug intermediates?
A: Yes, the process uses readily available raw materials and common non-polar solvents like petroleum ether. The simplified workflow, involving direct phase separation and cooling crystallization, is highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2R,3S)-Epoxidation Amino-Benzene Butane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of antiretroviral therapies. Our technical team has extensively evaluated the non-polar solvent cyclization route described in CN103044361A and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this innovative process are realized at an industrial level. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of (2R,3S)-epoxidation amino-benzene butane meets the exacting standards required for API synthesis. We are committed to delivering consistent quality and supply continuity for our partners in the pharmaceutical sector.
We invite procurement leaders and technical directors to collaborate with us on optimizing their supply chains for HIV drug intermediates. By leveraging our expertise in process chemistry, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can ensure a stable, cost-effective, and high-quality supply of this essential chiral building block, supporting the continued availability of life-saving medications worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
